Abstract
INTRODUCTION
This study investigated whether heavy metal levels were higher in people from Taiwan as compared to those from Western countries.
METHODS
We measured the level of heavy metals (lead, mercury, arsenic and cadmium) in the blood of 40 apparently healthy adults. Since mercury does not respond to ethylenediaminetetraacetic acid (EDTA) infusion, only urine lead, arsenic and cadmium levels were determined by applying the body burden concept after calcium disodium EDTA infusion. These three heavy metals were extracted from daily urine samples for three consecutive days.
RESULTS
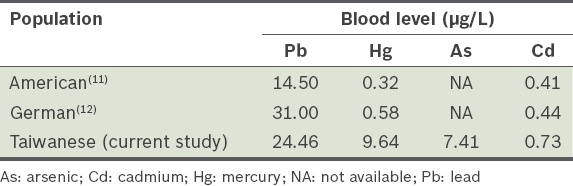
The mean blood lead, mercury and cadmium levels of the Taiwanese individuals (24.46 ± 9.69 µg/L, 9.64 ± 6.98 µg/L and 0.73 ± 0.27 µg/L, respectively) were greater than those of the Americans. The Taiwanese also had greater blood mercury and cadmium levels than the Germans. The first-day urine lead, arsenic and cadmium levels were 77.9%, 33.1% and 62.4%, respectively, of the total lead, arsenic and cadmium excretion during the three days. This indicates that the first-day urine lead and cadmium excretion represented most (> 60%) of the lead and cadmium excretion in those three days.
CONCLUSION
This study demonstrated that the Taiwanese population has higher blood mercury and cadmium levels than Western populations. To study the urine lead and cadmium body burden of patients, detection of first-day, rather than three-day, urine lead and cadmium levels can be done, as the former yields results that are fairly representative, and is more time- and cost-effective.
INTRODUCTION
Several studies have suggested that blood lead and cadmium levels that are well below the current safety standards are associated with health risks, such as peripheral arterial disease, impaired renal function and elevated blood pressure.(1-3) Menke et al reported that blood lead level was significantly associated with myocardial infarction and stroke mortality, even when it was below 0.48 µmol/L (10 µg/dL).(4) A Taiwanese study by Huang reported that seafood contamination was very common in Taiwan, and that the lead and copper levels of the rivers in Taiwan were the highest in the world.(5) The abstract of a study that investigated the concentration of heavy metals in the hair of 700 residents in Taiwan reported that most of the residents exhibited elevated heavy metal levels.(6) However, this abstract is unpublished and only found on the Internet.(6) To the best of our knowledge, there has been no formal report describing the heavy metal levels of healthy subjects in Taiwan. The two reports(5,6) indicate that the finding of elevated heavy metal levels in Taiwanese people might not be unusual. Therefore, for the present study, we hypothesised that there may be greater levels of four common heavy metals (lead, mercury, arsenic and cadmium) in the blood of a Southern Taiwanese population than in Western populations.
The most reliable methods for measuring body lead burden are bone X-ray fluorescence studies and calcium disodium ethylenediaminetetraacetic acid (CaNa2EDTA) mobilisation tests. This is because blood lead levels only reflect recent exposure to lead(7) and not the lead burden of the body. According to recent reports, body lead burden can be measured by collecting 72-hour urinary lead excretion after intravenous infusion of 1 g of CaNa2EDTA.(8,9) These reports recommend the daily collection of urine for 72 hours in a high-volume container before the urine is sent for lead measurement. If most of the lead (or other heavy metal) can be excreted during the first 24 hours after CaNa2EDTA infusion, then the level of heavy metal in the urine during the first 24 hours may be reflective of the body burden of these heavy metals. We hypothesised that the level of heavy metal in the urine collected in the first 24 hours would be positively correlated with the body burden of the heavy metal. Thus, the aim of the present study was twofold. First, we aimed to assess the levels of four common heavy metals (lead, mercury, arsenic and cadmium) in the blood of apparently healthy Taiwanese adults and to determine whether these heavy metal levels are greater than those of adults from Western countries. Second, we aimed to test our hypothesis that the level of heavy metal in first-day urine is reflective of the body burden of heavy metals (i.e. most heavy metals are excreted during the first 24 hours).
METHODS
A total of 40 apparently healthy persons (34 men, six women) were enrolled in the study, which was conducted from January 2012 to December 2014. The mean age of the participants was 53.1 ± 9.1 (range 30–75) years. All 40 participants resided in Southern Taiwan and 36 of them lived in Kaohsiung city. The occupations of the participants were diverse and included doctors, nurses, technicians, businessmen, businesswomen, artists, military officers, teachers and housewives. None of the participants worked in the vicinity of factories that could have led to heavy metal exposure. The participants had normal liver function and normal renal function. They did not have a history of diabetes mellitus, coronary artery disease, stroke or any connective tissue disease. Although some of the participants had a history of hypertension, they had received appropriate antihypertensive treatment and their blood pressure (BP) was below 140/90 mmHg. This study was approved by the Institutional Review Board of Kaohsiung Veterans General Hospital, Taiwan. All of the 40 participants gave written informed consent before participating in this study.
The exclusion criteria were: (a) persistent elevated BP (i.e. BP ≥ 140/90 mmHg for more than two days); (b) creatinine > 1.3 mg/dL in men or > 1.2 mg/dL in women; (c) the presence of proteinuria; (d) baseline platelet level < 100,000/cumm; and (e) cigarette smoker within the previous two years.
The study participants were instructed to avoid eating fish, clams, lobsters and crabs two days before the CaNa2EDTA injection. They were requested to drink at least 1,500 mL of water daily for three days to ensure a daily urine amount of more than 1,400 mL. To assess the side effects of CaNa2EDTA infusion, laboratory studies were done on the second day following CaNa2EDTA injection, laboratory studies were done of the participants’ creatinine level, fasting glucose level, haemoglobin A1c level, blood urea nitrogen level, alanine transaminase (ALT) level, aspartate transaminase (AST) level, blood cholesterol level, body burden of heavy metals and complete blood count. Other than that, the development of any symptom or discomfort after CaNa2EDTA infusion was also recorded.
To determine the blood heavy metal levels of the participants, fasting venous blood specimens were drawn and placed into heparinised blood collection tubes (Becton Dickinson, NJ, USA) before CaNa2EDTA infusion. If the heavy metal assays could not be completed within eight hours, the samples were refrigerated (4°C–8°C). All of the samples were examined within ten days after preparation. To determine the lead, arsenic and cadmium levels in the blood, a mixed solution was prepared by combining 3 mL of 5% nitric acid, 9 mL of Triton 100, 2 mL of ethanol and deionised water to a volume of 100 mL. For each participant, 0.4 mL of blood sample was added to 0.4 mL of internal standard and 3.5 mL of mixed solution for analysis. To determine the blood mercury levels, 0.5 mL of blood sample was added to 0.5 mL of EDTA, 0.5 mL of cystine, 0.5 mL of internal standard and 2.5 mL of 30% hydrochloric acid. The blood samples were well mixed at room temperature prior to analysis and the mixtures for determining the level of heavy metals in the blood were centrifuged for five minutes at 5,000 rpm. The lead, mercury, arsenic and cadmium levels in the blood were determined using Elan 6100 DRC Plus inductively coupled plasma mass spectrometry (Perkin Elmer, Waltham, USA). All the calibrators and blood samples were placed in the auto-sampler tray.
Since mercury does not respond to CaNa2EDTA infusion, only urine lead, arsenic and cadmium levels were determined by applying the body burden concept after CaNa2EDTA infusion. To determine the lead body burden, we assessed the level of lead in the participant’s urine using the modified EDTA mobilisation tests, as described in previous studies.(8-10) The study participants were instructed to have their breakfasts before the CaNa2EDTA infusion. Each participant received a slow intravenous infusion (over 2–3 hours) of 1 g CaNa2EDTA that was premixed with 250 mL of normal saline solution. The first five participants underwent CaNa2EDTA infusion for three hours, and the next five participants underwent the CaNa2EDTA infusion for 2.5 hours. Since none of these participants experienced any discomfort, the remaining 30 participants received the CaNa2EDTA infusion slowly for two hours.
Following the infusion, all participants started collecting their urine samples. The 24-hour urine samples were collected for three consecutive days in three 2-L bottles to assess the body burden of lead. The urine samples were collected by spontaneous voiding. The total amount of urine collected per day was recorded for each participant. For heavy metal quantification, 10 mL of urine from the total daily urine was used. The total daily urine would be well mixed before the extraction of the 10 mL for heavy metal quantification. Urine lead levels were quantified using the coupled plasma-mass spectrometry. Urine cadmium and arsenic levels were determined using the same method. The amount of heavy metal (i.e. lead, arsenic and cadmium) in the urine was calculated using the following formula: heavy metal amount (µg) = concentration of heavy metal in the urine (µg/L) × total daily urine volume (L).
Collected data was presented as mean ± standard deviation. Shapiro-Wilk test, which was used to examine whether the collected data (i.e. the heavy metal levels in blood and urine) followed a normal distribution, revealed that the data followed a Gaussian distribution. The statistical significance of the intergroup differences was determined using one-way analysis of variance and the two-tailed t-test with Bonferroni correction. In order to assess the reproducibility of the measurements of the heavy metal levels, measurements of urine heavy metal levels were repeated for 20 of the 40 participants using another 10 mL of urine from the total daily urine (that had been well-mixed). Pearson correlation analysis was employed to assess whether the measurements were reproducible. A p-value < 0.05 was considered statistically significant.
RESULTS
The levels of heavy metals in the blood and first-day urine of participants are shown in
Table I
Level of heavy metals in the blood and urine of the participants (n = 40).
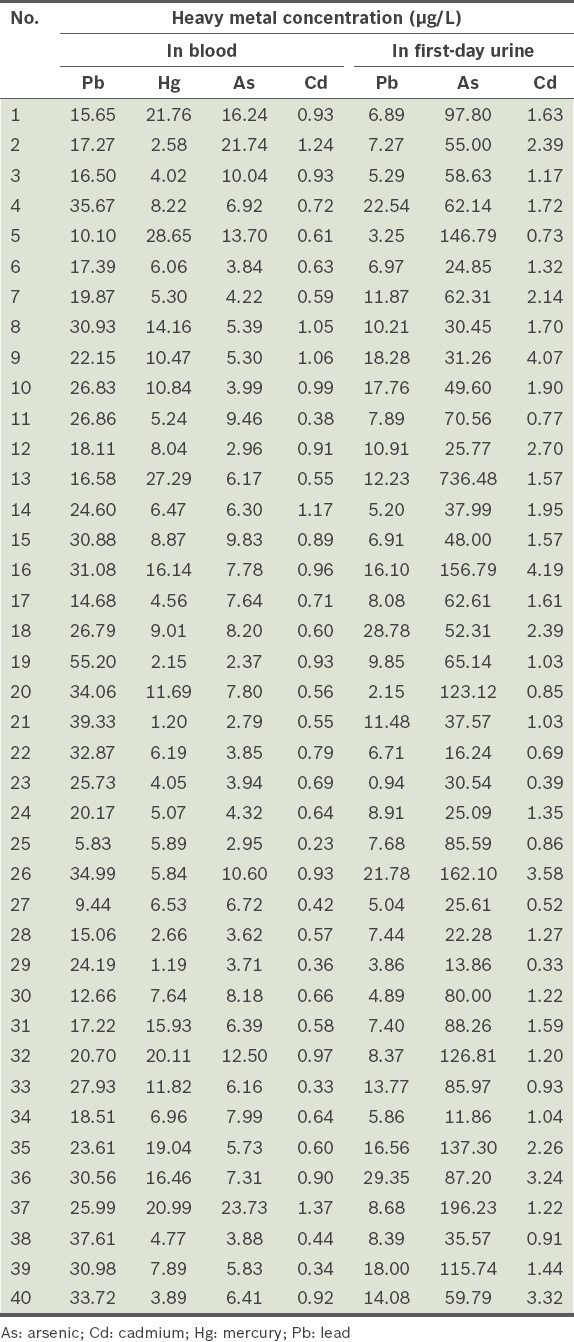
Fig. 1
Bar graphs show the levels of (a) lead, (b) arsenic and (c) cadmium in the urine of 40 participants over three days. NS: not significant
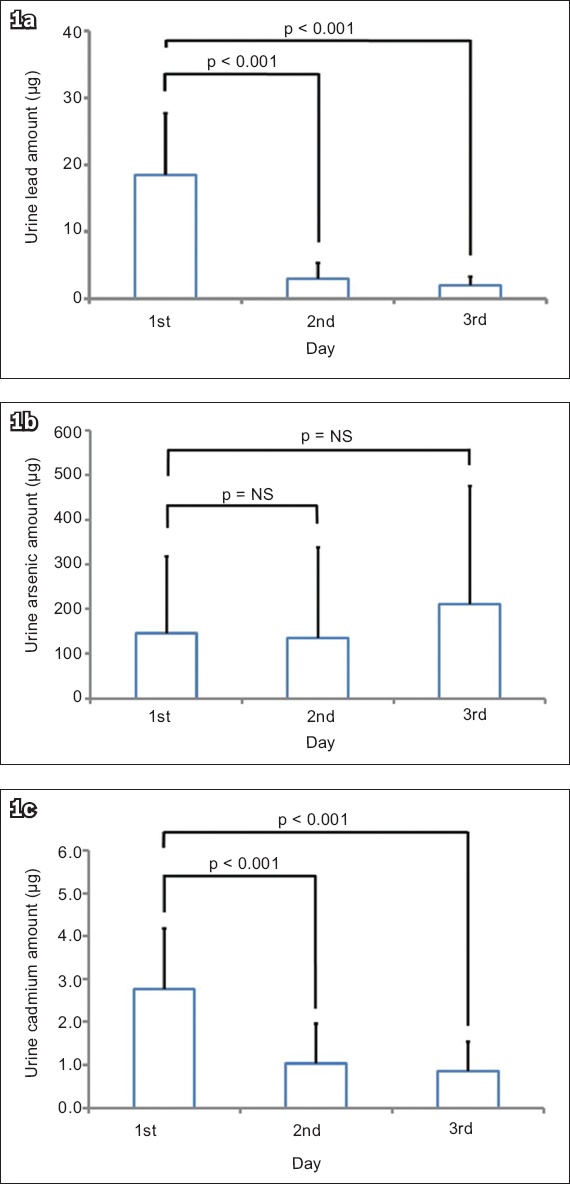
Percentage analysis showed that the amounts of lead and cadmium in the urine on the first day were 77.9% and 62.4%, respectively, of the total lead and cadmium excretion during the three days (
Fig. 2
Bar graph shows the ratio of the amount of heavy metal in urine on the first day to the total amount of heavy metal in urine for three days, for three heavy metals, lead, arsenic and cadmium.
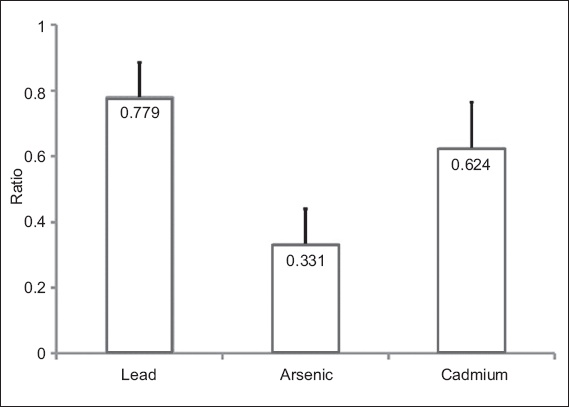
The reproducibility of the measurements of the heavy metals in urine (analysed in 20 participants) was good. The results of Pearson correlation analysis are shown in
Table II
Results of the Pearson correlation analysis testing the reproducibility of the heavy metal levels in the urine of 20 participants.
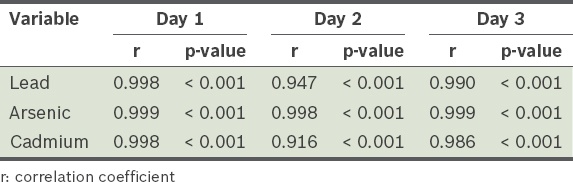
Table III
Comparison of blood heavy metal levels in three populations.
As a meticulous and slow infusion rate was used during CaNa2EDTA solution infusion and fasting was not required, all 40 participants underwent CaNa2EDTA solution infusion without experiencing any discomfort. There was no substantial change in the participants’ biochemical levels and complete blood counts before and after CaNa2EDTA infusion.
DISCUSSION
The present study investigated the blood heavy metal levels of apparently healthy adults from Southern Taiwan. It also examined the level of heavy metals in the urine (i.e. body burden of the heavy metals) of the same population after CaNa2EDTA infusion. Several studies have used the body burdens of lead to determine the relationship between environmental exposure to lead and progressive chronic kidney diseases.(8,9,13,14) These studies used the total lead excretion for three days to assess the lead burden after CaNa2EDTA infusions. The present study demonstrated that first-day lead and cadmium urine excretion could adequately reflect the body burdens of lead and cadmium. As shown in
A previous study reported that disodium EDTA was not safe, resulting in its subsequent removal from the market in June 2008.(16) On the other hand, CaNa2EDTA infusion has been approved for use in the treatment of heavy metal toxicity by the United States’ Food and Drug Administration. The present study used CaNa2EDTA infusion, which differs from the previously used disodium EDTA. Since mercury usually binds to either sodium 2,3-dimercaptopropane-1-sulfonate (DMPS) or meso 2,3-dimercaptosuccinic acid (DMSA),(17) we did not evaluate the body burden of mercury using CaNa2EDTA infusion. To assess the body burden of mercury, the chelation agent must be changed to either DMPS or DMSA. As heavy metals react differently to different reagents, it is important to choose appropriate tests (and reagents) for detecting the presence of heavy metals, depending on the suspected source of heavy metal exposure.
The present study revealed that the amount of arsenic in urine fluctuated considerably on different days. This phenomenon may be due to two reasons: (a) discontinuing the consumption of prohibited food types for only two days may have been insufficient; and (b) although the participants were instructed to avoid eating fish, clams, lobsters and crabs two days before the CaNa2EDTA injection, they consumed other foods that may have contained organic arsenic (which is not harmful to the body). The test used in the present study, as well as most available tests, detects both organic and inorganic arsenic. We speculate that this is probably the reason that no blood arsenic data was reported in the National Health and Nutrition Examination Survey III(11) and the German Environmental Survey III study(12) (
In the present study, the occupations of the 40 participants included doctors, nurses, technicians, businessmen, businesswomen, artists, military officers, teachers and housewives; none of the participants worked in the vicinity of factories, which may have led to heavy metal exposure. The working environments of the participants were better than those of persons from occupations associated with heavy metal exposure (e.g. chemical factory worker, painter, car repairman, airplane maintenance personnel, long-term gas station worker, construction worker, battery manufacturer and recycler, firing range instructor and plastic manufacturer). If we had included persons from the latter group in the study, the heavy metal levels obtained may have been higher. We speculate that the heavy metal levels of Taiwanese persons are substantially high, and this should be of concern to the public and medical community.
A previous study reported that prolonged exposure to lead may cause cardiovascular, renal and hepatic dysfunction.(18) Three epidemiological studies have demonstrated a positive association between blood lead levels and rate of renal function impairment.(19-21) Lin et al conducted EDTA mobilisation tests to assess the body burden of lead in patients with different degrees of renal insufficiency and no known lead exposure; their results suggest that long-term, low-level environmental lead exposure may be associated with the progression of renal insufficiency.(8,9) They also showed that long-term, low-level environmental lead exposure may be associated with renal tubular and glomerular damage in patients with immunoglobulin A nephropathy(22) and in residents who had been living near a lead battery factory for more than ten years.(23) The findings of these studies suggest that there may be a positive association between long-term lead exposure (resulting in a high body burden of lead) and impaired kidney function.
Regular monitoring of renal function over an extended period of time is needed to determine whether persons who are exposed to high levels of lead are more likely to develop renal issues as compared to persons who are exposed to lower levels of lead. A nationwide survey on heavy metal exposure should be conducted in Taiwan in order to investigate this important health issue; this survey should involve a large number of study participants, and be done using a standardised questionnaire and blood or urine heavy metal tests.
The present study was not without limitations. It involved a relatively small study population (n = 40). Also, all of the participants resided in Southern Taiwan. Hence, the results may not be representative of the general population of Taiwan.
Previous studies used the conventional method of collecting total urine for three consecutive days to reflect the body burden of lead in end-stage renal disease.(8,9,13,14) However, the findings of the present study demonstrated that first-day urine may be sufficient for determining the body burden of lead and cadmium in healthy persons. As all of the 40 participants had normal creatinine levels, it is uncertain whether this concept is applicable to patients with slightly elevated creatinine levels. Future studies are needed to determine whether this concept is applicable to patients with mildly abnormal renal function.
In conclusion, to the best of our knowledge, our study is the first to investigate the levels of four common heavy metals in the blood of healthy Taiwanese adults. It showed that Taiwanese people have higher blood mercury and cadmium levels than people from Western countries. The lead and cadmium excretion on the first day represented most of the lead and cadmium excreted in the three days after CaNa2EDTA infusion. Thus, the collection of only first-day urine, instead of total urine for three days, is a practical method for clinically evaluating lead and cadmium body burdens.
ACKNOWLEDGEMENTS
This study was supported by the Veterans General Hospital, Taiwan, and the University System of Taiwan Joint Research Programme (Grant No. VGHUST100-G3-2-1).


