Abstract
INTRODUCTION
Amblyopia treatment in the form of glasses and/or patching in children poses a great challenge to parents. This study aimed to assess the factors that influence visual outcome in amblyopia and children’s perception towards treatment.
METHODS
180 children (aged 3.0–7.0 years) with newly diagnosed amblyopia were recruited. The effects of age, gender, type of amblyopia, treatment and compliance on visual outcome at one year were assessed. Parents completed a questionnaire on children’s attitudes towards amblyopia treatment.
RESULTS
150 (83%) children with a mean age of 5.2 ± 0.8 years returned for follow-up at one year. 130 (87%) had refractive amblyopia and 20 (13%) had strabismic and refractive-strabismic amblyopia. Visual acuity (VA) of 6/9 or better in the amblyopic eye was achieved in 121 (81%) children. On multivariable analysis, poor responders were more likely to have initial VA of worse than 6/15 (relative risk [RR] 4.17, 95% confidence interval [CI] 1.58–11.00, p = 0.004), prescribed combined (glasses and patching) treatment (RR 2.83, 95% CI 1.02–7.83, p = 0.045) and poor compliance (RR 6.10, 95% CI 1.90–19.57, p = 0.002) after adjustment for age, gender and type of amblyopia. While 7% of children initially reacted poorly to treatment, 5% remained uncooperative at the first follow-up visit. Children had difficulty with schoolwork (5%), mood changes (6%) and social problems (2%) associated with treatment.
CONCLUSION
Most children with amblyopia respond well to treatment, but more care (i.e. more parental education and closer follow-up) may be needed in children who are non-compliant and have poorer initial VA.
INTRODUCTION
Amblyopia is characterised by suboptimal vision that has no demonstrable abnormality in the visual system or is not fully explained by an underlying ocular pathology. It can affect one or both eyes and is usually more responsive to treatment before the age of seven years.(1,2) Globally, it affects 0.2%–6.2% of children and teenagers.(3) In East Asia, the prevalence of amblyopia was 0%–2.2% in children aged 1.5–15.0 years.(3-8) Amblyopia is often treated by optical correction of the refractive error (if any) and occlusion patch therapy or therapeutic penalisation of the non-amblyopic eye.(9) The duration of treatment depends on the degree of visual deficit and improvement in vision over time.
The success of amblyopia treatment has been linked to the age at presentation, type of amblyopia, visual acuity (VA) at diagnosis and compliance with treatment.(10) Although outcomes tend to be better in younger children (< 7 years), studies suggest that the visual outcome within this age group was not different(2,11-15) and that visual improvement was still possible in older children.(16) Several studies reported better outcomes with anisometropic (refractive) amblyopia.(11,15) On the other hand, other reports suggested no difference in short-term(17-20) or long-term treatment outcome in children with strabismic and mixed strabismic-refractive amblyopia.(21) Reports of the effect of initial VA on visual outcome are also variable. While some investigators noted no association between initial VA and the visual outcome of the amblyopia treatment,(12,22) others reported that initial VA at presentation was correlated with the amount of improvement and final visual outcome.(11,13,17,18,23,24)
Compliance to amblyopia treatment is an important factor in determining outcome.(25) This is hindered by the psychological effect of the treatment modality used. Treatment (especially glasses and patching) was found to be more disabling than the condition in some children.(26) There are variable reports concerning the ability of children to tolerate amblyopia therapy. Several studies reported no significant change in behaviour and psychosocial well-being of children undergoing amblyopia treatment,(27-29) while others reported a negative impact.(26)
In Singapore, children are first screened by a school health screening service at age 4–5 years when they first attend kindergarten, with visual screening continuing annually till 14 years of age. Children suspected of having visual impairment are referred to ophthalmology services. The paediatric ophthalmology subspecialty at KK Women’s and Children’s Hospital (KKH), Singapore, is a major referral centre that receives amblyopia referrals from screening sites all over Singapore. The aim of this study was to determine the factors that affect visual outcome and to assess parents’ perception of children’s attitudes towards amblyopia treatment.
METHODS
In this prospective, hospital-based observational study, all children aged 3.0–7.0 years who were newly diagnosed with amblyopia over a period of six months at KKH (September 2007–March 2008) were recruited. Children were excluded from the study if they had a previous diagnosis and treatment for amblyopia, developmental delay, autism, syndromes (e.g. trisomy 21) or chronic medical conditions. Prior to examination, informed written consent was obtained from the parents or guardians, with an explanation of the nature of the study. The study was approved by the local research ethics committee and conducted in accordance with the tenets of the Declaration of Helsinki.
A detailed history of symptoms and past illnesses was obtained. An assessment of unaided VA using the Snellen chart was done. The Kay Picture Test was performed for those who could not be tested with the Snellen chart. Values were then converted to logMAR units for the purpose of analysis. An orthoptic assessment was done to examine ocular alignment and motility. Cycloplegic refraction was done for each child 30 minutes after instillation of proparacaine 0.5% eye drops, three drops of cyclopentolate 1% five minutes apart, and one drop each of phenylephrine 2.5% and tropicamide 0.5% eye drops. Spherical equivalent (SE; i.e. sphere + half cylinder) refraction was calculated. Best corrected visual acuity (BCVA) was recorded with full cycloplegic refraction or non-cycloplegic refraction, depending on which was better. All children underwent full ocular examination to exclude any pathology in their anterior and posterior segments.
Unilateral amblyopia was defined as BCVA worse than 6/12 in at least one eye in children < 6 years, or BCVA worse than 6/9 in at least one eye in children aged ≥ 6 years, or a two-line difference in BCVA between eyes. Bilateral amblyopia was defined as BCVA worse than 6/12 in both eyes in children < 6 years, and worse than 6/9 in children aged ≥ 6 years in the presence of amblyogenic risk factors. Amblyogenic risk factors included the presence of strabismus and significant refractive errors (bilateral ametropia of hyperopia of +4.00 D and worse; myopia of –6.00 D and worse, or astigmatism of –1.50 D and worse; and anisometropia of difference of 1.00 D SE or more in hyperopia; 2.00 D SE or more in myopia and 1.00 D or more in astigmatism, in any meridian). Mixed refractive amblyopia was diagnosed if more than one refractive amblyogenic factor was present. Strabismus was defined as manifest deviation for distance and/or near fixation with or without glasses. Refractive-strabismic amblyopia was diagnosed if the child had both strabismus and significant refractive error. Children with strabismic amblyopia alone were included in the refractive-strabismic group for ease of statistical analysis.
The aetiology of amblyopia was evaluated and treatment was started at the time of diagnosis. The type of treatment prescribed was selected by the attending doctor and included refractive correction (glasses), occlusion therapy (patching) or both.
On the follow-up visit at 4–6 months, parents completed a short questionnaire about their child’s first reaction to the treatment (very well, fairly well or poor) and attitude to the treatment (always cooperative, sometimes cooperative or rarely cooperative). They were also asked whether: glasses or patching was easier to implement among children who were prescribed both (glasses, patching or no difference), there was any change in mood or temperament since starting treatment (not at all, a little or a lot), the treatment affected the child’s schoolwork (not at all, a little or a lot) or social life (not at all, a little or a lot), and the parents were worried about their child’s treatment (not at all, slightly or a lot). Parents were asked about the strategies they found useful to make the child follow the treatment, such as explaining that it was necessary, rewarding the child, wearing patches/glasses themselves or making siblings wear them, putting glasses or patches on toys, allowing children to choose their own glasses/stickers for patches, or obtaining the support of others such as a teacher.
VA was measured at each visit and the level of compliance to treatment was recorded. Good and poor compliance were defined as following treatment for ≥ 50% and < 50% of the prescribed duration, respectively, and was based on the time reported by the parents. Visual outcome was determined at one year. A poor visual outcome at the end of one year was defined as VA worse than 6/9.
Proportions were expressed as percentages, and differences between proportions were analysed using chi-square test for proportions. Continuous measures were analysed using Student’s t-test. Multivariable analysis was performed to evaluate the risk factors that affect the visual outcome in children with amblyopia. Statistical analysis was done using commercially available software, Stata Statistical Software Release 12 (StataCorp LP, College Station, TX, USA).
RESULTS
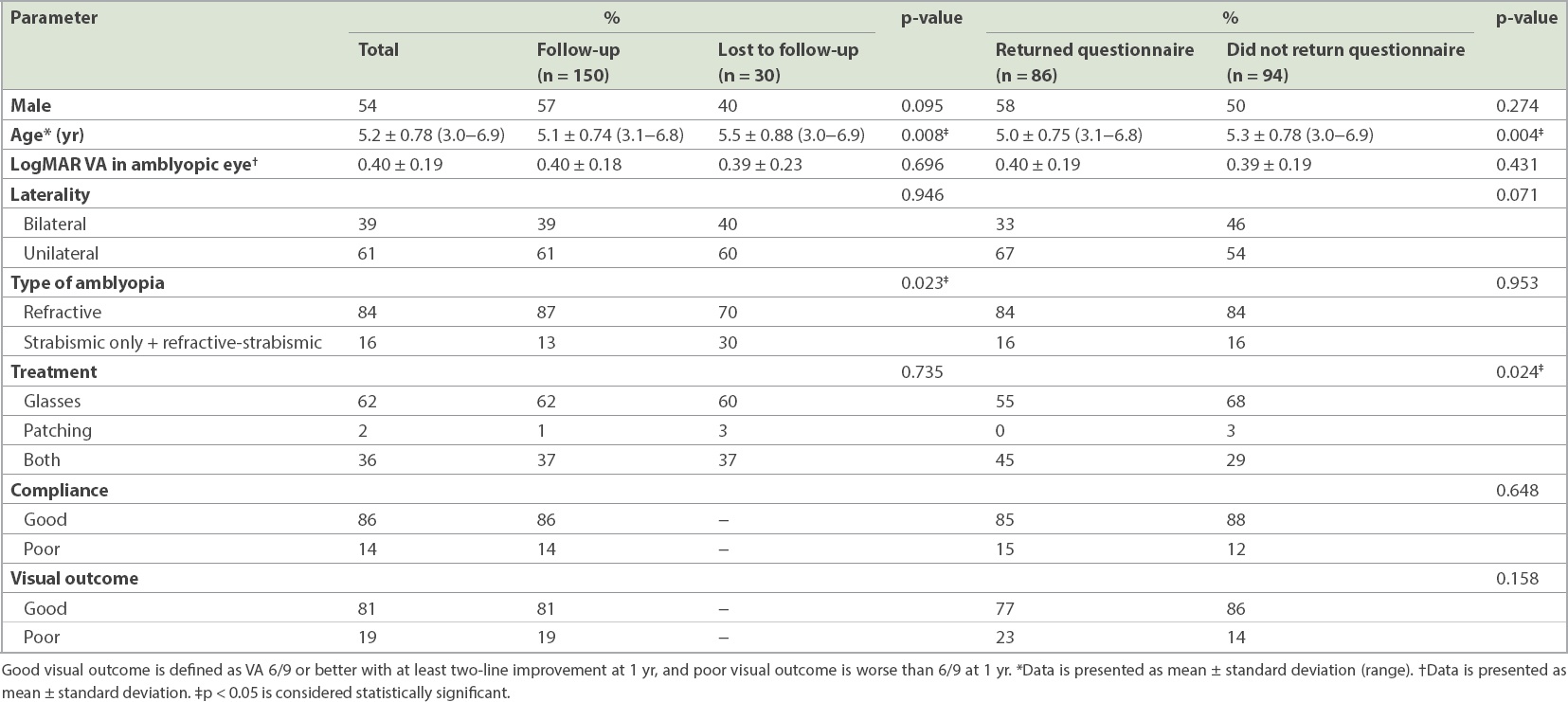
A total of 180 children were recruited. Their mean age was 5.2 ± 0.8 years and median age was 5.2 (range 3.0–6.9) years. 150 (83%) children returned for follow-up at one year. Children who returned were more likely to be younger (mean age 5.1 years vs. 5.5 years; p = 0.008) and to have refractive amblyopia (87% vs. 70%; p = 0.023) compared to those who were lost to follow-up (
Table I
Differences between children who returned and failed to return for follow-up at one year, and those who returned and failed to return the questionnaire.
Among the 150 children who returned for follow-up, 13% (n = 20) had strabismic-only (2%, n = 3) and refractive-strabismic (11%, n = 17) amblyopia, while the rest (87%, n = 130) had refractive amblyopia. More than half of the children with strabismic-only and refractive-strabismic amblyopia had exotropia (65%, n = 13). In the 130 children with refractive amblyopia, 51 children (39%) had anisometropic amblyopia, 49 (38%) had meridional amblyopia (astigmatism) and 30 children (23%) had a combination of meridional and ametropic amblyopia.
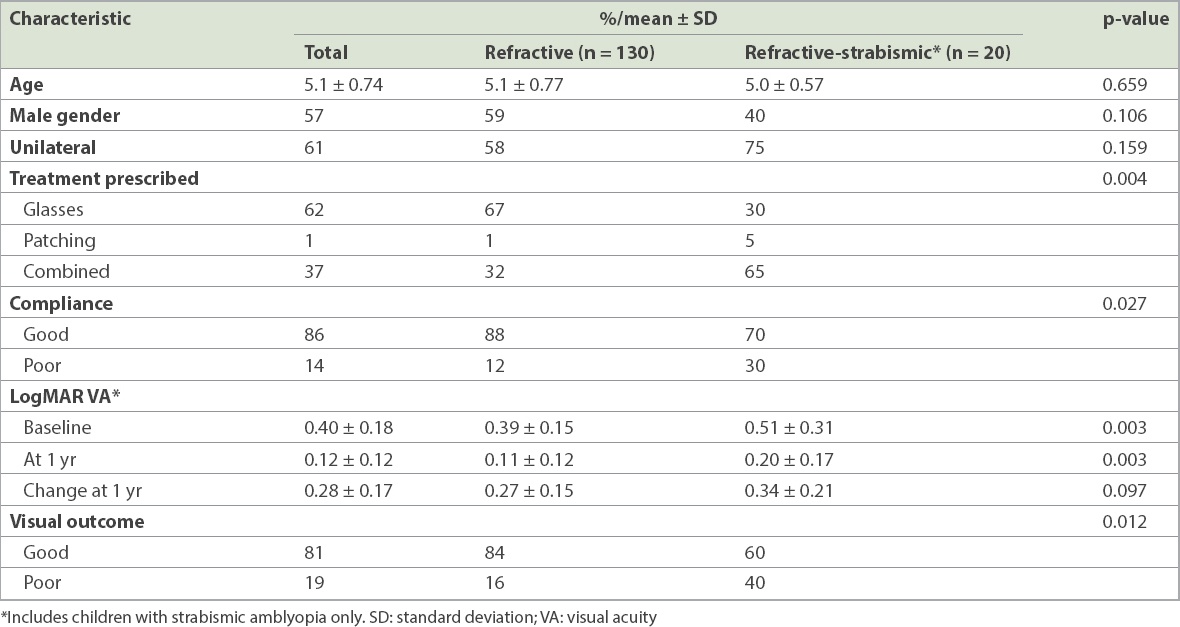
Children with strabismic-only and refractive-strabismic amblyopia were more likely to have poorer VA at presentation (logMAR VA 0.51; p = 0.003) and at one year of treatment (logMAR VA 0.20; p = 0.003) compared to those who had refractive amblyopia (logMAR VA 0.39 and 0.11, respectively) (
Table II
Differences in baseline characteristics and visual outcome in children with refractive and refractive-strabismic amblyopia.
Among the group who returned for follow-up, parents of 86 (57%) children returned the questionnaire. Those who returned the questionnaire were more likely to be younger (5.0 vs. 5.3 years, p = 0.004) and to have been prescribed both glasses and patching for treatment compared to those who did not return the questionnaire (45% vs. 29%; p = 0.024) (
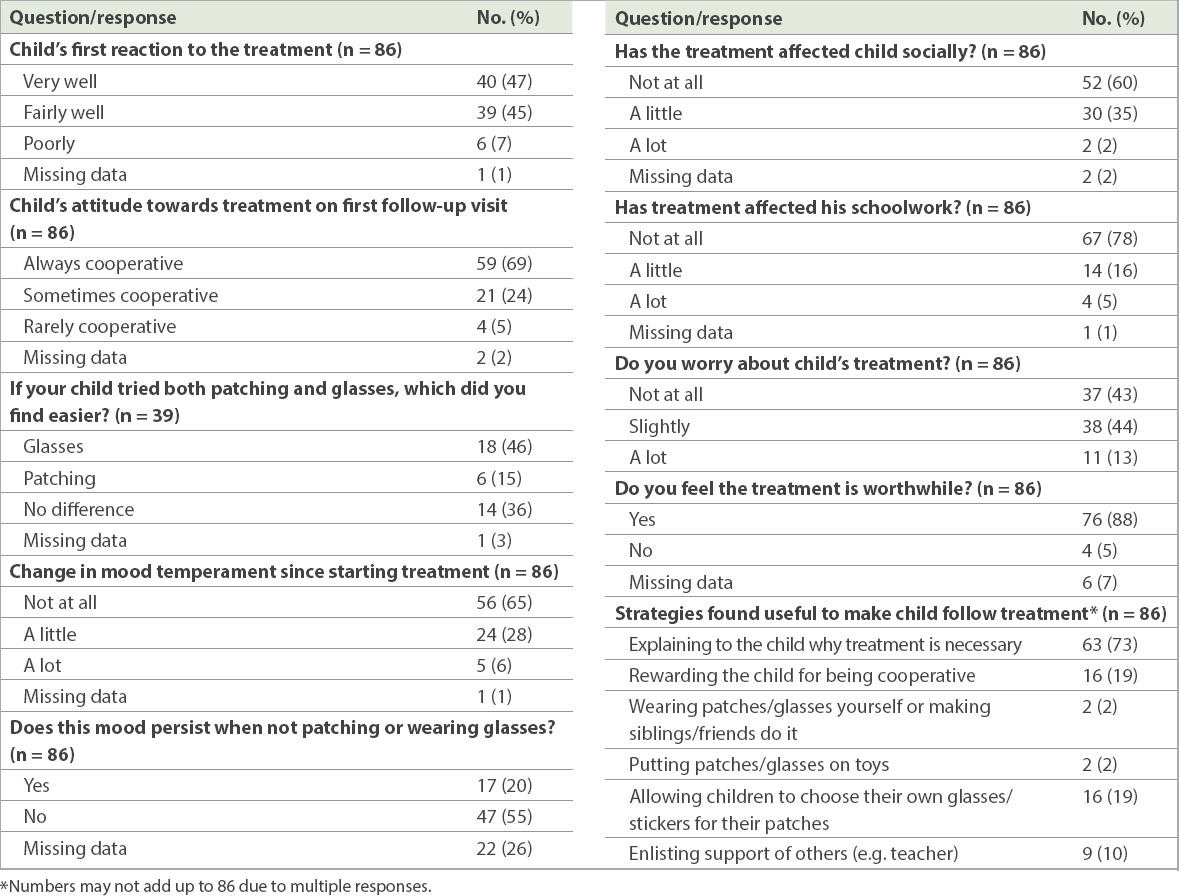
According to the parents, only 47% of the children were cooperative and accepted the treatment easily at initiation. However, 69% of them were cooperative by the first follow-up visit. Among those who were prescribed combined treatment (i.e. glasses and patching), 46% of parents found that making a child wear glasses was an easier option, but 36% found no difference between glasses and patching. Mood was adversely affected in 6% of the children. Parents felt that children had great difficulty with schoolwork (5%) and had a lot of social problems (2%) due to the prescribed treatment. The median age of the children who had difficulty with schoolwork and social problems was 5.2 (3.1–6.2) years and 5.2 (3.6–6.3) years, respectively. About 13% of parents worried a lot about their child’s treatment, and 5% felt that the treatment was not worthwhile. Most (73%) found that simply explaining the need for the treatment to their children was sufficient, while others said that rewarding the children or allowing them to choose their own glasses or stickers (19% each) and enlisting the support of teachers (10%) helped (
Table III
Children’s attitude towards the prescribed treatment.
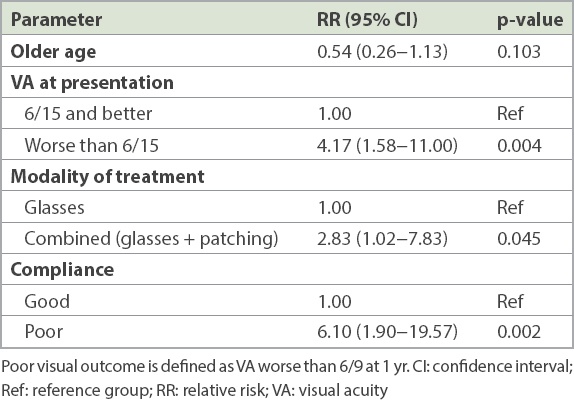
A good outcome (VA 6/9 or better with at least two-line improvement at one year) was noted in 81% (n = 121) of children. Univariate analysis showed that children who had a poor outcome were more likely to have strabismic-only and refractive-strabismic amblyopia (28% vs. 10%; p = 0.012), poorer logMAR VA at presentation (0.52 vs. 0.37; p < 0.001), prescribed combined treatment with patching and glasses (55% vs. 32%; p < 0.001) and poor compliance with the treatment (38% vs. 8%; p < 0.001) compared to those with a good outcome (
Table IV
Univariate analysis of factors that determined good and poor visual outcome.
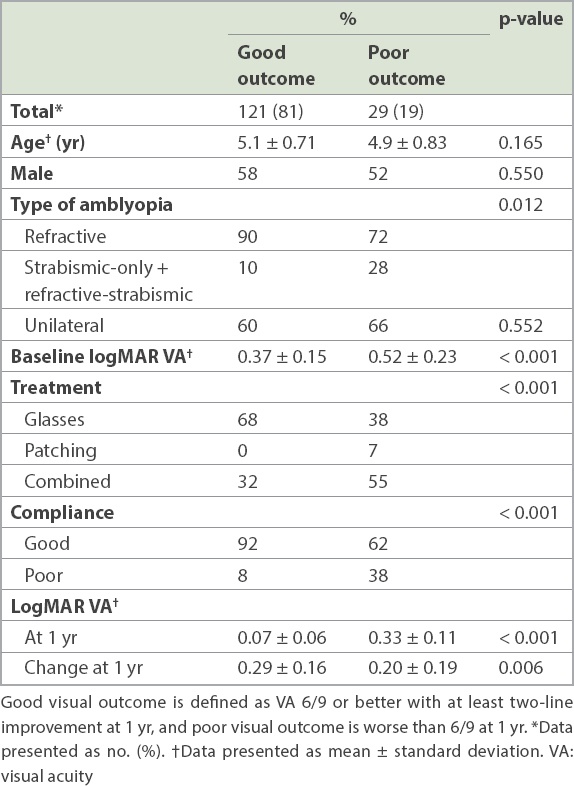
Table V
Multivariable analysis of factors determining poor visual outcome.
DISCUSSION
In this study, we evaluated the factors that may influence visual outcome in an Asian population of children with amblyopia and parents’ perception of their children’s reaction to the treatment. About 81% of the children achieved VA 6/9 or better with at least two-line improvement at one year after starting the treatment. In our study, poorer VA at presentation, combined patching and glasses (compared to glasses alone), and poor compliance were the main factors related to visual outcome on multivariable analysis. Most parents felt that their children accepted the treatment very well, with only a small proportion of them (< 10%) noting problems with mood, schoolwork and social issues.
Amblyopia was attributed to refractive error in about 85% and to strabismus in 15% of Singaporean preschool children in the population-based STARS (Strabismus, Amblyopia and Refractive Error in Singaporean Children) study.(3) We found similar proportions in our study, with 87% of the children having refractive amblyopia and 13% having strabismic-only and refractive-strabismic amblyopia. Low rates of strabismic amblyopia have also been noted in other East Asian countries.(4,8) In contrast, in Western or white populations, strabismus alone often accounts for a larger proportion (37%–48%) of amblyopia cases.(30-32)
The diagnosis of amblyopia causes anxiety to families, and compliance with treatment may depend on how well the condition and need of treatment are explained to parents. In our study, about 13% of parents were very worried about the treatment, similar to 12% of the parents of children aged four years in a study by Hrisos et al.(28) Fortunately, the acceptance of amblyopia treatment increased over time, probably buoyed by the gradual improvement in VA. We found that initially, only half of the children were cooperative, while almost two-thirds were always cooperative with the treatment by the time they returned for follow-up.
Comparison of success rates between studies was difficult due to differences in disease distribution and definitions of success. Our success rates were high, with about 81% of children achieving VA 6/9 or better with at least two-line improvement in the amblyopic eye, compared to other studies reporting a 74%–79% success rate in children who were younger than seven years of age and had refractive, strabismic and mixed types of amblyopia.(13,14,20) A few other studies reported improvements to VA 6/12 or better in 55%–83% of children (aged 11 months–12 years) with anisometropic amblyopia.(24,33,34) The best visual outcomes were reported in children with anisometropic amblyopia, intermediate outcomes in pure strabismic cases, and poorer outcomes in combined-type amblyopia.(35,36) Our higher success rates could be explained by the greater proportion of children with refractive amblyopia (87%) in our study group. Although we noted better visual outcomes in children with refractive amblyopia on univariate analysis, this association did not persist on multivariable analysis. Another reason for higher success rates could be because children who did not return for follow-up at one year were excluded from analysis.
A few studies have used follow-up visits as a surrogate measure for compliance, where children who missed appointments were considered non-compliant.(37,38) Improvement in VA was noted to be poorer in children with higher numbers of failed appointments.(39) In our cohort of children with amblyopia, about 17% (n = 30) were lost to follow-up, resulting in a possible bias towards compliant children. Among those who returned for follow-up, children with poorer compliance were 6.10 times more likely to have a poorer outcome. Although reporting bias may exist when parental feedback is used to define compliance, our results are consistent with other studies that showed an association of poor compliance with poor outcome.(11,24,35)
Interestingly, within our cohort of children aged 3–7 years, there was no correlation between visual improvement and age, suggesting that older children were as likely to respond to treatment as younger children.(17,23) This is similar to the lack of age effect observed in the Western studies, at least in children younger than ten years.(11-14)
In our study, the modality of the treatment was also a factor in the outcome, with more of the children in the poor outcome group requiring patching. Children with combined patching and glasses treatment also had a poorer outcome than those treated with glasses alone. Patching can cause psychological distress in both the child and parents,(28,40) which may negatively affect compliance. In contrast, there may be less resistance to glasses.(41) However, the mode of treatment may also be influenced by the perceived severity and type of the amblyopia. Advice to start patching may be more likely to be taken when children have greater asymmetry of vision or strabismic amblyopia.
Although children with poorer VA have the potential for greater visual improvement, studies suggest that poorer initial VA was often predictive of poor visual outcome.(11,13,17,41) Stewart et al reported a worse visual outcome in children with initial VA worse than 0.6 logMAR units compared to the rest; however, there was no difference in the ratio of VA improvement in the amblyopic eye compared to the possible improvement in VA similar to the fellow eye.(42) In our study, children with initial VA worse than 6/15 (0.4 logMAR units) had poor visual outcome after adjustment for age, gender and type of amblyopia. However, children with poor visual outcome had a smaller change in VA at one year.
The strength of our study was its prospective nature and the recruitment of a large number of consecutive cases of newly diagnosed children with amblyopia, who were referred after school screening. Limitations of the study included the variations in pre-amblyopia VA and the types of treatment offered by different doctors. Second, about 17% of the total study population were lost to follow-up, which may have resulted in bias towards children who might have been more compliant to the treatment. Third, a conclusion about the effect of amblyopia type on visual outcome cannot be made due to the small number of strabismic-only amblyopia cases in our population and their inclusion in the refractive-strabismic amblyopia group for statistical analysis. Fourth, we used a non-validated questionnaire to study parents’ perception of children’s attitudes towards amblyopia treatment. Finally, the response rate to the questionnaire was only 57% and we could not contact the parents who did not return the questionnaire.
In conclusion, a good visual outcome was obtained in most of the children. Unfortunately, children with strabismic-only and refractive-strabismic amblyopia were more likely to be lost to follow-up. As with previous studies done in Western populations, poorer baseline VA and compliance were predictive of poor visual outcome. This suggests that more care is required to ensure that parents are well aware of the importance of good compliance and adequate follow-up, particularly those with strabismic-only and refractive-strabismic amblyopia, and children with poorer visual acuity at presentation.


