Abstract
A 60-year-old man presented with abdominal pain. He was later diagnosed on imaging to have high-grade small bowel obstruction. The patient underwent surgery, and a hard, rounded bezoar resembling the endosperm of Nypa fruticans, colloquially known as attap chee, was found at the point of obstruction. Small bowel obstruction is a common acute surgical condition with multiple causes, including bezoars. We discuss the typical imaging features of bezoars causing small bowel obstruction as well as potential pitfalls that can mimic the appearance of a bezoar.
CASE PRESENTATION
A 60-year-old Chinese man with a past medical history of hypertension, hyperlipidaemia and previous cerebrovascular accident six years ago presented with a one-day history of acute abdominal pain and vomiting. This was associated with abdominal distension and symptoms of obstipation. He did not complain of any constitutional symptoms such as weight loss or loss of appetite. He did not report having any prior abdominal surgery.
Physical examination revealed mild, central abdominal tenderness. The abdomen was distended, but no guarding or rebound tenderness was elicited. No mass or lump was felt in the hernial orifices of the groin.
Abdominal radiography (
Fig. 1
(a) Supine and (b) erect abdominal radiographs.
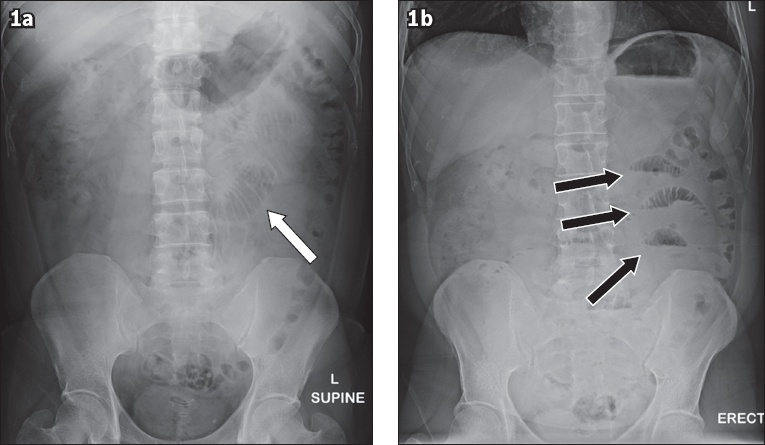
Fig. 2
(a) Axial and (b) coronal CT images of the abdomen and pelvis in the portal venous phase.
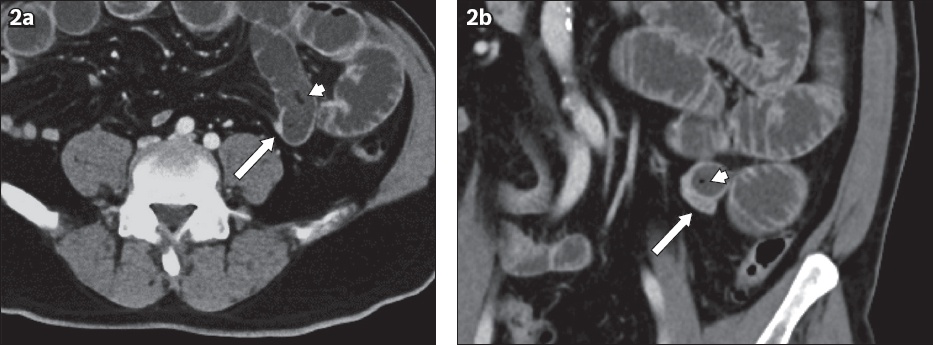
IMAGE INTERPRETATION
Abdominal radiography shows a few dilated small bowel loops on the supine view (arrow,
CT confirmed the presence of multiple dilated small bowel loops. A distinct transition point is seen in the left lower quadrant, where the dilated small bowel loops abruptly return to normal calibre (arrowheads,
DIAGNOSIS
Small bowel obstruction secondary to a bezoar.
CLINICAL COURSE
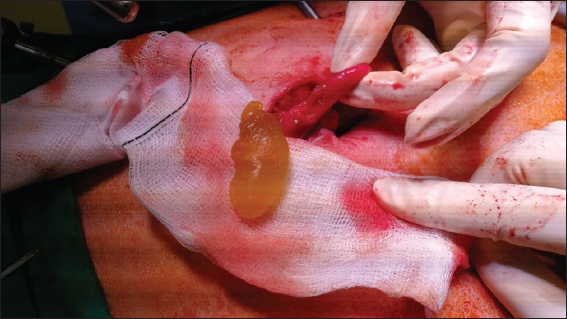
The patient was scheduled for urgent surgery in view of the CT findings of small bowel obstruction. He underwent a diagnostic laparoscopy, which confirmed the presence of small bowel obstruction with a discrete transition point. Distal to the transition point, the small bowel loops were collapsed. The bowel was otherwise healthy and viable. The cause of the obstruction was due to a hard intraluminal mass, presumably a bezoar. As such, the affected small bowel was exteriorised and an enterotomy made to remove the bezoar, with subsequent closure of the enterotomy. The extracted bezoar visually resembled a palm seed from Nypa fruticans, also known colloquially as an attap chee (
Fig. 3
Intraoperative photograph shows an ovoid bezoar resembling the endosperm of Nypa fruticans at the site of the obstruction.
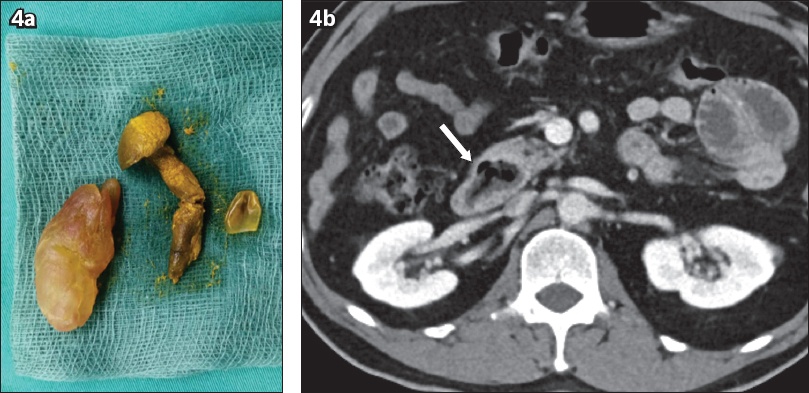
An on-table oesophagogastroduodenoscopy was also performed. It showed a periampullary diverticulum containing a bezoar that was subsequently retrieved endoscopically. On extraction, the bezoar within the periampullary diverticulum resembled a mushroom (
Fig. 4
(a) Photograph shows the probable attap chee bezoar (left) and the extracted mushroom (right). (b) Axial CT image shows the mushroom phytobezoar in the periampullary diverticulum (arrow).
DISCUSSION
Small bowel intestinal obstruction is a common surgical condition encountered in clinical practice. In patients who had previous abdominal surgery, the commonest cause of small bowel obstruction is adhesions. In patients with no prior history of abdominal surgery (i.e. the virgin abdomen), causes of intestinal obstruction are varied and can be classified as intraluminal, mural or extramural aetiologies (
Box 1
Causes of virgin abdomen intestinal obstruction:

Bezoars are a well recognised, albeit uncommon, cause of small bowel obstruction, contributing to 4% of cases.(1,2) They are masses composed of partially digested or undigested food material within the lumen of the gastrointestinal tract and are formed due to high fibre intake, inadequate mastication and reduced gastric motility. Hence, bezoars are prevalent among patients with delayed gastric emptying, such as after a vagotomy or gastrectomy, or as a result of autonomic dysfunction from diabetes mellitus.(3,4)
Bezoars are commonly seen in the stomach and the small bowel. Although gastric bezoars are more common, small bowel bezoars are more likely to present with intestinal obstruction. Most small bowel bezoars are located in the ileum, approximately 50–70 cm proximal from the ileocaecal junction.(5-7) It has been postulated that this is due to the smaller luminal calibre of the ileum, reduced bowel motility and the fact that a large amount of water reabsorption occurs in the small bowel. Water reabsorption causes hardening of the bezoar, resulting in impaction and obstruction. A few different types of bezoars have been described, with phytobezoars being the most common.(8,9) Other rarer forms of bezoars include trichobezoars, which are seen in psychiatric patients who ingest hair,(10) and lactobezoars, which are masses of milk coagulum found in infants.(11)
The clinical presentation of small bowel obstruction due to a bezoar is similar to any other case of small bowel obstruction. Patients present with colicky abdominal pain, vomiting, abdominal distension and constipation.(12) On conventional abdominal radiography, dilated small bowel loops and multiple air-fluid levels are seen. In the appropriate clinical context, this is in keeping with small bowel obstruction. The offending bezoar is usually not detected on radiography unless it is large.
Contrast-enhanced CT of the abdomen and pelvis is the imaging modality of choice in the evaluation of patients with intestinal obstruction. CT allows the radiologist to identify the site of obstruction (commonly referred to as the transition point) and, more importantly, elucidate the cause of obstruction. CT also helps to evaluate for the presence of complications such as perforation or bowel ischaemia.
The typical CT appearance of a bezoar is a discrete intraluminal mass that contains multiple gas locules in its interstices and is located at the transition point where it is causing intestinal obstruction (Figs.
Fig. 5
A 60-year-old male patient presented with intestinal obstruction. (a) Axial and (b) coronal CT images show a well-defined mass lesion (arrow) with a mottled appearance due to the presence of multiple gas locules trapped in its interstices. The patient underwent surgery, which showed a phytobezoar composed of undigested vegetable material.
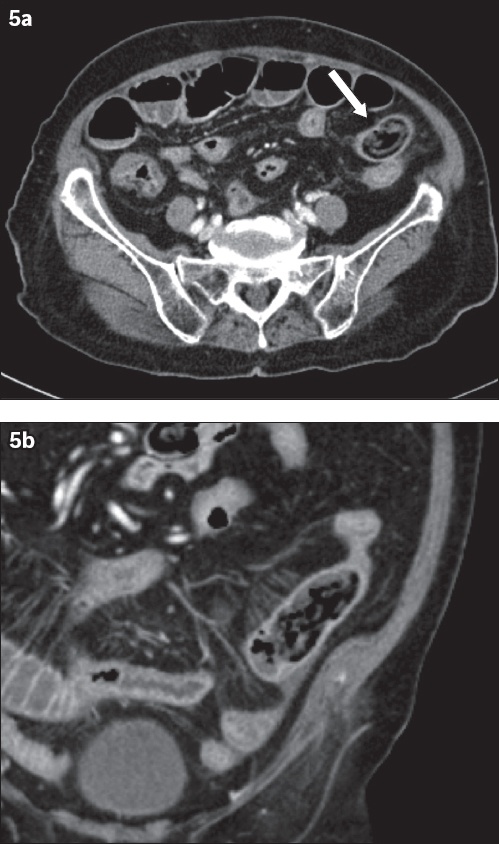
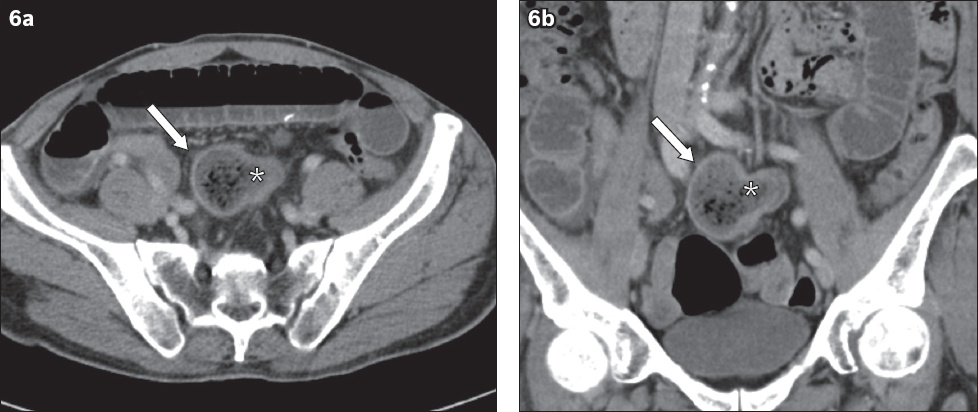
Fig. 6
A 83-year-old female patient who presented with intestinal obstruction. (a) Axial and (b) coronal CT images show a well-defined mass lesion (arrows) with a heterogeneous mottled appearance due to gas locules trapped within its interstices, in keeping with a bezoar. The bezoar has an unusual T-shaped appearance, as part of it was trapped within a Meckel’s diverticulum (asterisk). The obstructing bezoar was surgically removed and shown to be a phytobezoar.
An entity that can mimic the appearance of a bezoar is the ‘small bowel faeces’ sign (
Fig. 7
Axial CT image shows ‘small bowel faeces’ sign in a 75-year-old woman. Note that the appearance of the small bowel contents is very similar to the bezoars seen in Figs.
Distinguishing intraluminal bezoars from the ‘small bowel faeces’ sign is a challenge with many overlapping features on CT. Indeed, in a study by Delabrousse et al, several CT features that were thought to be exclusively seen in bezoars were also detected in cases that had the ‘small bowel faeces’ sign. Nonetheless, their study showed that features such as a well-encapsulated wall, a similar bezoar-like lesion in the stomach and the ‘floating fat density’ sign were useful in distinguishing cases of bezoars from ‘small bowel faeces’.(13) In our case, some foci of fat density were also seen proximal to the obstructing lesion (Figs.
Fig. 8
Selected axial CT images of the patient in
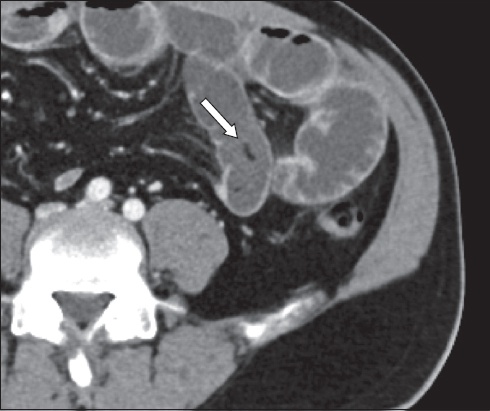
Fig. 9
A 67-year-old man presented with intestinal obstruction. (a) Axial and (b) coronal CT images show an obstructing bezoar (arrows) with a mottled appearance due to gas locules, proven on surgery to be a phytobezoar resembling a shiitake mushroom. (c) Axial CT image of the abdomen shows the presence of several small foci of fat-density material just proximal to the phytobezoar, which likely correspond to the ‘floating fat density’ sign.
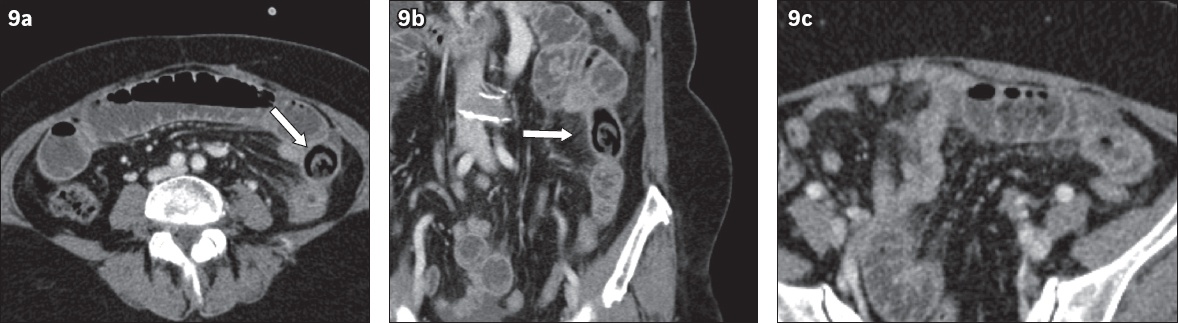
Our case is unique, as the bezoar that was extracted resembled an attap chee. Furthermore, the classic appearance of a bezoar was not found on CT. An ovoid low-density structure was seen on CT, instead of the usual gas-mottled appearance of bezoars.
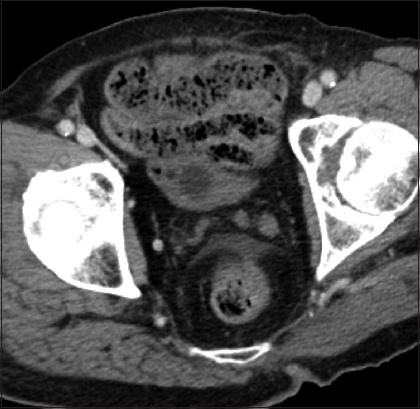
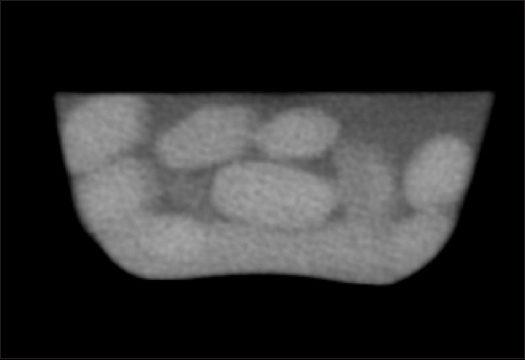
CT images of off-the-shelf seeds of Nypa fruticans showed that the seeds were hyperdense (
Fig. 10
CT image of commercially available Nypa fruticans seeds shows them to be hyperdense with an attenuation of approximately 50–60 Hounsfield units.
The definitive management of small bowel dilatation with a transition point in a virgin abdomen is often surgical. In our case, the initial working diagnosis was likely a congenital adhesion band, as it was not very evident to the surgeon that there was a bezoar on the images. However, on laparoscopy, a hard intraluminal bezoar mass was noted proximal to the transition point. As the calibre of the bowel tends to narrow as it progresses towards the caecum, it is unlikely for peristalsis alone to push the bezoar through to the more capacious colon. Surgical intervention aims to remove the bezoar as shown in this case or, in a softer phytobezoar, to manually break it intraluminally and milk it into the caecum, avoiding the need for an enterotomy. Surgery also allows an assessment of the proximal bowel to ensure that there are no other similar offending bezoars, such as another seed in a more proximal location, and to exclude other bezoars in the stomach, which could cause repeated problems.
In this clinical case, it is likely that the patient developed a phytobezoar from inadequate mastication, resulting in him swallowing the attap chee endosperm whole. Poor mastication is often seen in elderly patients with poor dentition as well as patients with neurological disorders (e.g. stroke) and results in large food particles being swallowed.(14) While studies have shown that poor dentition and mastication are risk factors for bezoar formation,(15) there is currently no published study available, to our knowledge, that correlates food particle size and the incidence of bezoar formation.
In conclusion, bezoars are an unusual cause of small bowel obstruction. Even bezoars with a classic appearance may be mistaken for the ‘small bowel faeces’ sign in longstanding high-grade small bowel obstructions. In the absence of the classic gas-mottled appearance of phytobezoars, the diagnosis can be challenging, as in this case.
SMJ-60-402.pdf


