Abstract
INTRODUCTION
Streptococcus agalactiae (Group B Streptococcus, GBS) is an uncommon cause of septic arthritis in the adult population. In recent years, there has been an increase in the incidence of GBS septic arthritis. This study aims to compare the clinical presentation, investigations, microbiology and outcome of management in patients with GBS and non-GBS septic arthritis.
METHODS
Retrospective review of hospital surgical records was done to identify all patients treated surgically at our institution from January 2011 to January 2016 for primary septic arthritis. Patients were categorised into two groups: those with culture-proven GBS septic arthritis and those with causative pathogens that were not GBS. Patients who were medically unfit for surgical intervention as well as those who declined interventional procedures were excluded from the study.
RESULTS
A total of 83 patients were included in the study: 62 (74.7%) had non-GBS septic arthritis and 21 (25.3%) had GBS septic arthritis. Patients with GBS septic arthritis were more likely to have polyarticular involvement (p < 0.001) and involvement of less common sites such as the elbow joint. They were also more likely to have elevated inflammatory markers (C-reactive protein > 150 mg/L; p = 0.017) and positive blood cultures (p = 0.02), and were typically healthy adults with no medical comorbidities (p = 0.012).
CONCLUSION
Patients with GBS septic arthritis were more likely to present with polyarticular involvement, positive blood cultures and higher levels of C-reactive protein on admission, and tended to be healthier individuals with no medical comorbidities.
INTRODUCTION
Septic arthritis is an orthopaedic emergency that has potentially severe complications, such as sepsis and limb loss, and an estimated mortality rate of 11%.(1) It generally presents as monoarticular, with polyarticular involvement being less common. Staphylococcus aureus and Streptococcus species are the usual causative organisms.(2-4) Streptococcus species can be broadly classified into two key groups; alpha-haemolytic (a-haemolytic) and beta-haemolytic (b-haemolytic) streptococci. b-haemolytic streptococci can be further subdivided into Group A Streptococcus and Group B Streptococcus (GBS). The latter is also known as Streptococcus agalactiae (S. agalactiae). Generally, streptococcal infections (all sites) are very common. The main causative micro-organisms are usually a-haemolytic streptococci and b-haemolytic Group A streptococci. On the other hand, b-haemolytic GBS are most often associated with severe infections in neonates, pregnant women and immunocompromised hosts such as the elderly population.(5)
However, in more recent times, some studies have observed that GBS has become an increasingly common pathogen in adult orthopaedic patients(6-11) and that the incidence of S. agalactiae-associated septic arthritis is rising.(6-8,10) The 2015 outbreak of GBS-associated infections in Singapore sparked nationwide interest in this pathogen and its associated infections. Not surprisingly, we also noticed a rise in the number of GBS infections as well as a corresponding increase in GBS-related septic arthritis here, particularly in 2015. In this study, we sought to study the clinical, epidemiologic characteristics and inpatient outcomes of patients with GBS septic arthritis compared to those with non-GBS septic arthritis.
METHODS
This study was conducted at National University Hospital (NUH), Singapore, a 1,200-bed tertiary acute care hospital located in western Singapore. The institution contains 41 specialised outpatient clinics/centres with an additional 19 specialised service centres. NUH has strong research and teaching affiliations with the National University of Singapore and is one of Singapore’s major tertiary referral centres, serving up to one-fifth of the population.
We retrospectively reviewed the hospital surgical records of all 83 patients who were treated surgically for primary septic arthritis from January 2011 to January 2016. Septic arthritis was defined as the presence of positive synovial fluid culture from the affected joint. In the same time period, patients who were treated non-surgically (i.e. managed purely with antibiotics and/or had needle aspiration of intra-articular pus) were excluded from this study. 68 (81.9%) patients were treated with open surgical drainage and debridement, and the remaining 15 (18.1%) patients were treated with arthroscopic debridement and washout of the affected joint, followed by completion of an appropriate antibiotics regime recommended by the infectious disease physicians. All patients were referred to and co-managed with physicians from the infectious disease department. Empirical antibiotics were commenced once the diagnosis of septic arthritis was made and then changed appropriately once the identities of the causative organisms were known. Following the advice of the infectious disease physicians, the antibiotics were continued until a successful outcome was attained, as indicated by complete clinical resolution of symptoms and downtrending of inflammatory markers.
The following parameters were obtained from hospital inpatient records: age, ethnicity, gender, type of comorbidities, blood, biochemical markers (including C-reactive protein [CRP] levels on admission), radiographic investigations, antibiotics treatment, type of surgical procedures undergone and outcome of patients during both inpatient as well as outpatient follow-up visits. The study cohort was divided into two groups, patients with culture-proven S. agalactiae (i.e. GBS) septic arthritis and patients with septic arthritis secondary to other pathogens. Patients who were medically unfit or moribund for joint aspiration or surgery, and those who refused to give consent for any form of interventional procedure were automatically excluded from this study, because pure medical treatment of septic arthritis could have skewed our observations.
Statistical analysis was performed using IBM SPSS Statistics version 22.0 (IBM Corp, Armonk, NY, USA). Wilcoxon rank-sum test and Mann-Whitney U test were used for continuous variables, while chi-square test and Fisher’s exact test were used for categorical variables. Logistic regression was performed on the significant variables identified. A p-value < 0.05 was considered to be statistically significant.
RESULTS
A total of 83 patients with proven septic arthritis were treated between January 2011 and January 2016. Of these patients, 62 (74.7%) had non-GBS septic arthritis, while 21 (25.3%) had GBS septic arthritis.
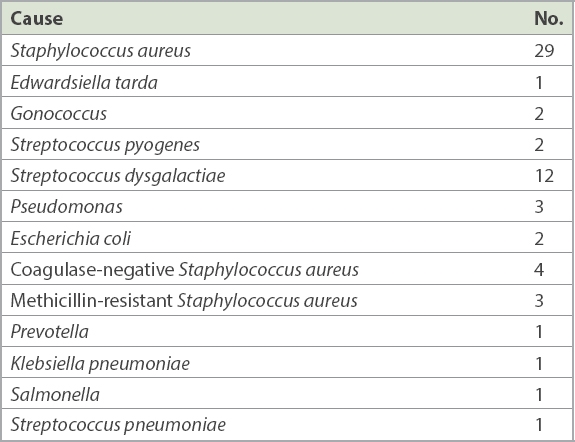
Table I
Microbiological identification of the non-Group B Streptococcus cases (n = 62).
Taking into account the ten patients in the same admission who died due to septic arthritis as a primary or secondary cause, the remaining 73 patients were followed up over a mean period of 8.13 (range 2–58) months. The wide range, after excluding the deceased patients, was mainly due to patients who were on long-term follow-up for other unrelated chronic orthopaedic problems such as concomitant osteoarthritis, fractures in other regions of the musculoskeletal system and gouty arthritis.
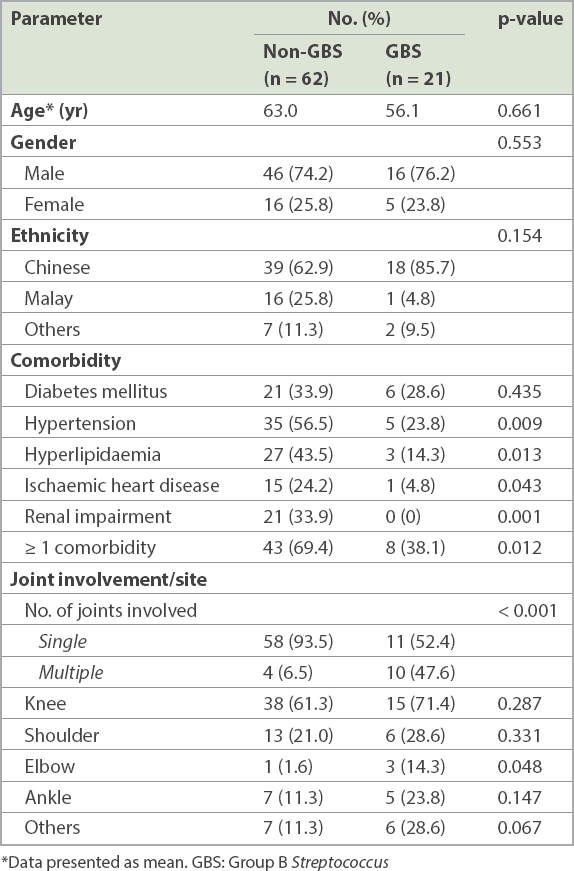
Table II
Patient demographics, comorbidities and presentation.
As shown in
Table III
Inpatient outcomes and investigations.
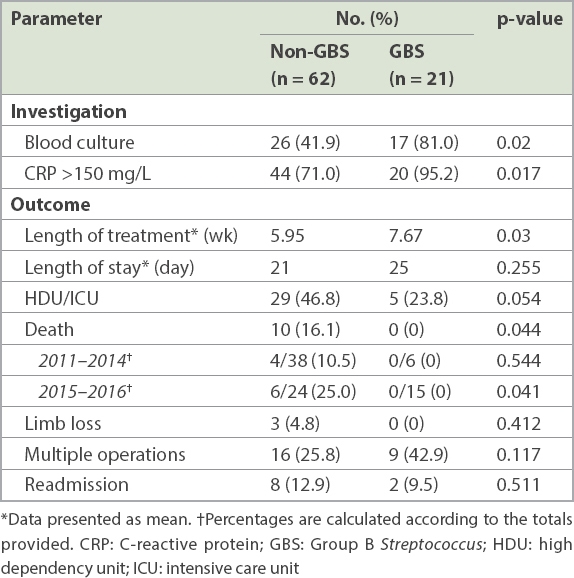
All 83 patients included in the study underwent surgical intervention in the form of either open arthrotomy debridement and washout (81.9%, n = 68) or arthroscopic debridement and washout (18.1%, n = 15) of the affected joint. 5 (23.8%) out of 21 patients with GBS septic arthritis underwent the arthroscopic procedure, as compared to 10 (16.1%) out of 62 patients with non-GBS septic arthritis. In terms of differences in the type of surgical intervention elected and performed, we did not find any significant difference between the two groups of patients (p = 0.313). The mean duration of treatment for the entire cohort of patients was 6.39 (median 6) weeks. When the two groups were compared, patients with GBS septic arthritis required a mean of 7.67 (95% confidence interval [CI] 6.58–8.76) weeks of treatment, which was significantly longer than those with non-GBS septic arthritis, who required 5.95 (95% CI 5.27–6.63) weeks (p = 0.03).
Outcomes studied included length of stay, need for monitoring at the high dependency unit (HDU) or intensive care unit (ICU), number of operations undergone, need for readmission within 30 days, limb loss, and death (
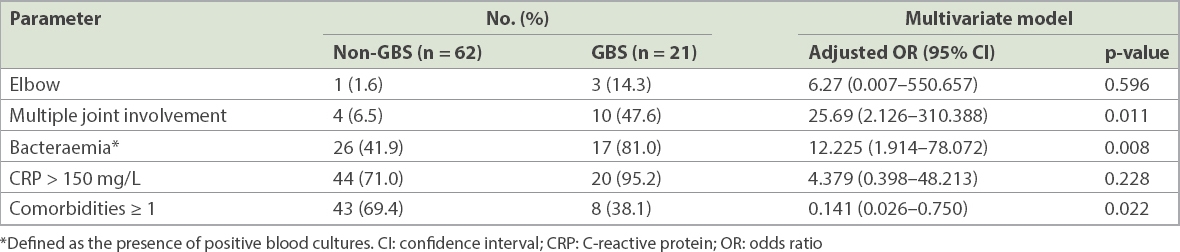
Table IV
Differences in clinical features and comorbidities between Group B Streptococcus (GBS) septic arthritis and non-GBS septic arthritis.
DISCUSSION
GBS, also known as S. agalactiae, is a type of Gram-positive bacteria and a common commensal bacterium of bowel and genital tract flora. Known transmission routes include sexual contact with colonised individuals, as well as vertical transmission from mother to neonate.(12,13) Colonisation usually does not lead to disease. Invasive GBS infections in healthy adults are not as common compared to severe infections in pregnant women, neonates, the elderly and immunocompromised individuals.(5,12,14-19)
Previous studies have described GBS causing bovine mastitis(20) and disease in fish.(21) However, our understanding of the links between the routes and likelihood of interspecies transmission and invasive disease in humans remains lacking. One study has drawn attention to a possible link in interspecies transmission of GBS from animals to humans, resulting in GBS colonisation in healthy adults.(13) Foxman et al observed an increased risk of GBS colonisation (capsular subtype Ia and Ib) with fish consumption in healthy adults.(13) However, the study only looked at the association between GBS colonisation and fish consumption, and hence could not establish a link between fish consumption and GBS infections in humans.
The recent outbreak of GBS-associated infections in Singapore sparked a widespread interest in this pathogen and its associated infections. Two recent local studies, conducted in the aftermath of the outbreak, identified a strong association between consumption of raw or undercooked fish and GBS Serotype III ST283 bacteraemia/invasive infections in men and non-pregnant women.(22,23) Their findings were further supported by the presence of GBS Serotype III ST283, the particular sequence associated with invasive disease in human beings, in fish samples tested by the Agri-Food and Veterinary Authority of Singapore and National Environment Agency. Tan et al(22) retrospectively studied a cohort of 40 case patients with GBS bacteraemia, comparing them with 58 case controls, and found that the odds ratios of GBS bacteraemia among patients who ate raw fish were 8.58 times that of those who had not eaten raw fish. Further genetic testing of bacteria isolates from case patients showed that all patients who consumed raw fish (n = 19) had Serotype III ST283, whereas case patients who did not eat raw fish had Serotypes Ia, II, III, VI and VII. The authors also found that the mean age of case patients infected with ST283 was significantly younger than those with non-ST283 (57.4 years vs. 68.6 years; p = 0.014). In addition, the authors reported that a significantly lower proportion of patients infected with ST283 reported having underlying medical conditions, compared to non-ST283 patients (56% vs. 100%; p = 0.003).(22) A second prospective case control study of 22 case patients with GBS bacteraemia or infections, conducted by Rajendram et al(23) during the epidemiologic weeks of the outbreak, drew similar observations. The authors found that patients infected with ST283 tended to be younger than those with non-ST283 (median age 59.4 years vs. 74.0 years; p = 0.033) and that they were less likely to have pre-existing medical conditions (33.3% vs. 84.6%; p = 0.014). Both studies also observed a sharp drop in the number of invasive GBS cases after a public advisory was issued in July 2015 to halt the sales of certain species of raw freshwater fish (Asian big head carp and snakehead fish). This lends substantial support to the association between raw fish consumption and invasive GBS infections.
Staphylococcus aureus is the most common micro-organism found in septic arthritis, accounting for up to 60% of cases,(2-4,24,25) followed by Streptococci species. In recent years, however, various studies have highlighted GBS as an increasingly important pathogen in adult septic arthritis.(6-8,10) As early as 1984, Laster et al(6) first alluded to the possible emergence of GBS as a pathogen in septic arthritis. They observed that for decades, only 18 cases had been described (eight cases after 1976), and that their two cases of GBS septic arthritis presenting within the same year should therefore not be attributed to pure coincidence. Findings from a more recent study by Louthrenoo et al(8) seemed to support this theory. The authors reported an incidence of 15.57% for S. agalactiae septic arthritis in patients treated for the infection between 1990 and 2010, of which 90% were diagnosed between 2008 and 2010. However, the authors were unable to find a cause for the increased infection rates. Instead, they speculated that it was related to the change in climate.
To our knowledge, no studies to date have specifically looked at the impact of the recent GBS outbreak on joint infections. In our study, about one-quarter of the septic arthritis cases were caused by GBS. Among these patients, those diagnosed in the January 2015–January 2016 period made up 38.5% of the septic arthritis cases treated during this period, significantly more than the proportion of 13.7% during the January 2011–December 2014 period (p = 0.009). This seems to concur with previous findings in the literature. In addition, all five patients who underwent further serotyping of the isolated bacteria yielded the same serotype (Serotype III ST283) of S. agalactiae. Our results are consistent with the findings of the Singapore Streptococcus agalactiae Working Group, which identified the same serotype of S. agalactiae in local patients with GBS infections as part of the outbreak investigation.(26)
Septic arthritis usually presents with monoarticular involvement. In our study, we observed that only slightly more than half (52.4%) of all GBS cases presented with single joint involvement compared to septic arthritis from other causes, of which 93.5% were monoarticular (p = 0.011). The most common sites of septic arthritis in both sets of patients were the knee and the shoulder, similar to the findings of Nolla et al(7) and Louthrenoo et al.(8) However, compared to patients with non-GBS septic arthritis, a higher proportion of patients with GBS septic arthritis presented with less typical primary sites of infection such as the elbow joint (14.3% vs. 1.6%, p = 0.048). In our multivariate analysis, however, this finding was not statistically significant (p = 0.596). Although S. agalactiae is known to affect the small joints of the body, such as the sternoclavicular joint and wrist joint,(27,28) we did not detect any significant difference between the two groups of patients with regard to small joint involvement (p = 0.067).
Septic arthritis is a condition that usually arises in the very young or old, patients with underlying joint disease (e.g. rheumatoid arthritis), individuals with underlying medical illnesses (e.g. diabetes mellitus) and immunocompromised patients.(29,30) In previous studies, up to two-thirds of patients with S. agalactiae infections had underlying conditions that predisposed them to infection.(7,31) These conditions included cancer, diabetes mellitus, systemic inflammatory disease (e.g. rheumatoid arthritis) and chronic liver disease, as well as diseases requiring corticosteroid therapy. Some authors have theorised that age and immunosuppression are important predisposing factors.(7,27-29,31) This was not so for our cohort, where logistic regression showed that GBS septic arthritis tended to affect healthier individuals with no significant comorbidities (p = 0.022). This difference from previous studies highlights the fact that the particular virulence factors of this disease still remain unknown.
No studies to date have managed to produce significant diagnostic results capable of differentiating between GBS and non-GBS septic arthritis. Our study identified some differences between the two entities. We observed that 81.0% of patients with GBS septic arthritis had positive blood cultures as compared to 41.9% of patients with non-GBS septic arthritis, which was significant on logistic regression (p = 0.008). This finding was comparable to various studies that also reported similar associations between GBS infections and bacteraemia.(8,19,22) In addition, our results showed that patients with GBS septic arthritis were more likely to have significantly higher CRP values, with 95.2% of these patients having CRP > 150 mg/L. In contrast, only 71.0% of patients with septic arthritis from non-GBS micro-organisms had CRP > 150 mg/L on univariate analysis (p = 0.017). However, on multivariate logistic regression, this difference was not statistically significant (p = 0.228).
This study included only patients who were treated surgically and excluded those who were managed purely with antibiotics and/or had needle aspiration of intra-articular pus. The intention was to exclude patients who were moribund or medically too unwell for surgery, sometimes due to the severity of the disease. Some authors might argue that regardless of disease severity, compared to appropriate initial surgical management, managing septic arthritis non-surgically (i.e. needle aspiration and/or antibiotics) does not affect the outcome. Indeed, a study by Goldenberg et al(32) comparing needle aspiration to surgery as an initial mode of treatment in septic arthritis found that neither treatment was superior to the other. However, at our institution, adequate debridement and drainage of the infective focus, which can only be achieved via surgical means, remains the gold standard in successful management of pyogenic joint infections.
Both groups of patients underwent surgical treatment in the form of either open arthrotomy washout or arthroscopic washout, and there was no difference in the type of surgical intervention performed. However, we did observe that patients suffering from GBS septic arthritis required a significantly longer duration of treatment compared to patients with non-GBS septic arthritis (7.67 weeks vs. 5.95 weeks; p = 0.003). This difference in treatment duration can potentially confound intermediate to long-term outcomes rather than inpatient or short-term clinical outcomes (i.e. mortality rates, limb loss, length of hospitalisation, HDU/ICU care, limb loss, requirement of multiple surgeries and readmission rates), which were the main focus of our study.
Our study had several limitations. Firstly, the large diversity of causative organisms within the non-GBS group required the assumption that the septic arthritis caused by the various micro-organisms has similar characteristics. However, we do not have conclusive evidence on this, and our numbers within the group were too small to perform further subgroup analysis. The control arm remains too diverse to make definitive conclusions about the virulence factors of the bacteria and their role in joint infections. Next, because our study straddles a unique time when there was an outbreak of invasive GBS (Serotype III ST283) infections, and 15 out of 21 cases were in the 2015–2016 period, we recognise that our results could be confined to this particular outbreak sequence type and possibly cannot be extrapolated to GBS septic arthritis in general. In addition, the lack of isolate collection and genetic characterisation of all cases of GBS-associated septic arthritis did not allow us to examine the particular risk and virulence factors of the different GBS strains. Lastly, as this was a retrospective study with a relatively short mean follow-up duration of 8.13 months, we were unable to investigate long-term outcome measures such as late recurrence rates and long-term complications such as risk of early osteoarthritis. A follow-up study of this cohort with a longer follow-up duration would hopefully allow us to draw more conclusions about the long-term sequelae of GBS septic arthritis.
This study has yielded several interesting observations on the differences between the two groups of patients. However, these findings are by no means conclusive. The management of all kinds of infections, and not just orthopaedic-specific sepsis, hinges upon understanding the complex interplay among host factors, specific micro-organism virulence factors and treatment-related specifics. Further follow-up and research in the form of prospective matched case-control studies are necessary to help orthopaedic physicians to better manage septic arthritis.
In conclusion, patients with GBS septic arthritis are more likely to be healthy individuals with absence of predisposing comorbidities. They also tend to present with multiple joint involvement and positive blood cultures. In healthy patients presenting with septic arthritis and S. agalactiae bacteraemia, care must be taken to exclude multiple sites of septic arthritis. They should then be treated with appropriate antibiotics as well as surgical drainage of all affected sites.


