Abstract
This paper aims to identify emerging evidence for endolymphatic sac surgery (ESS) in the treatment of Meniere’s disease since the landmark study by Thomsen et al, published in 1998 (conducted from 1981 to 1989). Using the MEDLINE database (PubMed), a systematic review of the literature published from January 1990 to June 2014 was performed. We included all English-language, peer-reviewed randomised controlled trials (RCTs) and controlled studies. Single-arm cohort studies were included if the sample size was ≥ 90 with a response rate > 60%. Altogether, 11 studies fulfilled our inclusion criteria; one was an RCT, two were controlled trials and eight were single-arm cohort studies. There currently exists a low level of evidence for the use of ESS in the treatment of Meniere’s disease. Further studies, in particular RCTs and/or controlled studies, are required to fully evaluate this modality. However, there are difficulties in designing a valid placebo and achieving adequate blinding of observers and investigators.
INTRODUCTION
Meniere’s disease presents difficulties both in terms of diagnosis and management. In medically refractory cases, surgery may be indicated. Options include vestibular neurectomy, labyrinthectomy and endolymphatic sac surgery (ESS). ESS is the more favoured option, as it enables both the hearing and balance functions of the ear to be preserved. However, since the landmark Danish study by Thomsen et al, published in 1998,(1) the applicability of ESS has been called into question. The aim of this paper is to identify emerging evidence for the use of ESS in the treatment of Meniere’s disease.
METHODOLOGY
To identify evidence for ESS in Meniere’s disease, we conducted a search of the MEDLINE database (PubMed) for literature published from January 1990 to June 2014, using the subject headings and keywords ‘endolymphatic sac surgery’, ‘Meniere’s disease’ and their variants. Inclusion criteria were as follows: (a) studies published in English-language, peer-reviewed journals; (b) randomised controlled trials (RCTs) and controlled studies; and (c) single-arm cohort studies with a sample size ≥ 90 and a response rate > 60%. Excluded from the review were studies that employed revision cases and studies in which some other form of concomitant surgery was performed (e.g. vestibular nerve section) or which employed variants of ESS (e.g. enhancement surgery, use of mitomycin).
RESULTS
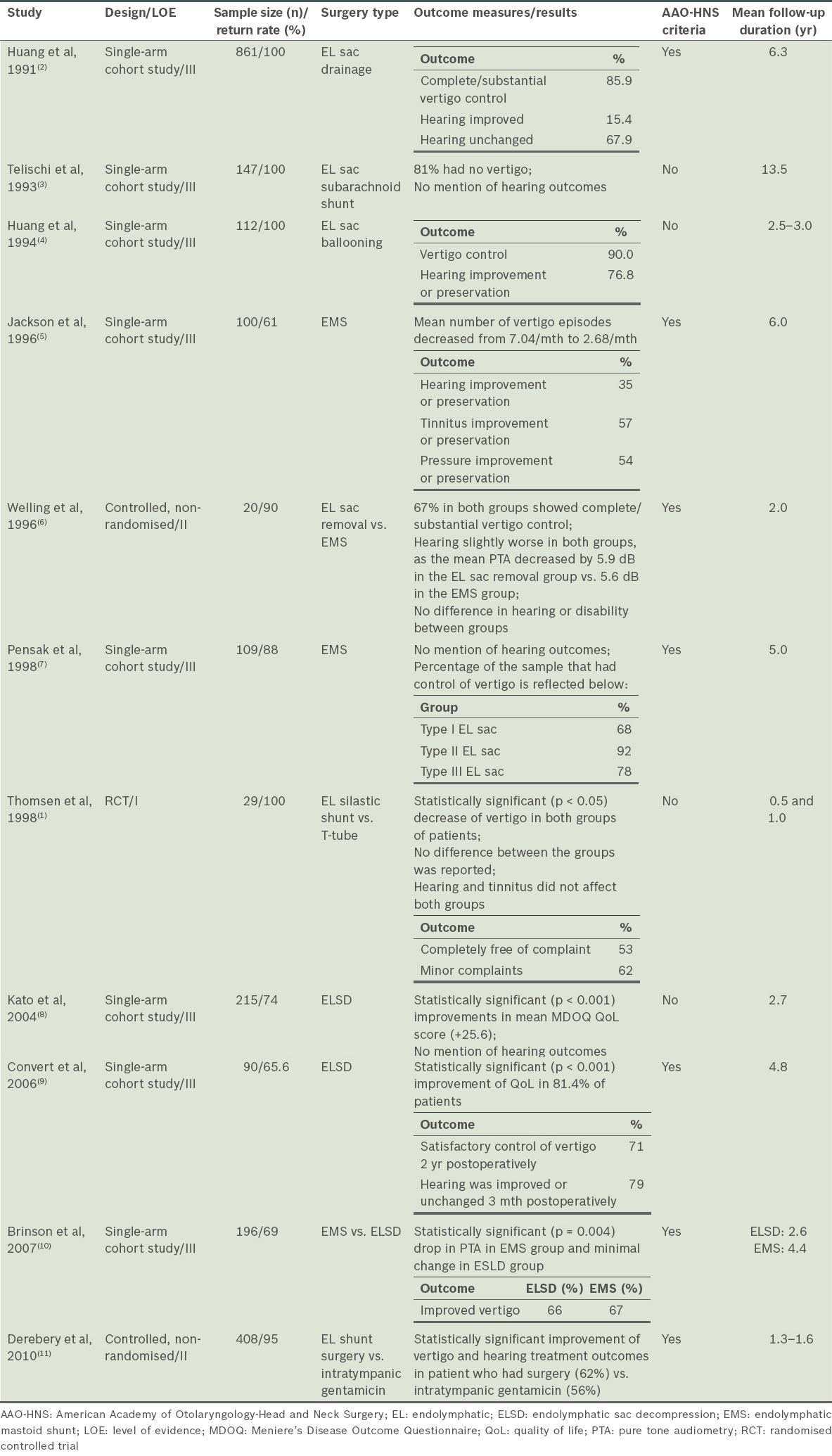
A total of 11 studies(1-11) fulfilled the inclusion criteria.
Table I
Major characteristics and outcomes after treatment of the studies reviewed.
Evidence for a particular treatment modality was categorised to reflect the validity of evidence (
Table II
Levels of evidence.

Seven out of the 11 studies(2,5-7,9-11) employed the American Academy of Otolaryngology-Head and Neck Surgery (AAO-HNS) criteria for determining control of vertigo symptoms. Based on the AAO-HNS criteria, 7%–86% of patients in these studies had complete control of their vertigo symptoms and 14%–56% had substantial improvements in symptoms, following ESS. In combination, 68%–90% of patients had substantial improvement and complete control of symptoms. However, it must be noted that the follow-up period of the various studies ranged from 16 months to 13.5 years, with the outcomes of surgery recorded at different end points. With the exception of four cases of anacusis in the EL sac shunt group,(1) no serious side effects were mentioned in any of the studies reviewed.
Various outcome measures were employed in the 11 studies. Vertigo control featured in all except Kato et al’s study,(8) which used the Meniere’s Disease Outcome Questionnaire (MDOQ); the authors found a statistically significant (p < 0.001) improvement in MDOQ scores following EL sac decompression.(8) Convert et al’s study in 2006(9) utilised both the AAO-HNS criteria and MDOQ questionnaire to assess the outcome of EL sac decompression.
DISCUSSION
History of ESS use
ESS was first described by Portmann in 1927(13) and since then, it has been employed in the treatment of medically refractory Meniere’s disease.(14) However, the efficacy of ESS was called into question in the 1980s when the Danish study by Thomsen et al(1) found no significant difference in vertigo relief between patients who had undergone the surgery and those who had undergone simple mastoidectomy. The same results applied at the one-, three- and nine-year follow-up. Despite these findings, the study’s small sample size has been criticised and the validity of using simple mastoidectomy as a placebo operation questioned.
Pathophysiology of endolymphatic hydrops and postulated causes of vertigo
Although there are still uncertainties surrounding the aetiology of Meniere’s disease, it is generally agreed that the underlying pathology is that of EL hydrops, which may be caused by overproduction of endolymph at the stria vascularis or compromised absorption of endolymph.(2) Absorption is thought to occur primarily at the EL sac.(6) Besides absorbing endolymph, the EL sac also secretes glycosaminoglycans. This is thought to be in response to pressure changes and produces an osmotic gradient to attract fluid from the cochlea and vestibule into the sac. A damaged sac may impair the secretion of these proteins and contribute to EL hydrops.(6)
The cause of vertigo in EL hydrops is controversial. Schuknecht(15) was the first to propose that vertigo is secondary to the rupture of Reissner’s membrane due to high pressure in the EL system. Alternatively, Shea(16) suggested that vertigo may be caused by a simple distension of the membranous labyrinth without rupture. Gibson and Arenberg(17) proposed that the rapid movement of fluid out of the inner ear in response to the secretion of osmotic particles is the cause of vertigo.
Rationale for ESS
The primary aim of ESS is to reduce vertigo while maintaining or improving hearing. The available forms of ESS include: (a) simple decompression with or without ballooning; (b) insertion of a shunt; and (c) ablation of the EL sac. The rationale behind the first two methods is in line with the theories of Schuknecht(15) and Shea(16) that draining the EL sac by simple decompression or shunting may reduce vertigo by decreasing the pressure in the EL system. Ablation of the EL sac, the third method, is more in line with the theory proposed by Gibson and Arenberg,(17) in which destruction of the EL sac prevents the secretion of osmotically active glycosaminoglycans that contribute to dizziness. All three methods have been shown to be effective. Thus, it is possible that the aetiology of vertigo may be multifactorial.
Natural history of Meniere’s disease
As the majority of studies in our review are single-arm cohort studies without a control group, it is important to interpret the results of these studies in light of the natural history of the disease. Despite the different rationales behind ESS, there is evidence in the literature to suggest that the natural progression of the disease is spontaneous resolution. In particular, we highlight the study by Silverstein,(18) which compared long-term vertigo control rates between post-EL sac surgical patients and those for whom surgery was recommended but declined. The study found no difference in the long-term vertigo control rates between the two groups (i.e. 70% complete control at 8.7 years and 71% complete control at 8.3 years, respectively). Similarly, a second study by Quaranta et al(19) (not included in our review because it did not fulfil the inclusion criteria of sample size ≥ 90) showed that, although in the first 2–4 years, vertigo control was better in the EL sac mastoid shunt group vs. the non-operative group, longer-term (i.e. six years) vertigo control rates were not statistically different between the two groups (85% vs. 74%). However, Stahle et al’s study(20) found that the frequency of vertigo attacks in Meniere’s patients declines only slightly over a 20-year period and that disability from dizziness may in fact increase with time. Taken together, it appears that the natural progression of Meniere’s disease varies, which reinforces the view that the outcomes of single-arm cohort studies should be interpreted with caution.
Evidence for ESS
Based on our literature review, since Thomsen et al’s study in 1998, no other RCT that studied the effects of ESS has been published. Thomsen et al(1) found no difference in efficacy between the EL silastic shunt and T-tube placement. This paper raised the possibility of the placebo effect of ESS. However, the sample size used in the study was small, making it difficult to draw any definitive conclusions regarding the true value of ESS.
The study by Welling et al,(6) which compared two forms of ESS, was relatively more impactful. However, the absence of a non-ESS control group reduces its level of evidence from IIa to III. A third study by Derebery et al,(11) which compared EL sac shunt surgery with intratympanic gentamicin, showed a small but statistically significant (p < 0.001) difference in vertigo control rates, thus favouring surgery. This would effectively be a level II study, as it included a control group. However, the control group consisted of patient data obtained from previous studies, which may be a limitation in interpreting the results.
The other eight single-arm studies provided level III evidence for ESS. These studies demonstrated both short-(2,4,5,7-10) and long-term(3) (i.e. more than ten years) control of vertigo. In the majority of these studies, vertigo control was achieved in at least 80% of the patients. The problem with interpreting level III studies is that the absence of a control means the study could potentially be confounded by the placebo effect of surgery as well as improvement of symptoms due to the natural progression of disease.
Furthermore, three of these studies(3,7,8) did not mention hearing outcomes, an important end point in any intervention of Meniere’s disease. The remaining five studies(2,4,5,9,10) showed variations in hearing preservation or improvement rates (i.e. improvement of 15.4%–79%). Interestingly, the studies that achieved better hearing preservation or improvement involved simple EL sac drainage or ballooning surgery,(2,4) whereas the study that achieved lower hearing preservation or improvement rate involved the use of a shunt.(5) The results of Brinson et al’s study(10) also showed a statistically significant (p = 0.004) deterioration in pure tone audiometry (PTA) results in the EL mastoid shunt group, whereas this was not observed in the EL sac decompression group, further supporting the aforementioned point.
It should be noted that, although some of the studies employed a comprehensive workup in the diagnosis of Meniere’s disease,(2,4,7,9) only Brinson et al’s study(10) used both the American Academy of Ophthalmology and Otolaryngology (AAOO) and AAO-HNS criteria to make the diagnosis. However, the majority of the studies employed the AAOO/AAO-HNS criteria in reporting the results of vertigo in the control group.(1,2,5-7,9-11)
The study by Kato et al(8) merits further discussion, as it employed a disease-specific, quality of life questionnaire to assess responsiveness to treatment. The results of their study showed significant improvements in the actual quality of the patient’s life, including vertigo control, following ESS. In addition to measuring disease-specific symptoms, they also analysed three other aspects that contributed to quality of life, namely physical, mental and social wellbeing. These results are similar to those in Convert et al’s 2006 study,(9) which showed that 81.4% of their patients had a significant improvement in quality of life, based on the MDOQ and AAO-HNS criteria.
The 2002 study by Huang(12) initially met our inclusion criteria but was eventually excluded for reasons stated earlier. Furthermore, this paper documents the personal experience of the author, and thus provided a low level of evidence for the efficacy of ESS. It includes the following observations: (a) the typical abnormally high summating potential and action potential ratio in Meniere’s disease (using intraoperative electrocochleography) decreases during sac incision and endolymph drainage; (b) class A patients (AAOO 1972 criteria) have better long-term results, which makes the placebo effect less probable, as it is unlikely to be class selective; and (c) in cases where conspicuous seepage of endolymph occurs during surgery, there are anecdotal reports of a dramatic reduction in aural fullness and often improvements in hearing. These observations provide level IV evidence for ESS in the treatment of Meniere’s disease.
Evidence for EL sac decompression vs. shunt surgery
In analysing the efficacy of EL sac decompression vs. shunt surgery, it is difficult to draw definitive conclusions, as the assessment time-point is different for each of the studies reviewed. Moreover, as the surgeons involved in the various studies are different, surgeon bias and experience cannot be ruled out.
The study by Brinson et al(10) showed that EL mastoid shunt surgery and EL sac decompression led to significant improvements in 67% and 66% of the patients, respectively. The authors concluded that both interventions were equally effective in reducing the incidence and severity of vertigo attacks. However, patients who underwent EL mastoid shunt surgery had a statistically significant (p = 0.004) decline in PTA results compared with a minimal decline in those treated with EL sac decompression. One confounding factor could be that patients who underwent EL sac decompression were more likely to have transtympanic dexamethasone delivered both pre- and postoperatively, which might have been a contributing factor to the improvement observed in these patients.
Convert et al(9) categorised patients who had EL sac decompression into those who had a silastic stent inserted to maintain the opening in a narrow, fibrous sac and those who had a simple decompression. Although the study found no significant difference in outcome, it must be noted that the small sample size did not allow any difference to be fully appreciated. Hence, the current evidence is insufficient to determine if EL sac decompression is better than EL mastoid shunt surgery in controlling vertigo and hearing symptoms.
Mastoid vs. subarachnoid shunts
No definitive conclusions could be made regarding the difference in efficacy between mastoid and subarachnoid shunts, as the 11 studies reviewed had minimal analysis done. However, one notable study published by Brackmann and Nissen in 1987(21) demonstrated that equivalent results (using vertigo control as an outcome measure) were obtained with the two shunts. This study also reported that the use of the mastoid shunt yielded lower morbidity. Hence, an argument could be made for the use of the mastoid shunt over the subarachnoid shunt.
Evidence for EL sac ballooning vs. primary sac surgery
Endolymphatic sac ballooning surgery (ESBS) involves the insertion and fan-folding of a thin silastic sheet into the EL sac. ESBS is characterised by expansion of the EL sac lumen with preservation of EL sac integrity. It is, however, difficult to make a direct comparison between ESBS and standard ESS in our review due to the inevitable differences in parameters measured and controlled for in a systematic review.
However, an analysis of two studies performed by Huang et al(2,4) allows for a more valid comparison. The initial series of 339 ESS cases, followed up over 45 months and published in 1991, had a success rate of about 80% (based on AAO 1972 criteria).(2) This result was compared with that of their second series of ESBS cases published in 1994.(4) In the latter study, cases were followed up for a mean of 30 months, and a success rate of about 90% was reported. The slight difference in mean follow-up time between the two groups may suggest the superiority of ESBS over ESS.
EL shunt surgery vs. intratympanic gentamicin
Derebery et al’s 2010 study(11) compared 183 patients who underwent EL sac shunt surgery against 203 patients who were treated with gentamicin (obtained from six published gentamicin treatment studies). Their results showed that both the surgical and gentamicin groups had a statistically significant (p < 0.001) improved outcome for combined vertigo and hearing, although the shunt group had a better outcome than the gentamicin group (i.e. 62% vs. 56%). The use of vertigo and hearing improvement as a combined outcome was unique to this study. However, the authors also reported that there was a statistically significant (p < 0.003) variability in outcome across patients who had gentamicin injection. This was postulated to be due to a mutation in mtRNA (A1555G), which has been linked to aminoglycoside-induced and non-syndromic sensorineural deafness. Hence, before intratympanic gentamicin can be used to great effect, the factors that affect its outcome must be better isolated.
CONCLUSION
There is currently low (level III) collaborative evidence for ESS in the treatment of Meniere’s disease. Evidence provided by this review, however, is potentially confounded by the placebo effect and the natural progression of disease. There is no large, well-designed randomised controlled trial to provide clear and conclusive evidence on the subject. To date, only one controlled study in the literature has suggested the benefit of ESS over intratympanic gentamicin. Comparisons between EL sac decompression and shunt surgery, as well as EL mastoid and subarachnoid shunts, cannot be easily made due to various confounding factors and different study designs, although there is some evidence that ESBS surgery may be more efficacious than primary ESS.
In order to fully evaluate the efficacy of ESS for the treatment of Meniere’s disease, randomised, double-blind, placebo-controlled trials with better designs are required. Large controlled studies are also necessary to eliminate the confounding effects of natural disease progression. Nevertheless, there still remain difficulties in choosing a suitable control group and adequately blinding subjects and observers.


