Abstract
INTRODUCTION
Lactogenesis II (LaII) failure can be prevented in at-risk mothers with simple proactive interventions. In a randomised trial, we investigated the efficacy of early and regular breast milk expression in establishing LaII, using an electric double-breast pump.
METHODS
Mothers with uncomplicated singleton deliveries were randomised to intervention (n = 31) or control (n = 29) groups. The former commenced breast milk expression with an electric pump within one hour of delivery and maintained regular expression with direct breastfeeding. Control mothers directly breastfed without regular pump expression. Expressed milk volumes were analysed for citrate, lactose, sodium and protein.
RESULTS
Median time of LaII was Day 3 (interquartile range [IQR] 1 day) with intervention and on Day 4 (IQR 1 day) among controls (p = 0.03). Biochemical steady-state concentrations were achieved around early Day 4 (sodium, total protein) and Days 4–5 (citrate, lactose). Sodium, protein and lactose levels were similar in both groups over seven days, at 5.80 mM, 0.68 mM and −13.38 mM, respectively. Mean daily milk volume with intervention was 73.9 mL on Day 3 and 225.2 mL on Day 7, greater than controls (25.4 mL on Day 3 and 69.2 mL on Day 7; p < 0.2). Mean infant weights were similar on Day 8 at 3,477 g with intervention and 3,479 g among controls.
CONCLUSION
LaII is established by postnatal Day 3 with early initiation of regular breast milk expression, a useful intervention for mothers at risk of early-onset breastfeeding failure.
INTRODUCTION
Exclusive breastfeeding (EBF) rates in developed countries are currently below the proposed global target of at least 50% during the first six postnatal months, as determined by the United Nations System Standing Committee on Nutrition in 2012. About one-third of mothers practise EBF, with lower rates expected in developed countries.(1) Although the incidence of initial breastfeeding reached 83% between 2005 and 2010 in the United Kingdom (UK), only 31% of babies remained on EBF at six weeks,(2) while in Singapore in 2013, only 1% of babies remained on EBF at six months despite a high initiation rate of 99%.(3) Given the many established health benefits, a concerted effort to overcome identifiable obstacles to sustained breastfeeding is urgently required, particularly in at-risk mothers.(4,5)
Lactogenesis II (LaII), also known as secretory activation,(6) is the onset of copious milk production, which typically occurs at 40–72 postnatal hours,(7) and increases rapidly and plateaus at ~96 postnatal hours.(8) Conditions associated with delayed LaII (DLII, onset > 72 postnatal hours)(9,10) include maternal obesity,(11) insulin-dependent diabetes mellitus,(12) preterm delivery(13) and Caesarean section.(14,15) As DLII is an important contributing factor to early breastfeeding cessation,(16,17) interventions that reduce DLII should be implemented in all maternity units. Evidence suggests that breastfeeding initiation within a few hours of birth decreases LaII failure in at-risk women.(18,19) Because the initial interaction between mother and baby may be iatrogenically delayed owing to operative delivery or perinatal complications,(20-24) a sub-group of at-risk women can be identified for early assessment and intervention in the maternity unit. Milk expression with electric breast pumps (EBPs) is a common practice in Singapore, with 68.5% of Chinese mothers in a local study using pump-expressed breast milk at three months.(25) EBPs are often utilised for milk collection and storage for future feeds or to determine the volume of milk fed to the infant. Early initiation independently contributes to improved breastfeeding rates at six months.(26) In postpartum situations that physically prevent direct breast feeding, early breast milk expression by at-risk mothers may fulfil a similar role.
In this randomised study, we investigated the hypothesis that DLII is reduced when EBP expression is initiated early and performed regularly after delivery. Specifically, we determined the time at which LaII was successfully established by coupling simple observational assessment with biochemical validation, and analysed total daily milk volume expressed and infant weight change between the start and end of the first postnatal week.
METHODS
This study was funded by a National Healthcare Group (NHG) research grant, NHG RPR 04027, and was conducted with the approval of the Domain Specific Review Board (DSRBD/04/119). The trial was registered with ClinicalTrials.gov (NCT00393640). Recruitment and interventions were initiated at the National University Hospital (NUH), Singapore.
Studies on lactation following preterm delivery have shown compromised initiation of lactation in up to 82% of mothers.(13) Based on this, we required a sample size of 30 women in each arm to show a 50% reduction in DLII incidence with a significance level of 0.05 and a power of 90%. We recruited women with term deliveries (≥ 37 completed weeks of gestation) who met the eligibility criteria for the trial: minimum age of 21 years, a desire to breastfeed, agreement to use the breast pump and no contraindications to breastfeeding. All patients provided written informed, independent consent to trial participation and were subsequently randomised by minimisation of the stratification factors of previous live births and breastfeeding experience (
Fig. 1
Flowchart shows the enrolment and randomisation processes.
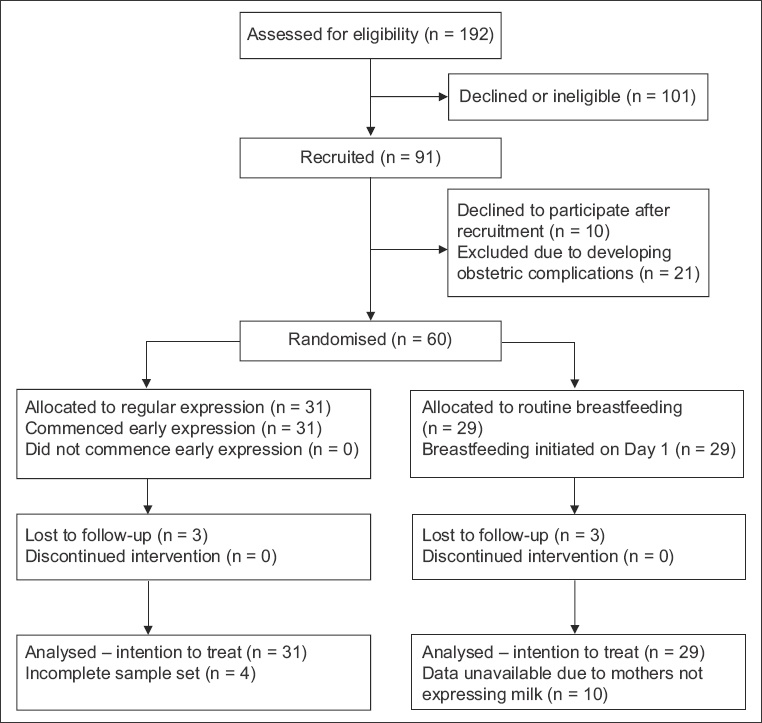
Patients with uncomplicated singleton deliveries were assigned to the intervention or control groups at the time of recruitment according to a computer-generated randomised list, but were only informed of their specific assignments after delivery. Exclusion criteria included maternal diabetes mellitus, hyperprolactinaemia, congenital fetal anomalies and multiple pregnancies. A standard questionnaire was used to record demographic information, details of the home environment and attitudes towards breastfeeding. All mothers had access to literature on the benefits of breastfeeding and were given the NUH Breastfeeding Booklet. The intervention group had a strict regimen of breast milk expression starting within one hour of delivery and were instructed to continue two- to three-hourly expression throughout the week, in addition to direct breastfeeding whenever desired. Participants were visited by the research coordinator within the first postnatal hour and educated on the use of a double-flange EBP (Ameda Egnell or Pump In Style®, Medela AG, Baar, Switzerland). Simultaneous breast expression was performed six to eight times during the day and at least once at night, with each session limited to 15 minutes. Participants were taught to feed expressed breast milk (EBM) to infants by spoon or cup to avoid nipple confusion. They were also encouraged to breastfeed directly after expression. Mothers in the control group were given appropriate instructions on expression and storage of breast milk and were allowed independence in determining their feeding schedules (for both direct and expressed milk feeds).
In addition to providing the required expressed milk samples, mothers from both control and intervention groups were encouraged to breastfeed or express milk according to hospital recommendations and were expected to use their own breast pumps. The primary outcome measure was maternal perception of copious milk flow, the most commonly used indicator of LaII. Secondary outcome measures included biochemical changes in breast milk, volume of breast milk expressed daily, and type and frequency of breastfeeding. Mothers in both groups collected breast milk samples twice daily from the day of delivery for the first week. The first 24 hours counted as Day 1, and total daily breast milk volume was recorded over each subsequent 24-hour period for seven days. All mothers recorded EBM volume using a calibrated syringe, after which the EBM was used immediately or stored appropriately for future use. Mothers recorded the total volume expressed at the end of each 24-hour period in their breastfeeding diaries. 1 mL of milk was extracted from the first and last yields of the day for biochemical analyses. Syringes and polypropylene containers were used to aliquot and store EBM samples at −20°C in the mother’s home freezer until collection at the end of the week. Long-term samples were stored at −80°C.
Neonatal feeding data consisting of childbirth, perinatal and infant feeding details were collected via standardised forms at the first postnatal visit conducted at Week 1. In the first week, participants prospectively recorded in their breastfeeding diaries their perception of when their milk first came in (as an indicator of LaII),(27) EBM volume, frequency and duration of direct breastfeeding, other fluids fed and breastfeeding problems encountered (if any). Exclusive breast milk feeding was defined as feeding only breast milk (expressed or direct), while mixed feeding was defined as giving breast milk with formula in any amounts. Infant weights were recorded using a Seca 334 scale (seca Deutschland, Hamburg, Germany) on Days 2 and 8.
For biochemical analysis of samples, batch testing of EBM samples was performed to determine the concentrations of total protein, citrate, lactate and sodium. Milk samples were thawed at room temperature, centrifuged and defatted as described by Arthur et al.(7) Total protein, sodium, citrate and lactose concentrations were assayed using methods previously described.(13)
Descriptive statistics were reported as mean ± standard deviation for continuous variables and percentages for categorical variables. Differences between groups were assessed using chi-square analyses, apart from infant weight, which was assessed by independent sample t-test. We serially examined milk volume and the corresponding biochemical profile (i.e. sodium, lactose, citrate and protein concentrations) over seven days with linear mixed-effects models that took into account the correlation between repeated measures from the same individual and incomplete outcome data, using the assumption that it was missing at random. Maximum likelihood estimation was used and a diagonal working covariance matrix for random effects variables (intercept and slope) was chosen. The Akaike information criterion statistic facilitated the model selection, and final models included time-intervention interactions to estimate the change in milk volume and biochemical profile over time between intervention groups. All statistical analysis was conducted using IBM SPSS Statistics version 19.0 (IBM Corp, Armonk, NY, USA) and GraphPad Prism Version 6.04 (GraphPad Software, La Jolla, CA, USA).
RESULTS
A total of 192 women were approached for this study and 91 were initially recruited. After excluding women with pregnancies complicated by diabetes mellitus, hyperprolactinaemia and congenital anomalies, or with multifetal pregnancies, 60 mothers were randomised into the intervention group (n = 31) and control group (n = 29,
Table I
Demographic data of women enrolled into the regular expression and routine breastfeeding groups.
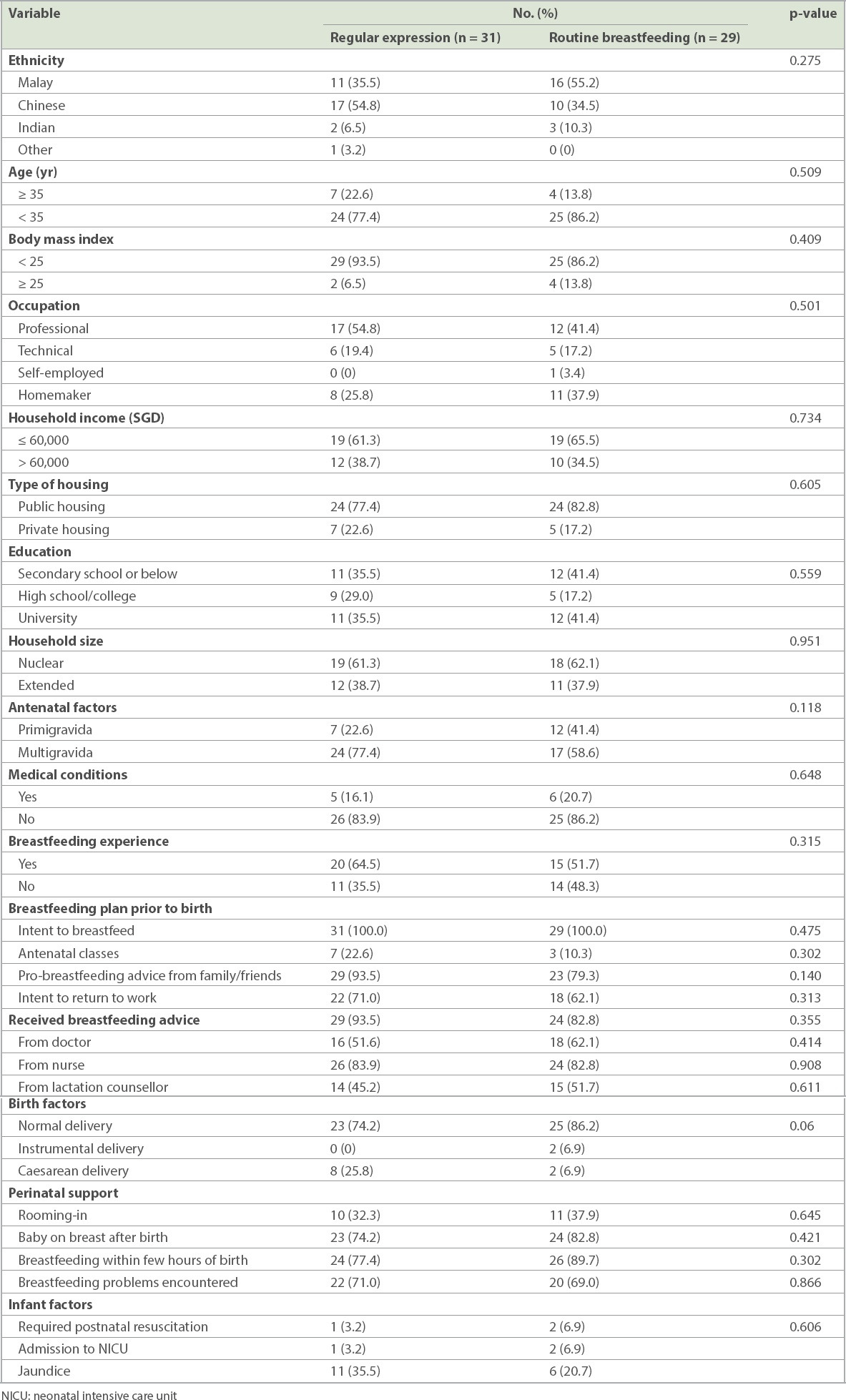
Mothers in the intervention group described copious milk flow from Day 2 onwards, except for two mothers who recorded this event in the first half (Day 1.0) and second half (Day 1.5) of Day 1, with the median time being Day 3 (72 completed hours) (interquartile range 1 day). Among the control group, copious milk flow was first reported on Day 2, with the median time being Day 4, when 14 mothers reported copious milk flow (
Fig. 2
Charts show a comparison of the first perception of breast fullness and copious milk flow between (a) the intervention group and control group; and (b) Malay and Chinese mothers.
When mothers of different races were compared, Chinese and Malay mothers demonstrated comparable time points at which copious milk flow was reported (
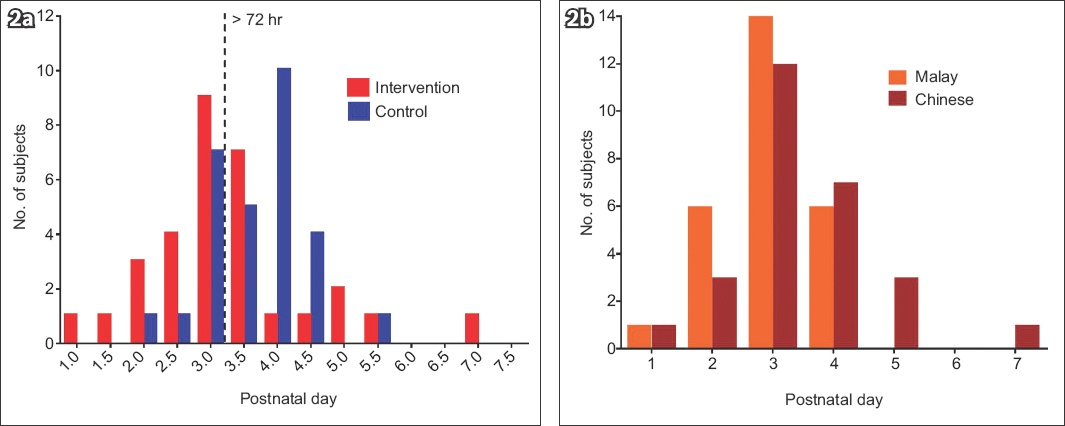
Regarding the biochemical levels of the two groups, sodium and total protein concentrations peaked on Day 1 and reached steady state at around early Day 4, while citrate and lactose levels achieved steady state at Days 4–5. Over the seven-day period, when mean concentrations of the biochemical markers were compared between the intervention and control groups, we found no significant differences in sodium, protein and lactose levels, which were 5.80 (95% CI –3.03, 14.62) mM, 0.68 (95% CI −2.38, 3.75) mM and 13.38 (95% CI −28.73, 1.97) mM, respectively (Figs.
Fig. 3
Charts show a comparison of breast milk concentrations of (a) sodium; (b) total protein; (c) citrate; and (d) lactose between the intervention and control groups.
Mothers in both groups fed their infants either breast milk, alone or supplemented with water only, or a combination of breast milk and milk formula (
Fig. 4
Charts show a comparison of (a) breastfeeding practices and (b) intervals among mothers in the two groups.
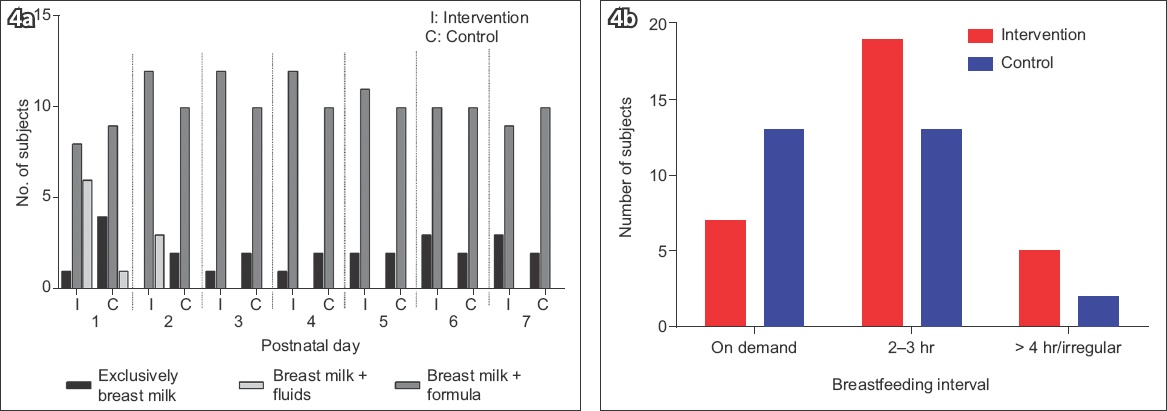
Overall, the mean difference in EBM volume for intervention versus control group was 73.9 (95% CI 46.3, 152.3) mL on Day 3 and 225.2 (95% CI 98.1, 352.3) mL by Day 7 (p < 0.02). There was significant interaction between EBM volumes and time (F-statistic = 2.28, p = 0.049). In the first 48 hours, the mean EBM volume in the intervention group was similar to that of the control group, but it was significantly higher (p < 0.02) from Day 3 to Day 7 (99.3 [95% CI 46.3, 152.3] mL to 294.4 [95% CI 179.3, 409.5] mL) compared to the control group (Day 3: 25.4 [95% CI 3.9, 46.9] mL to Day 7: 69.2 [95% CI 32.3, 106.1] mL) (
Fig. 5
Charts show (a) expressed breast milk (EBM) volume in the intervention group, who had regular milk expression; (b) comparison of formula feeds and total daily fluid volume fed in the intervention and control groups; and (c) comparison of mean infant weights in the two groups from Day 2 to Day 8.
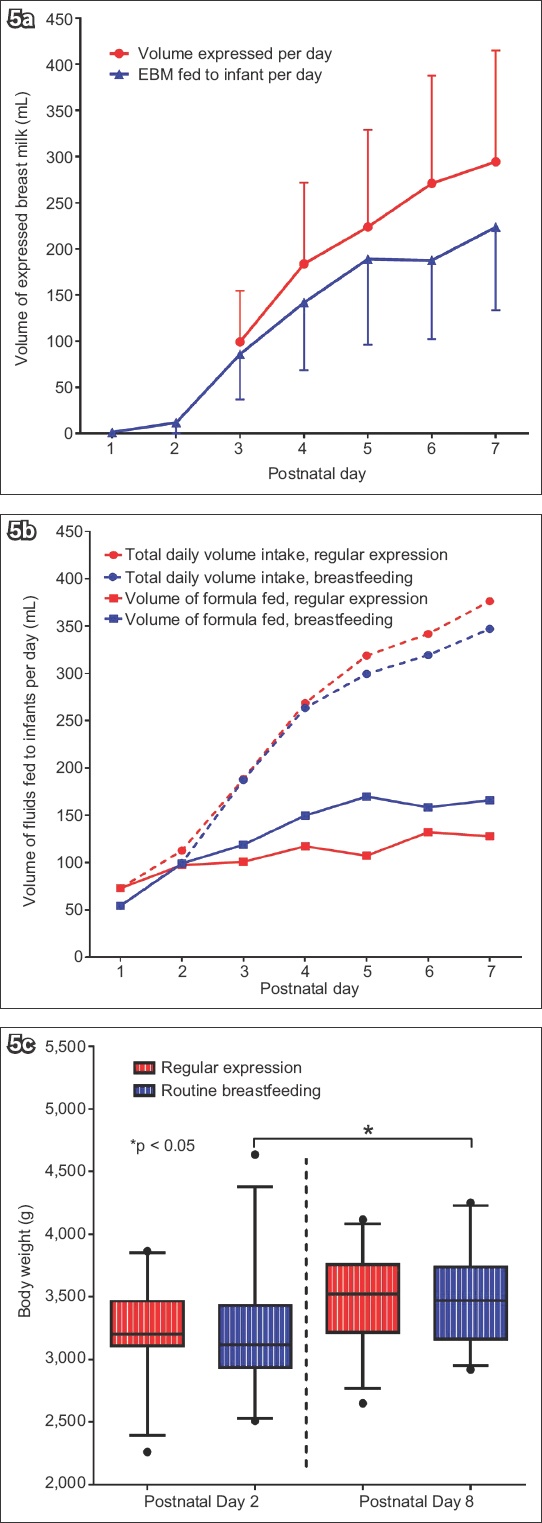
Milk formula was fed to infants in both groups to supplement breast-milk feeds at the mothers’ discretion. Comparing the intervention and control groups, there was no significant difference in the mean daily volumes of formula (108.0 mL vs. 130.9 mL,
Babies were weighed at the start and end of the first postnatal week. There were no differences in mean weights between the intervention and control groups, which were 3,227 (95% CI 3,094, 3,359) g and 3,218 (95% CI 3,034, 3,402) g, respectively, on Day 2 (
DISCUSSION
In this study, the majority of women experienced voluminous flow on around the third to fourth postnatal days, an event often interpreted by nursing mothers as confirmation of successful breastfeeding initiation. Earlier onset LaII and a trend towards a lower incidence of DLII was observed in mothers performing a simple intervention of regular breast-milk expression using a standard EBP compared to unscheduled expression.
All mothers had copious flow by the end of Day 5, with the exception of one patient. Control group mothers reported copious flow by Day 4. The biochemical profile of the breast milk demonstrated the expected changes that indicate LaII, with decreasing sodium and total protein, and increasing citrate and lactose. Biochemical steady states for each marker were reached around postnatal Day 4 in both groups, responding to clinical onset. While there is no significant difference in biochemical content between both groups on Day 4, earlier perception of lactogenesis, i.e. maternal perception of copious milk flow within the intervention group may result in greater confidence with establishing breast milk supply, a lower likelihood of supplementing feeds with formula, possibily resulting in a higher breastfeeding yield.
No ethnicity-specific breastfeeding practices were observed during the first week, but the number of participants reporting on breastfeeding practices was small. In support of other studies, our data confirms a direct correlation between milk volumes and frequency of expression and an inverse correlation with delivery-to-milk expression interval.(28) Past studies have looked into the effects of breast pump suction patterns on the initiation and continuation of lactation for premature infants,(29) as well as the usage of manual methods of milk expression coupled with electrical pumps for preterm infants.(30) Examining breast milk expression and its frequency hence proves critical in furthering our understanding of breast milk supply for infants.
Mothers often encounter barriers to early direct breastfeeding, particularly when perinatal complications preclude early physical contact between mother and infant.(31,32) Because early removal of breast milk is associated with successful transition to LaII, our data supports the clinical utility of scheduled expression for at-risk women. This proactive intervention is relatively easy, low cost and can be initiated with widely available equipment, while possibly avoiding pharmacological agents. It can be applied to women who are too ill to breastfeed after delivery, to enable lactation initiation in preparation for breastfeeding when they recover, and to mothers of multiples, preterm or low-birth-weight infants at higher risk of breastfeeding failure.(33) Encouraging such a practice would be helpful in communities with suboptimal breastfeeding initiation and where early cessation is prevalent due to maternal perception of insufficient milk. Although this intervention might not appeal to every mother who intends to breastfeed, with judicious antenatal education and proper case selection, a protocol of early and regular breast milk expression can be a suitable addition to a healthcare policy promoting breastfeeding, giving women an acceptable alternative to initiating LaII if direct early breastfeeding is not feasible. There are few contraindications to using a breast pump if the mother or her caregiver is physically able to perform the necessary manoeuvres. Possible obstacles to widespread implementation of this practice include access to and the expense of acquiring hospital-grade EBPs with a double collection kit,(34) since simultaneous breast emptying is more efficient than consecutive single-breast emptying.(35)
An important factor influencing the smooth transition from lactogenesis I (gestational development of mammary lactocytes) to LaII that this practice emulates is the early removal of breast milk, acting in concert with progesterone withdrawal and other perinatal factors.(36,37) Early initiation of breastfeeding within the first half hour of life is the fourth step in the Baby-Friendly Hospital Initiative criteria,(38) and is a critical strategy in the effort to elevate breastfeeding rates, contributing to significantly higher total milk production and earlier LaII in mothers of very-low-birth-weight babies.(19) This is in contrast to the decreased milk production associated with late initiation (up to six days postpartum) in a similar cohort.(28) Another important contributor is complete breast emptying. Maternal milk production and the breast’s storage capacity both respond closely to the infant’s demand for milk and completeness of breast emptying. Proper breast drainage also improves milk production efficacy, by increasing the capacity of the breast to produce milk in excess of the volume needed to satisfy the infant’s hunger.(39) Studies of milk production in dairy animals and humans have identified a local inhibitory factor that caused reversible disruption of milk flow, via local feedback inhibition, in a dose-dependent manner in the absence of effective milk removal, subsequently contributing to failed LaII.(37,40)
Encouraging sustained and exclusive breastfeeding following international recommendations is a key strategy in national programmes aimed at addressing inequalities in health and improving population well-being.(41,42) The difficulty of sustaining this practice is underlined by the decline in breastfeeding rates over the course of the neonatal period. While initiation rates were encouragingly high, from 74% in the United States (US, 2006 data) and 83% across the UK (2010 data), to 99% in Singapore (2013 data), continued breastfeeding of any type declined steeply by six months to reach 42% in Singapore (EBF, 1%), 43.5% in the US (EBF, 14.1%) and 25% across the UK (EBF, negligible).(2,3,41,43,44) There is an unfulfilled opportunity for obstetric healthcare providers to support breastfeeding women by offering a safe and effective intervention that may positively impact the quality and duration of breastfeeding.
Among the strengths of this study are that it provides useful information about the expected breast milk volume in our local Asian population consisting predominantly of Malay and Chinese women, which are substantially lower than reference volumes derived from relatively homogenous Caucasian populations. Mean EBM volume by Day 5 was around 220 mL/day, and total fluid volumes consumed by infants approximated 230 mL/day. The regaining and surpassing of birth weight within the first week demonstrate that these infants were not compromised by lower milk consumption and should reassure Asian mothers that their breastfeeding efforts are likely to be sufficient.
The limitations of this study include not being able to accurately gauge the amount of breast milk consumed by breastfed infants in the control group, as we were not able to test weigh the infants after feeds. This data would have informed us about the true volume intake of breastfed infants. Furthermore, these interventions were designed not to interfere with the normal postnatal activities of trial participants; thus, we have several incomplete data sets because collection of samples and recording of data were not strictly enforced. Additionally, the data set is incomplete, as several mothers were uncontactable after they were recruited. Mothers were also not surveyed about their satisfaction with their individual breastfeeding experience, which would serve as a plausible research focus for future papers.
In conclusion, early and regular breast milk expression significantly contributes to the timely establishment of LaII, probably through mechanisms involving frequent and complete removal of intramammary inhibitors of breast milk production. This was supported by appropriate changes in biochemical markers by Day 3 in both groups. This simple intervention may be beneficial to women who are at risk of delayed LaII and premature breastfeeding cessation, and may contribute to promoting breastfeeding rates in industrialised countries.
ACKNOWLEDGEMENT
This study was funded by the National Healthcare Group grant NHG RPR 04027.


