Abstract
INTRODUCTION
We compared the effects of various surgical positions, with and without the Valsalva manoeuvre, on the diameter of the right internal jugular vein (RIJV).
METHODS
We recruited 100 American Society of Anesthesiologists physical status class I patients aged 2–12 years. The patients’ heart rate, blood pressure, peripheral oxygen saturation and end-tidal CO2 pressure were monitored. Induction of anaesthesia was done using 1% propofol 10 mg/mL and fentanyl 2 µg/kg, while maintenance was achieved with 2% sevoflurane in a mixture of 50/50 oxygen and air (administered via a laryngeal mask airway). The RIJV diameter was measured using ultrasonography when the patient was in the supine position. Thereafter, it was measured when the patient was in the supine position + Valsalva, followed by the Trendelenburg, Trendelenburg + Valsalva, reverse Trendelenburg, and reverse Trendelenburg + Valsalva positions. A 15° depression or elevation was applied for the Trendelenburg position, and an airway pressure of 20 cmH2O was applied in the Valsalva manoeuvre. During ultrasonography, the patient’s head was tilted 20° to the left.
RESULTS
When compared to the mean RIJV diameter in the supine position, the mean RIJV diameter was significantly greater in all positions (p < 0.001) except for the reverse Trendelenburg position. The greatest increase in diameter was observed in the Trendelenburg position with the Valsalva manoeuvre (p < 0.001).
CONCLUSION
In paediatric patients, the application of the Trendelenburg position with the Valsalva manoeuvre gave the greatest increase in RIJV diameter. The reverse Trendelenburg position had no significant effect on RIJV diameter.
INTRODUCTION
Central venous catheterisation (CVC) refers to the insertion of a catheter into a vein that empties directly into the heart. Central venous catheters come in different diameters (2–15 mm), lengths (20–60 cm) and lumen diameters (1–4 mm). The type of catheter used depends on its purpose and entry site (peripheral or central) and the patient’s age. The material used to make catheters is chemically inert (so as not to encourage thrombus formation), flexible and radiopaque.(1)
The advent of clinical ultrasonography (US), which can visualise vascular anatomy very clearly,(2) revolutionised CVC. US-guided catheterisation is often successful in situations where the anatomical landmark (AL) technique fails.(1) Furthermore, US-guided catheterisation is associated with fewer complications.(3) US guidance for venous interventions was first described in 1984,(4) and since then, its benefits have been clearly demonstrated.(5) In 1999, Keyes et al utilised US successfully for peripheral venous access in emergency medicine,(6) reporting a 91% rate of successful cannulation when access to the brachial artery was 2%. Furthermore, US can be performed at the bedside, does not involve ionising radiation, and is cheap and time efficient.(7,8)
Gordon et al(9) showed in their study that increasing the cross-sectional area of the jugular vein facilitates catheterisation and reduces complications. While the Valsalva manoeuvre is one of the most effective methods to increase jugular vein cross-sectional area, positive intrathoracic pressure and positive end-expiratory pressure (PEEP) in patients under anaesthesia have similar effects.(10-13) Trautner et al(14) showed that in the Trendelenburg position, internal jugular vein size is directly proportional to PEEP.
The aim of the present study was to examine the effect of head-down and head-up tilts (i.e. Trendelenburg and reverse Trendelenburg positions, respectively), with and without the Valsalva manoeuvre, on the diameter of the right internal jugular vein (RIJV) of paediatric patients.
METHODS
The prospective and randomised study was performed in the Health Application and Research Center of the Uludag University Faculty of Medicine (UUFM). Written approval was obtained from the Research Ethics Committee. Patients who participated in the study were admitted for paediatric surgery (i.e. circumcision, anoplasty, orchidopexy, hypospadias repair, hydrocelectomy, lymphadenopathy excision, cystoscopy, vaginal dilatation, ureteroneocystostomy and inguinal hernioplasty). A total of 100 patients, aged 2–12 years and categorised as American Society of Anesthesiologists (ASA) physical status class I, were included in the study. Written consent was obtained from all parents after they were verbally informed about the purpose and details of the study. Patients who were categorised into ASA physical status classes II–IV and who were haemodynamically unstable or had pathology in the neck area were excluded.
Patient information (i.e. age, height, weight and type of surgery) was recorded after the patients were taken into the operating room. Datex-Ohmeda Cardiocap™/5 (General Electric Healthcare, Helsinki, Finland) was used to monitor the patients’ heart rate and rhythm (with electrocardiogram lead II), systolic and diastolic blood pressures and mean arterial pressure noninvasively; peripheral oxygen saturation and capnography pressure of end-tidal carbon dioxide (ETCO2) were measured by pulse oximetry. Fluid infusion was started with 5% dextrose and 0.45% sodium chloride solution at 5–10 mL/kg/hour. No premedication was given.
Following pre-oxygenation with 100% oxygen for 2–3 minutes, 1% propofol (10 mg/mL) and fentanyl 2 µg/kg were administered until the patient’s eyelash reflex disappeared. Ventilation was performed using a face mask until a laryngeal mask airway could be inserted. Anaesthesia was maintained with 2% sevoflurane in a mixture of 50/50 oxygen and air. Positive pressure mechanical ventilation was applied with 8–10 mL/kg tidal volume to ETCO2 of 35–40 mmHg.
US image of the RIJV was brought to the middle of the screen of a 12-MHz linear transducer (LOGIQ-e Compact Ultrasound; General Electric Healthcare, Pittsburgh, PA, USA). All diameters were measured at the level of the cricoid cartilage by rotating the patient’s head 20° to the left; all measurements were made by the same physician. In order to exclude respiratory effects, measurements were made at the end of inspiration.
After the RIJV diameter was measured in the supine position, the Valsalva manoeuvre was applied at a pressure of 20 cmH2O for 15 seconds and a further measurement was taken. Measurements were then taken in the Trendelenburg and reverse Trendelenburg positions, with and without the Valsalva manoeuvre. The Trendelenburg position was maintained by placing the operating table at a 15° head-down angle, while the reverse Trendelenburg position was maintained by placing the operating table at a 15° head-up angle of the operation table. The sequence was as follows: supine; supine + Valsalva; Trendelenburg; Trendelenburg + Valsalva; reverse Trendelenburg; and reverse Trendelenburg + Valsalva.
Data collected was analysed by the Biostatistics Department of UUFM using the IBM SPSS Statistics version 20.0 (IBM Corp, Armonk, NY, USA). Continuous variables were presented as descriptive statistics, such as mean ± 2 standard deviations and median (range) values. Categorical variables were presented as frequency and percentages. The Wilcoxon signed-rank test was used for group comparisons. p < 0.05 was considered statistically significant.
RESULTS
None of the 100 patients recruited were excluded from the study. The demographic data of the patients is summarised in
Table I
Patient demographics (n = 100).

Table II
Type of surgery the patients underwent.

The mean RIJV diameter in the supine position was 1.66 cm; this diameter was taken as the baseline. The mean RIJV diameter was 1.76 cm (p < 0.001) in the Trendelenburg position and 1.68 cm in the reverse Trendelenburg position (p = 0.100) (
Table III
RIJV diameters of the patients in different positions, with and without the Valsalva manoeuvre.
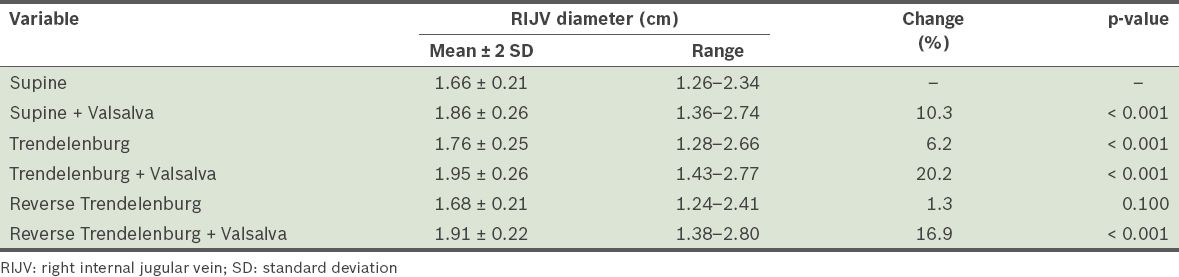
The mean RIJV diameter increased by 10.3% in supine + Valsalva, 6.2% in Trendelenburg, 1.3% in reverse Trendelenburg and 16.9% in reverse Trendelenburg + Valsalva. The greatest change (20.2%) was seen in Trendelenburg + Valsalva. In 15% of the patients, the RIJV was more medial than normal and in front of the carotid artery, while in 5% of the patients, it was more lateral. In the remaining 80% of the patients, the RIJV was anterolateral to the carotid artery.
DISCUSSION
AL and US guidance are techniques often used for CVCs. As AL techniques are ‘blind’ and require experience to perform, they have greater complication and failure rates when done by learners. Performing 30–50 CVCs successfully is considered adequate training for CVCs in adults; however, more experience is required for CVCs in paediatric patients. US reduces complications during CVCs,(15,16) provides good-quality imaging of the vascular anatomy at a low cost and can be applied at the bedside.(7,8,17-20) With US guidance, CVCs can also be performed safely in patients with bleeding disorders.(21,22)
The success rate of CVCs in children is lower and the complication rate is greater.(23,24) English et al(25) studied internal jugular vein catheterisation by the percutaneous method in 415 adult patients and 85 paediatric patients; the success rates of catheterisation in the adult patients and paediatric patients were 96% and 91%, respectively. Hayashi et al(26) reported a success rate of 97.2% for percutaneous cannulations in 106 children; however, this success rate decreased to 81.3% when the patient was aged below three months.
In a study by Leyvi et al,(27) internal jugular vein catheters were inserted in 149 paediatric patients via either the AL or US-guided technique. Although the authors found that the overall success rate for US-guided catheterisation was higher than that for AL-based catheterisation (94.7% vs. 75.9%), no difference was found between the two methods for patients aged below one year and for patients who weighed less than 10 kg.(27) Asheim et al’s study,(28) which involved 42 paediatric patients aged 0–177 months and weighing 3–45 kg, reported a 100% success rate in US-guided CVCs (95% at the first attempt) and no complications. Denys et al(20) also reported a 100% success rate in US-guided CVCs (78% at the first attempt).
Carotid artery injury is a serious complication of internal jugular vein catheterisation.(29) Although arterial entry rates between 4.3% and 25% have been reported,(4,16,19,24,30,31) arterial puncture is less common with US guidance.(19,23,32) Similarly, although posterior wall perforation can occur on occasion, this happens less frequently with US guidance.(33)
The anatomy of the internal jugular vein is variable.(2) It lies anterior and lateral to the carotid artery in 77% of patients,(9) fully lateral to the carotid artery in 2% and fully medial to the carotid artery in 1%.(34) In another study, 4% of the patients had an internal jugular vein that was smaller than normal, but located at the correct anatomical site; the vein was medial to the carotid artery in 10% of the patients and more lateral than normal in 2% of the patients.(35) In the present study, the internal jugular vein was located more medially than usual in 15% of the patients, and lateral to the carotid artery in 5% of the patients. In the remaining 80%, the internal jugular vein was anterolateral to the carotid artery, similar to that reported in other studies.(9,35)
The position of the patient’s head during CVCs affects the internal jugular vein’s anatomical relationships and diameter. When the head is rotated to the left at increasing angles, the internal jugular vein and carotid artery come closer to each other; at angles above 40°, may even overlap.(29,36,37) While keeping the patient’s head in an almost neutral position helps retain normal anatomical relationships and decreases the probability of arterial puncture, the position of the mandible may be a problem in the neutral position.(29,36-38) For this reason, the heads of the paediatric patients were rotated 20° to the left in the present study.
As catheterisation is easier when the cross-sectional area of the internal jugular vein is large, various positions and manoeuvres are applied during cannulation. The use of various surgical positions (e.g. Trendelenburg position and head rotation) and manoeuvres (e.g. hepatic compression, Valsalva manoeuvre and carotid pressure) have been reported.(10-12,39-41) Although some studies have reported the placement of a support under the patient’s shoulder during catheterisation, this method is ineffective in children.(42)
The Trendelenburg position has a positive effect on vein diameter and also prevents air embolisms. Its use helps to improve the success rate of CVCs. The maximum degree of tilt required for this position was reported as 10° in one study(43) and 25° in another.(44) A 25° tilt, however, may not be tolerated by an awake patient. We used the Trendelenburg position with a 15° head tilt in the present study and observed a significant increase in RIJV diameter.(12,33,36)
The Valsalva manoeuvre increases intrathoracic pressure, which then increases the diameter of the internal jugular vein.(11,39,43,45) This manoeuvre, however, is not without risks; it can result in haemodynamic instability and increase the likelihood of complications such as arterial rupture and local haematoma. The ideal pressure in order to achieve increased vein diameter, successful cannulation and a low complication rate, has been suggested to be 20 cmH2O.(33) At a pressure of above 20 cmH2O, hypotension, bradycardia and other complications are more common.(33) Thus, we used 20 cmH2O pressure for 15 minutes in the present study. Our finding of increased RIJV diameter during the Valsalva manoeuvre in the three surgical positions investigated is similar to that of other studies.(11,33,40,44) No complications were encountered.
Verghese et al(12) studied the effect of hepatic compression, Trendelenburg position and the Valsalva manoeuvre, singly and combined, on the diameter of the internal jugular vein of patients aged one month to six years. They found that the Valsalva manoeuvre was the most effective method and that the combination of all methods maximally increased the diameter of the internal jugular vein;(12) this finding is similar to that of the present study. Botero et al(10) concluded that a 20° Trendelenburg head tilt was not as effective as 20 cmH2O positive airway pressure, a finding that is also essentially similar to that of the present study.
In conclusion, the results of the present study demonstrate that both the Trendelenburg position and the Valsalva manoeuvre are able to increase RIJV diameter. While the reverse Trendelenburg position alone was not found to have any significant effect on RIJV diameter, it significantly increased RIJV diameter when used in combination with the Valsalva manoeuvre. The Trendelenburg position in combination with the Valsalva manoeuvre resulted in the most significant increase in RIJV diameter.


