Abstract
INTRODUCTION
This study describes the physician experience relating to the effectiveness of incobotulinumtoxinA and patient satisfaction with its use for the treatment of glabellar frown lines (GFLs).
METHODS
A total of 17 patients from six dermatological clinics, aged > 18 years and with mild to very severe GFLs at maximum frown, were included. Patients were excluded if they had treatment with resorbable fillers and botulinum toxins in the preceding six months, or non-resorbable fillers or surgery in the treatment area. Injection sites (range 3–5) were chosen depending on their severity (dose range 12–20 U), covering corrugators and procerus muscles. Physicians assessed improvements to GFLs using the Merz scale on Days 4 and 14 after treatment. Patients completed a self-reported questionnaire on their facial wrinkles on Days 2 and 4 after treatment.
RESULTS
Most (76.5%) patients were women. The mean age of the patients was 46.9 ± 10.0 years. Mean severities (on the Merz scale) for at-rest and dynamic (with expression) GFLs at baseline were 1.3 ± 1.10 and 3.4 ± 0.38, respectively, and decreased on Day 14 (p < 0.05). Treatment response rates (> 1-point improvement) for at-rest and dynamic (with expression) GFLs on Day 4 were 40% and 100%, respectively. All patients reported being satisfied or very satisfied, and 64.3%–71.4% indicated that their facial wrinkles had improved on Day 2.
CONCLUSION
IncobotulinumtoxinA was fast acting with visible improvements by Day 4 and all patients expressed satisfaction with their treatment after two days. GFLs saw the most improvement among the facial characteristics measured.
INTRODUCTION
Botulinum neurotoxins (BoNTs) are bacterial exotoxins that are known to reduce muscular contraction by inhibiting vesicular neurotransmitter release through their interaction with the exocytotic release mechanism.(1) This mechanism of botulinum Type A (BoNT/A) has been harnessed to relax facial muscles via local injections, leading to its commercialisation for use in aesthetic dermatology to treat facial wrinkles in patients.(2) Variants of BoNT/A that have been produced and marketed commercially include onabotulinumtoxinA (Allergan Inc, Irvine, CA, USA), abobotulinumtoxinA (Ipsen Ltd, Slough, Berkshire, UK) and incobotulinumtoxinA (Merz Pharmaceuticals GmbH, Frankfurt am Main, Hesse, Germany).(1)
IncobotulinumtoxinA (marketed under the trademark Xeomin®) has been shown to be effective for the treatment of glabellar frown lines (GFLs).(3-6) It contains only the active neurotoxin and none of the complexing proteins commonly found in other BoNT/A products. As these complexing proteins are not required for the neurotoxin activity, incobotulinumtoxinA has a higher specific biologic activity in each dose.(7) Thus, the onset of treatment effect is more rapid for incobotulinumtoxinA as compared to other variants of BoNT/A.(6) A multicentre European study found that overall treatment satisfaction in using incobotulinumtoxinA specifically for GFLs was high among physicians and patients.(3) The drug was also well tolerated and had a longer duration of effect, permitting a gap of five months or more between injections.(3,6,8) However, reported data from clinical trials has typically been obtained from a week post injection(9) to as long as 30 days after treatment,(4) as the focus was often on efficacy rather than the onset of effect. As a result, the efficacy of incobotulinumtoxinA for the treatment of GFLs has been widely published, but its rapid onset of effect is less well documented. There are, consequently, limited reports about the onset of treatment effect during the first week following the injection.(10)
The Asian Doctors Hands on Experience through Real-life Efficacy (ADHERE) Program is a regional sampling programme designed to provide doctors with firsthand experience with using incobotulinumtoxinA (Xeomin®) for the treatment of GFLs. Through the ADHERE Program, dermatologists were able to assess the effectiveness of incobotulinumtoxinA over a treatment period of at least two weeks and test their patients’ level of satisfaction with the treatment. This article describes the physician experience of the effectiveness of incobotulinumtoxinA and patient satisfaction with its use for the treatment of GFLs.
METHODS
This was a single-arm, prospective, observational, clinical experience study of patients with GFLs. Data was collected through a patient survey conducted by aesthetic specialists (physicians) who were on the ADHERE sampling programme for early use of the drug. Under this programme, 30 patients would be enrolled within the first six months after incobotulinumtoxinA (Xeomin®) was available on the market. Patient recruitment was conducted from February to July 2015 at six aesthetic clinics in Singapore. All survey respondents provided informed consent.
Inclusion criteria were: patients who were aged > 18 years, and had GFLs that were rated by the physician as mild, moderate, severe or very severe at maximum frown. Patients were excluded if they had: (a) treatment with resorbable fillers and botulinum toxins in the preceding six months, or non-resorbable fillers or surgery in the treatment area; (b) allergies to the study medication; (c) contraindications to botulinum toxin treatment (including pregnancy and breastfeeding); (d) severe concomitant disease; and (e) circumstances that would not allow regular participation in the study.
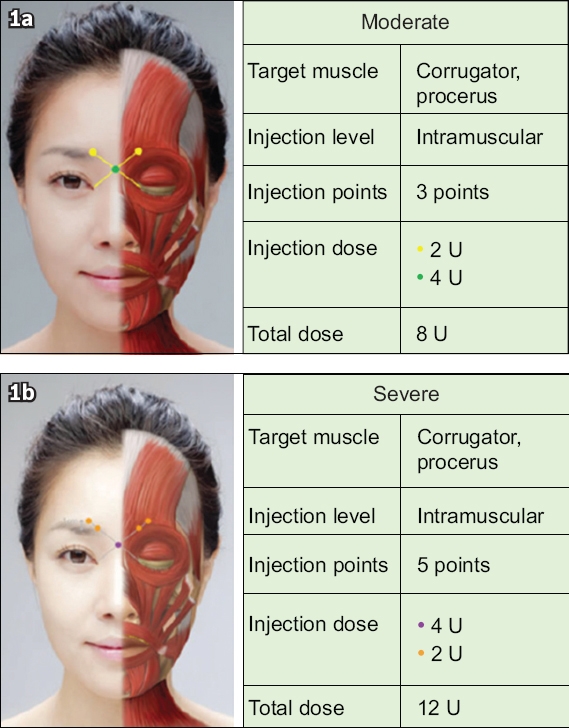
The sponsor of the study supplied the drug used and eligible patients were not required to pay for the treatment. The drug dosage was based on the Asian consensus recommendations.(11) However, the decision on the final dosage (dose range 12–20 U) was made by the attending physician and it depended on the severity of the GFLs. Specifically, 3–5 injection sites were chosen, covering corrugators and procerus muscles, as shown in
Fig. 1
Diagram shows injection sites chosen for patients with (a) moderate and (b) severe glabellar frown lines, based on Ahn et al’s study.(11) In our study, the dose range is 12–20 U (i.e. moderate to severe). Picture reprinted with permission from Wolters Kluwer Health Inc.
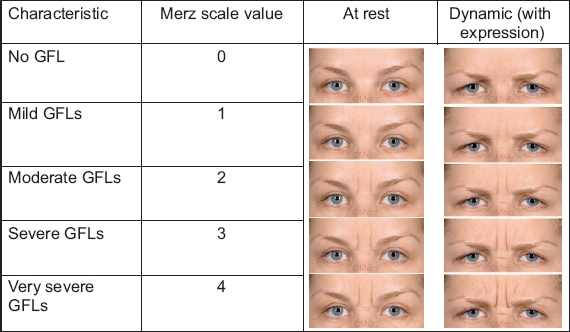
The attending physician assessed the improvement to the GFLs (at rest and dynamic [with expression]) using the Merz scale(12,13) to rate the severity of the wrinkle lines on the features of the patients (
Fig. 2
Photograph shows rating of at-rest and dynamic (with expression) glabellar frown lines (GFLs) using the 5-point Merz scale, with a score range of 0–4.(12)
Descriptive statistics, such as mean ± standard deviation, percentages and/or median (range), were used to assess patient demographics, Merz scores and patient questionnaire responses. The treatment was deemed to be effective from the physician’s perspective if a minimum of a 1-point change from baseline was observed for the values at Days 4 and 14 after treatment. Similarly, a 1-point change in the patient response observed from baseline to Days 2 and 4 after treatment was rated as an improvement from the patient’s perspective. A 1-point change in response would be from ‘a lot’ to ‘a little’, ‘a little’ to ‘not at all’ or ‘a lot’ to ‘not at all’. The McNemar test was used to determine if there was an improvement in the mean Merz score and patient response, i.e. ≥ 1-point improvement was seen between Days 4 and 14 after treatment. The paired t-test was used to determine if there was a change in the Merz score at Days 4 and 14 after treatment compared to baseline. All tests of significance were assessed at the 5% level and statistical analysis was performed using IBM SPSS Statistics version 23 (IBM Corp, Armonk, NY, USA).
RESULTS
A total of 17 patients were recruited from six aesthetic clinics in Singapore. The demographic details of these patients are shown in
Table I
Demographics of patients receiving Xeomin® for treatment of glabellar frown lines (n = 17).
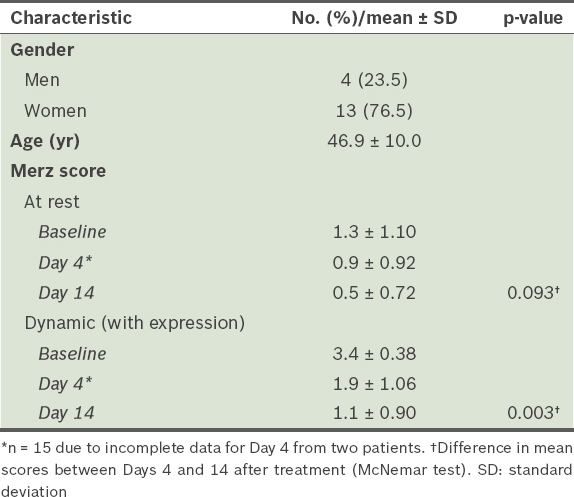
Fig. 3
Photographs show injection sites before and after (on Days 4 and 14) treatment, with the glabellar frown lines rated using the 5-point Merz scale (score range 0–4) illustrating assessment of the treatment outcome.
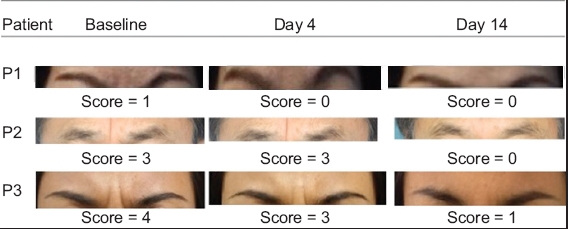
Table II
Mean change in the severity of glabellar frown lines (GFLs), as measured on the Merz scale.
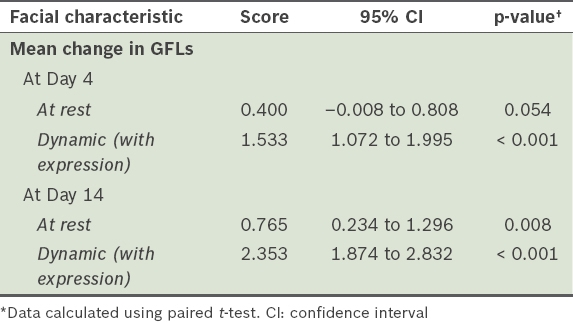
Table III
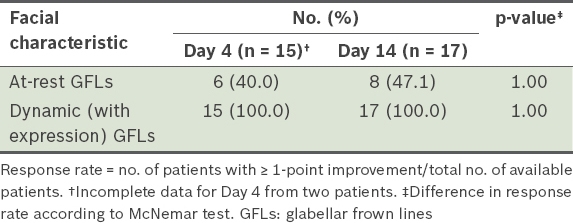
Response rate provided by physicians for patients with ≥ 1-point improvement over baseline on the Merz scale.
Among the patients in the study cohort, 14 completed the self-reported questionnaire (
Table IV
Patient satisfaction rate according to the self-reported questionnaire (n = 14).
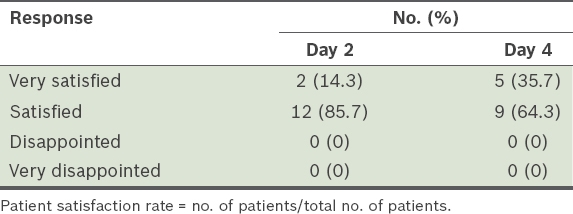
Table V
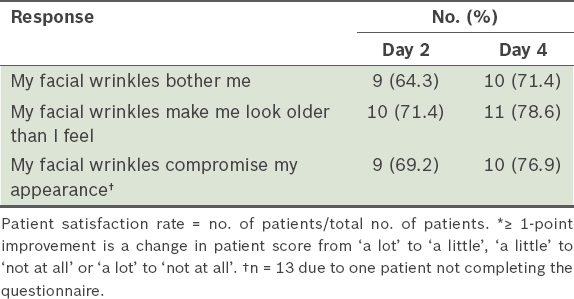
Patient satisfaction among patients with ≥ 1-point improvement* (n = 14).
DISCUSSION
Our study showed that the incobotulinumtoxinA (Xeomin®) drug was fast acting, with observable improvements by Day 4 after treatment. In a similar single-arm, prospective, proof-of-concept study of 23 patients, a 1-point improvement (also measured on the Merz scale) in GFLs at maximum frown was observed in 95.2% of patients within four days.(8) It was also estimated that 84% of the maximum effect would have been achieved by this time. Similar to the results of other studies.(3,8,14) effective treatment of dynamic (with expression) GFLs was achieved in all our treated patients by Day 4. Our physicians also noted significant changes in mean scores from Days 4 to 14 after treatment.
While additional improvements to facial characteristics were recorded from Day 4 to Day 14, there was no corresponding increase in the treatment response rate. The response rates for GFLs at rest increased only marginally from 40.0% on Day 4 after treatment to 47.1% at Day 14, although the difference in Merz scores from baseline to Day 14 was significant. In contrast, the treatment for dynamic (with expression) GFLs achieved a 100% response rate on Day 4 after treatment, and the drug continued to have effect on GFLs at rest beyond Day 4. Phase III trials of the neurotoxin using higher doses (up to 20 U of the drug) to treat GFLs at rest have also reported continued effect and that a higher percentage (range 77%–94%) of patients responded to the treatment by Day 30.(14,15) Similarly, pooled data from investigator- and subject-assessed trials for GFLs showed that the maximum response rate of 93.1% was achieved at about 30 days after treatment.(16) Thus, the shorter duration of 14 days in our study may not be sufficiently long for the drug to demonstrate its maximum effect.
The perception of satisfaction can be personal and subjective depending on the respondent. In such a study, patient satisfaction includes an assessment of the treatment, procedure and outcome, which each have a varying degree of importance for patients. The number of outcome measures and scales used in such studies is large and varied. A review of patient-reported studies showed high rates (range 65%–100%) of patient satisfaction for treatment of GFLs using BoNT/A regardless of the measurement method.(2) In our study, all patients expressed satisfaction with their treatment after two days and a majority reported improvement to their facial wrinkles.
To conclude, we are aware of some limitations of our study. For one, the small number of patients in our study means that the study population may not be representative of the general Singapore population. Furthermore, the short duration of the study may not have allowed the neurotoxin to demonstrate its maximum effect and, thus, limited observable improvements. There may also be some bias in the reported observations, as the same attending physician who performed the treatment also assessed its outcome. As this is an early-use programme, its aim was to provide physicians with first-hand experience on the use of incobotulinumtoxinA to treat GFLs. The study showed the rapid onset of effect of incobotulinumtoxinA, and the positive treatment results achieved by our patients were consistent with and similar to those of studies carried out elsewhere.
ACKNOWLEDGEMENTS
This study is supported and funded by Merz Asia-Pacific, which also provided the study drug. We would like to acknowledge the following physicians who enrolled their patients into our study: Dr Tam Elias, Dr Soon-Tee Wong, Dr Khek-Kah Chew, Dr Bee-Lan Lam. Data management, statistical and editorial support was provided by Sen-Kwan Tay of Research2Trials Clinical Solutions Pte Ltd and funded by Merz Asia-Pacific.
Supplementary Material
The full versions of the patient assessment questionnaire and patient satisfaction survey used in this study are available online at http://www.smj.org.sg/sites/default/files/SMJ-58-606app.pdf


