Abstract
INTRODUCTION
Septic arthritis (SA) is a devastating infection with a high rate of sequelae. The aim of this retrospective study was to determine the epidemiology, clinically significant sequelae and risk factors for developing these sequelae in children admitted to our hospital with SA.
METHODS
Patients with bacteriologically and/or radiologically confirmed SA from January 1999 to December 2014 were identified from discharge and laboratory records. Data was collected through a retrospective review of the case notes.
RESULTS
A total of 75 patients (62.7% male) met the inclusion criteria. The median age at presentation was six years (range two weeks to 15 years), and six patients were neonates. Microbiologic aetiology was determined in 40 (53.3%) patients, with Staphylococcus aureus being the most common organism. 68.0% of the patients underwent arthrotomy, and the average hospital stay was 15.3 days. Sequelae of SA were observed in nine patients on follow-up. Univariate and multivariate statistical analyses showed that young age, pyogenic bacterial isolation and concomitant osteomyelitis were significant risk factors for developing sequelae.
CONCLUSION
Our study demonstrated that young age, pyogenic bacterial isolation and concomitant osteomyelitis are associated with a high risk of sequelae. Timely microbiologic diagnosis by novel polymerase chain reaction methods and the use of magnetic resonance imaging in high-risk children to identify adjacent infection could possibly prevent lifelong disabling sequelae in SA.
INTRODUCTION
Septic arthritis (SA) is a potentially devastating condition with a high incidence of severe and long-lasting sequelae, particularly in growing children. In the skeletally immature child, SA can lead to physeal injury and growth arrest, resulting in leg length discrepancy or angular deformity.(1) Prompt recognition and treatment of SA in children is crucial to prevent the risk of such long-term sequelae. Few international studies have analysed the risk factors for developing long-term sequelae in children with SA.(2-4) A systematic review in 2009 reported that young age and delayed treatment are poor prognostic factors in children with SA.(5)
The aim of this retrospective study was to determine the epidemiology, clinically significant sequelae and risk factors for developing these sequelae in children admitted to our hospital with SA. In an effort to standardise care and improve outcomes, our study specifically analysed the risk factors for developing long-term sequelae of SA that are prevalent among the local population. We also reviewed the recent literature for current developments in the management of paediatric SA.
METHODS
This was a retrospective study conducted at KK Women’s and Children’s Hospital (KKH) from January 1999 to December 2014. KKH is a tertiary paediatric centre with 857 inpatient beds that serves most of the child population in Singapore. All children aged younger than 16 years who had been diagnosed with SA were eligible. All patients discharged with a final diagnosis of SA were identified through a search of the hospital databases coded according to the International Classification of Diseases, 10th revision, Clinical Modification (ICD-10-CM). The following ICD-10-CM codes were included: M00.0 (Staphylococcal arthritis and polyarthritis); M00.1 (pneumococcal arthritis and polyarthritis); M00.2 (other streptococcal arthritis and polyarthritis); M00.8 (arthritis and polyarthritis due to other specified bacterial agents); and M00.9 (pyogenic arthritis, unspecified). The diagnosis of SA was established by a multidisciplinary primary care team and was based on characteristic clinical, and bacteriological and radiological findings commonly seen in SA.
We included all patients who fulfilled the following criteria: (a) clinical presentation consistent with a joint infection (fever, joint swelling, joint pain and abnormal physical examination); and/or (b) positive blood or synovial fluid/tissue culture; and/or (c) presence of radiological signs consistent with SA (such as synovial effusion); and/or (d) surgical finding of pus in joint. Children with immunosuppression and penetrating joint injury were excluded from the study. The medical records of the patients were reviewed and the following data was collected: demographics; clinical and laboratory parameters at presentation; imaging studies; surgical procedures; antibiotic treatment; duration of hospitalisation; complication; total duration of therapy; and sequelae. A C-reactive protein (CRP) level ≥ 20 mg/L, erythrocyte sedimentation rate (ESR) ≥ 10 mm/hr and white blood cell (WBC) count ≥ 15 × 109/L were considered elevated. Bacterial cultures of blood were processed using the BD BACTEC 9240 blood culture system (Becton, Dickinson and Company, Franklin Lakes, NJ, USA), and synovial fluid and bone tissue were inoculated on agar or broth media. Organisms were identified using standard biochemical methods or the Vitek® 2 system (bioMérieux Inc, Marcy I’Etoile, France). Antibiotic susceptibility testing was performed using the Calibrated Dichotomous Susceptibility Test. Patients were classified as having culture-positive SA if growth of pathogenic bacteria from blood or joint aspirate was observed. Concomitant osteomyelitis was diagnosed with radiographic evidence of bone infection by ultrasonography and/or magnetic resonance (MR) imaging. The presence of sequelae was determined by orthopaedic surgeons in the outpatient clinic during follow-up. The study protocol was approved by the KKH Institutional Review Board. The above thresholds were chosen so as to take into account milder forms of SA such as Kingella kingae infection.
Patient demographics, clinical and laboratory variables, and complications were summarised as median (interquartile range) for continuous variables and counts (%) for categorical variables. Differences between patients with bacteriology-positive cultures and those with negative cultures were compared using Fisher’s exact test for categorical variables and Wilcoxon’s two-sample test for continuous variables. Risk factors associated with sequelae were analysed using univariate and multivariable logistic regression analyses. Statistical analysis was performed using SAS version 9.4 (SAS Inc, Cary, NC, USA). All statistical tests of significance were two-tailed and p < 0.05 was considered statistically significant.
RESULTS
A total of 75 patients (62.7% male) met the inclusion criteria and were included in the analysis. The median age at presentation was six years (range 2 weeks to 15 years), and six patients were neonates. Fever and localised pain were the main presenting symptoms (84.0% and 85.3%, respectively).
Table I
Demographics and clinical/laboratory data (n = 75).
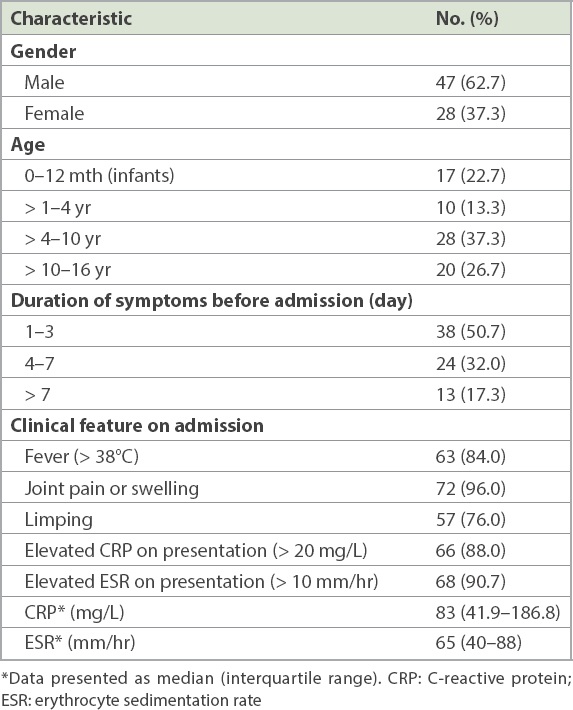
The joints of the lower extremities (hips, knees and ankles) were affected in most of the patients (79.9%), with the hip being the most commonly affected joint (46.6%;
Fig. 1
Graph shows the distribution of joint involvement in the cohort.
At the time of presentation, WBC count and CRP test were performed in all the patients. ESR (median 65 mm/hr; range 5–165 mm/hr), CRP (median 83 mg/L; range 5–390 mg/L) and WBC (median 15.5 × 109/L; range 4.25–32.4 × 109/L) were elevated in 90.7%, 88.0% and 69.3% of patients, respectively. There was no significant correlation between laboratory parameter values and duration of symptoms before hospitalisation.
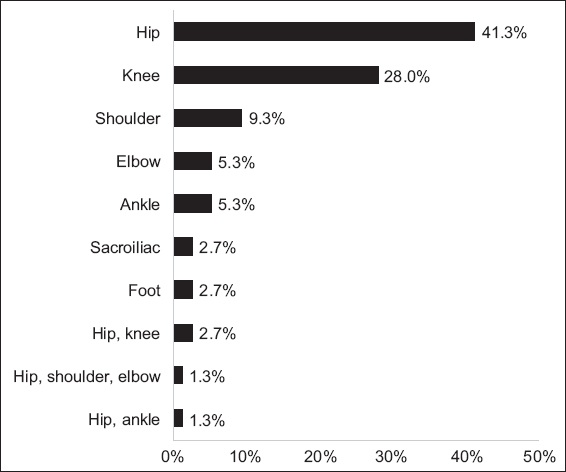
Blood cultures and synovial fluid cultures were performed in 90.7% and 80.0% of patients, respectively. Microbiologic aetiology was determined in 40 (53.3%) patients. Blood cultures and synovial fluid cultures were positive in 33.4% and 38.7% of the patients, respectively. Staphylococcus aureus (S. aureus) was the most common organism (33.3%), followed by Streptococcus pneumoniae (4.0%) (
Table II
Distribution of pathogens (n = 75).
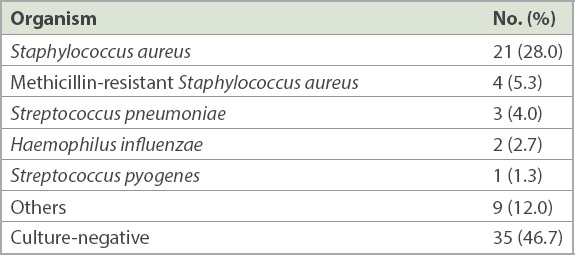
Table III
Differences between bacteriology-positive and bacteriology-negative patients.
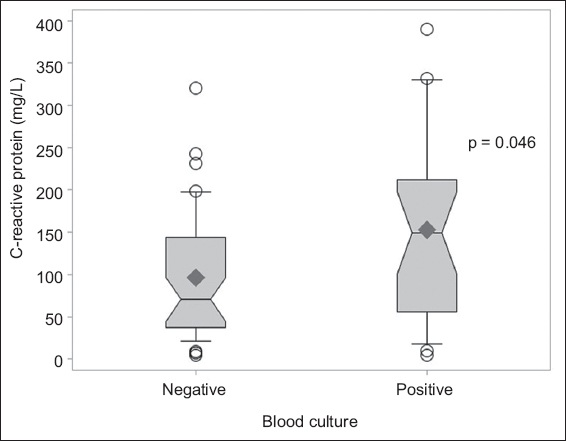
Patients with bacteraemia were more likely to have a higher median CRP level on admission (149.0 mg/L vs. 70.6 mg/L, p = 0.046;
Fig. 2
Graph shows the distribution of C-reactive protein in blood culture-positive and blood culture-negative septic arthritis.
Among the 75 children, 10 (13.3%) had concomitant osteomyelitis, and it was more common in boys (90.0% vs. 58.5%, p = 0.080) and infants (40.0% vs. 20.0%, p = 0.221). Children with concomitant osteomyelitis were more likely to have positive bacteriology (80.0% vs. 49.2%, p = 0.094) and sequelae (50.0% vs. 6.2%, p = 0.001). They also received antibiotics for a longer duration (42.7 days vs. 37.4 days, p = 0.013) and had longer hospital stay (22.7 day vs. 14.2 days, p = 0.021) than their counterparts.
Radiological abnormalities were noted in 84.0% of the patients. Initial radiography was the most frequently used technique (89.0%), and the diagnostic yield was low at 31.3%. Ultrasonography, the second most frequently used technique (77.3%), had the highest diagnostic yield (89.7%). MR imaging and bone scintigraphy were infrequently used.
Initially, all patients were treated with parenteral antibiotic therapy (range 4–42 days), followed by oral administration to be continued after discharge (range 0–43 days). The most frequently used intravenous (IV) antibiotic was cloxacillin, followed by the cloxacillin-ceftriaxone combination. The most used oral antibiotic was cloxacillin, followed by cephalexin. The mean total duration of antibiotic use was 38 (range 21–56) days. 68.0% of the patients underwent arthrotomy, 10.7% underwent needle aspiration and 5.3% underwent arthroscopic drainage. The mean duration of hospital stay was 15.3 (range 4–83) days.
About 92% of the patients were seen in the follow-up clinic, and the mean duration of follow-up was 16.7 months. Sequelae of SA were observed in 9 (12.0%) patients during follow-up, and the most frequently seen sequelae were destructive changes with limited range of motion as well as deformity (
Table IV
Long-term sequelae in the cohort (n = 75).

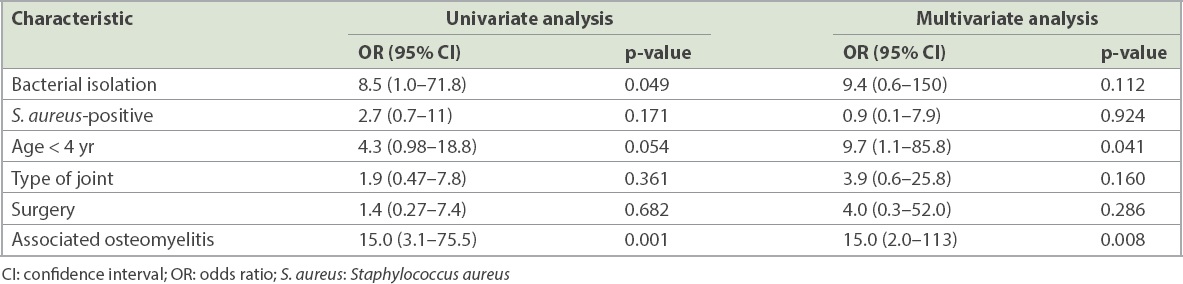
Univariate analysis showed that bacterial isolation and concomitant osteomyelitis were risk factors for the development of sequelae. In multivariate analysis, concomitant osteomyelitis remained a significant risk factor for developing sequelae, while age less than four years was a strong risk factor for sequelae. S. aureus infection, surgery and type of joints were not associated with the development of sequelae (
Table V
Factors associated with sequelae in septic arthritis.
DISCUSSION
To our knowledge, this is the first study from Singapore that described the burden of long-term sequelae associated with paediatric SA and analysed the risk factors for developing these sequelae. Our study examined the prevalence, spectrum of clinical presentation, epidemiological characteristics and long-term outcomes of children with SA, with special emphasis on the risk factors for developing sequelae.
The age and gender distribution in our cohort were comparable with those of previous studies.(4,6-9) Similar to other studies, we found that lower limb joints were more commonly affected, with joint pain or swelling being the most frequently reported symptom.(1,4-9) In our cohort, S. aureus continued to be the most common pathogen in children with SA,(1,2,4-9) and the rate of culture-positive cases (53.3%) was consistent with that reported in previous studies.(2,4-8) Increasing prevalence of MRSA was noted in our neonatal cohort. Our study revealed that 12.0% of children with SA developed long-term sequelae, and this is consistent with the rates in previous reports from the developed world.(2,4)
A retrospective study from Australia that evaluated the outcome of SA in 44 children reported that 10% of their cohort developed clinically significant sequelae at 12 months following diagnosis.(2) A Taiwanese retrospective study that analysed 58 children with SA found that 17% of their cohort eventually developed sequelae of SA.(4) Most notably, studies from developing countries have reported significantly higher rates of sequelae in up to 66% of their cohorts.(10,11)
Our study demonstrated that young age, pyogenic bacterial isolation and concomitant osteomyelitis were associated with a high risk of sequelae. We opine that prompt recognition and aggressive management of children at high risk of developing sequelae could possibly improve the long-term outcome. Young age has been well established as a risk factor for poor prognosis in SA.(2,4,5,12) SA in infancy often presents with atypical or minimal symptoms, leading to delayed diagnosis. Elevation of inflammatory markers may not be present, which may further compound the situation. Moreover, infants have a higher rate of coexisting osteomyelitis and SA, because the spread of infection from the metaphysis to the joint space is enhanced by the unique transphyseal vasculature in this age group.(12) In addition, young infants with hip SA have a high risk of developing avascular necrosis.(13) As SA is more challenging to diagnose in young children, a high level of suspicion is required to make an early diagnosis. We propose that prompt evaluation of infants with suspected SA by orthopaedic surgeons could improve the outcome through timely diagnosis and early surgical intervention.
Previous studies have shown that children with culture-positive SA have a poorer prognosis compared with those with culture-negative SA.(14) Howard-Jones et al studied the rate of long-term residual function in children with SA and concluded that culture positivity is a poor prognostic factor, with increased rate of clinically significant dysfunction (2) Our present series likewise showed significantly higher sequelae in culture-positive cases (20.0% vs. 2.9%, p = 0.032). Bacterial isolation by synovial fluid or tissue culture is considered the gold standard for diagnosing pyogenic SA. However, the long turnaround time could potentially delay the administration of appropriate antibiotics, particularly in MRSA infections. Although detection of Kingella kingae by polymerase chain reaction (PCR) is well established,(15,16) the clinical utility of PCR for diagnosing pyogenic SA is still in development. Rapid identification of pyogenic infection from synovial fluid using novel PCR-based methods offers great benefit in selecting appropriate antibiotics for SA in children. In a prospective study, Choe et al compared the diagnostic performance of real-time PCR assay to microbiological culture methods in 20 children with SA, using methicillin-resistant Staphylococcus-specific PCR and broad-range universal PCR for rapid differentiation between Gram-positive and Gram-negative organisms; the study concluded that real-time PCR has higher sensitivity than microbiological culture for the diagnosis of children with SA.(17) Montgomery and Rosenfeld recommended that the use of real-time PCR could help to guide antibiotic therapy while the results from traditional microbiological culture are still pending.(18) We propose that the use of real-time PCR as a supplemental tool for rapid identification of pyogenic infection and reliable selection of appropriate antibiotics could potentially improve the outcome of children with SA. In countries with a high rate of MRSA, rapid identification of MRSA by real-time PCR could restrict the widespread usage of vancomycin, thus contributing to major reductions in antibiotic resistance.
Synovasure, a novel diagnostic test to detect prosthetic joint infection in adults, is gaining popularity owing to its ease of use and rapid turnover time.(19) This test determines the presence of alpha defensin, a novel biomarker in the synovial fluid, and has been found to have excellent sensitivity and specificity. Although it had never been tested in the paediatric population, it could potentially be developed as a point-of-care test in the future for rapid diagnosis of SA in children.
The incidence of concurrent osteomyelitis in our cohort was 13.3%, which was significantly lower than previously reported.(1,20,21) Our data demonstrated that concomitant osteomyelitis was significantly associated with bacteraemia, longer duration of hospital stay and increased risk of sequelae. Previous studies have reported that children with concomitant SA and osteomyelitis have a less favourable clinical course. Therefore, awareness of the presence of concomitant osteomyelitis is vital to guide further clinical management.(1,21) Branson et al described children with concomitant SA and osteomyelitis as more likely to have a longer duration of symptoms on presentation, positive blood culture, longer duration of hospital stay and increased risk of sequelae.(21) Rosenfeld et al developed a prediction algorithm to distinguish SA with adjacent infections from isolated SA, identifying five independent predictors of adjacent infection: age > 4 years, CRP > 13.8 mg/L, duration of symptoms > 3 days, platelets < 314 × 103 cells/mL and absolute neutrophil count > 8.6 × 103 cells/μL. The study indicated that patients with SA who meet three or more criteria are at a high risk of having associated adjacent infections and should undergo preoperative MR imaging to improve their outcome.(22) Of note, Rosenfeld et al’s study included only culture-positive SA and excluded children below one year of age.
Montgomery et al reported that advanced imaging (computed tomography, bone scintigraphy and/or MR imaging) of high-risk children could potentially limit infection-related sequelae by identifying concurrent infection earlier. Risk factors in the study included age less than four months or 13–20 years, shoulder involvement and presence of symptoms for more than six days. The study also found that pyogenic SA has a significantly greater frequency of concurrent infection.(20) Browne et al reported that the sensitivity of MR imaging for detecting skeletal infection caused by community-acquired S. aureus in infants and young children is significantly improved by the addition of gadolinium-enhanced sequences. The study recommended that contrast-enhanced sequences be performed in all infants with suspected skeletal infection in locales where community-acquired S. aureus is endemic.(23) We propose that children with risk factors such as young age and bacteraemia be carefully evaluated for the presence of adjacent infection; if found to be at risk, they could benefit from advanced imaging techniques such as MR imaging to ensure good prognosis.
The hip joint was the most frequently affected joint in our series. Of the 35 patients with hip SA, 13 (37.1%) met all four predictors of Kocher’s criteria (fever, non-weight-bearing status, ESR ≥ 40 mm/hr and WBC > 12 × 109/L). We noted that patients who satisfied Kocher’s criteria(24) more frequently underwent surgical intervention as compared to their counterparts (84.6% vs. 68.0%, p = 0.282). However, there was no significant difference in culture positivity between these two groups (46.2% vs. 54.5%, p = 0.631). This serves as a reminder that although clinical prediction rules provide physicians with some much-needed objectivity during the decision-making process, physicians must still exercise their best clinical judgement in order to make a definitive diagnosis and create an appropriate management plan.
Traditionally, children with SA were treated with several weeks of IV antibiotics. In our series, 87% of the cohort received IV antibiotics for at least one week. Recent evidence supports a shorter duration of IV antibiotics with a prompt switch to oral therapy in uncomplicated cases of SA.(25) Jagodzinski et al performed a prospective bi-centre study and found that a shortened course of IV and oral antibiotic therapy was effective in the management of acute uncomplicated bone and joint infection in children. The study included 70 children in whom the duration of IV antibiotics was determined by careful assessment of clinical response and by stabilising CRP. Overall, 86% of patients were converted to oral therapy by Day 5. Oral therapy was continued for only three weeks in the majority (83%) of patients, and the remaining received prolonged antibiotic therapy in view of abnormal radiographic findings. At the one-year follow-up, all the patients were clinically, haematologically and radiologically normal.(26)
Peltola et al performed a randomised, multicentre prospective trial in 130 children with culture-positive SA and concluded that treatment with large doses of well-absorbed antimicrobials for about ten days (IV for the first 2–4 days) is not less effective than a 30-day treatment course for childhood SA, provided that the clinical response is good and the CRP level normalises quickly. Two late-onset infections occurred in one child, and 12% of the patients did not have a one-year follow-up visit.(27) Of note, both studies(26,27) excluded complicated cases such as combined SA with osteomyelitis. Children with SA who are at high risk of developing sequelae need a cautious, conservative approach in terms of the duration of antibiotics treatment. This duration should be extended if there is no adequate clinical response or if there is imaging evidence of accompanying osteomyelitis.
Timely diagnosis, coupled with prompt and aggressive antimicrobial therapy and surgical irrigation, is necessary to ensure a good prognosis for SA.(28) Open arthrotomy is considered the gold-standard treatment for SA of tightly encapsulated joints, such as the hip and shoulder, for rapid reduction of intra-articular pressure to prevent avascular necrosis.(25) Recently, less invasive procedures such as arthroscopy and serial ultrasonography-guided aspirations have shown acceptable results. El-Sayed performed a prospective randomised controlled trial of 24 children with hip SA and reported that minimally invasive arthroscopic drainage is a valid alternative to open arthrotomy.(29) Of note, the study excluded children under three years of age with concomitant osteomyelitis. Another prospective study by Thompson and Gourineni, which included 24 young children with various joint involvements, concluded that arthroscopic lavage appears to be a safe, quick and effective alternative to open arthrotomy in very young children.(30) Givon et al performed a retrospective study on children with hip SA who were treated with serial ultrasonography-guided aspirations without the need for general anaesthesia. Out of their 28 patients, four eventually underwent arthrotomy, with no long-term complications observed on follow-up. The study concluded that repeated aspiration is a safe and efficacious treatment method for hip SA.(31) Given the small sample size and selective inclusion of children with uncomplicated SA, further prospective studies comparing the outcomes of open arthrotomy with those of arthroscopic lavage are needed to confirm the efficacy of these less invasive procedures.
SA continues to cause significant morbidity despite prompt antibiotic therapy and surgical intervention. There is growing evidence that the use of adjuvant therapy such as corticosteroids to downregulate the heightened immune response may improve outcome. A retrospective study by Fogel et al demonstrated that children with SA who were initially treated with a short course of adjuvant dexamethasone showed earlier improvement in clinical and laboratory parameters compared to children who were treated with antibiotics alone.(32) Of note, the majority of cases in this cohort were culture negative and the follow-up period was only six months. A double-blind, randomised placebo-controlled trial in 123 children by Odio et al compared the use of low-dose IV dexamethasone therapy, given in conjunction with antibiotic therapy, with antibiotic therapy alone. The study, which evaluated only culture-positive patients and had a follow-up period of 12 months, concluded that a short course of low-dose dexamethasone given in conjunction with antibiotic therapy reduces residual joint dysfunction and significantly shortens the duration of symptoms in children with SA.(33) More research, however, is needed to recommend the routine use of steroids as adjuvant therapy in SA.
The limitations of our study included the inherent bias associated with a retrospective study design. As a single-centre study, our results may not be applicable to all settings. In our centre, PCR for Kingella kingae was not routinely performed and thus, a portion of the culture-negative cases could have been attributable to Kingella kingae infection. The lower prevalence of concomitant osteomyelitis in our cohort could be attributed to the marginal usage of MR imaging. Despite these limitations, our study highlighted the importance of prompt recognition of children at high risk of developing sequelae to ensure good prognosis. We also provided recommendations on the utilisation of modern diagnostic tools such as novel PCR methods and MR imaging to improve outcomes in high-risk children. In addition, our study reviewed recent literature on current developments in the management of paediatric SA.
In conclusion, the present study demonstrated that young age, pyogenic bacterial isolation and concomitant osteomyelitis were associated with a high risk of sequelae in SA. Timely microbiologic diagnosis by novel PCR methods and the use of MR imaging in high-risk children to identify adjacent infection could possibly prevent lifelong disabling sequelae in SA. Furthermore, a cautious and conservative approach to the duration of antibiotic use is required in high-risk children. Currently, there is insufficient evidence for the routine use of dexamethasone as adjuvant therapy. Further prospective studies addressing the risk factors for developing sequelae in SA are needed to improve long-term outcomes.
ACKNOWLEDGEMENT
We would like to thank Adj A/Prof Thoon Koh Cheng, Senior Consultant and Head, Paediatric Infectious Disease service, KK Women’s and Children’s Hospital, Singapore, for his assistance and support for this project.


