Abstract
Combined oral contraceptive pills (COCs) remain one of the most popular forms of contraception to prevent unwanted pregnancy in women. While it is known that COCs can cause sexual dysfunction in women, there is currently no recommendation to screen for sexual function before and after initiation of COCs. We propose that, based on the evidence available, assessment of sexual function should be done at initiation of COCs, as well as at regular intervals thereafter. This would allow COC-related sexual dysfunction to be managed early, such as by switching the patient to newer-generation COCs or other forms of contraception.
Mr and Mrs Lim had consulted you three months ago for contraception, as they had completed their family with two children aged four and six years old, and wanted to start birth control to avoid another pregnancy. Mrs Lim was prescribed combined oral contraceptive pills consisting of levonorgestrel 150 mcg and ethinylestradiol 30 mcg. She returned for follow-up with complaints of decreased libido and occasional discomfort during intercourse for the past one month. Her menses were regular and of normal flow. This had affected the couple’s relationship and they wanted to know if anything could be done to address her symptoms.
WHAT IS FEMALE SEXUAL DYSFUNCTION?
Sexual dysfunction is defined as a persistent or recurrent sexual problem that causes marked distress or interpersonal difficulty. The 2013 Diagnostic and Statistical Manual of Mental Disorders(1) classifies female sexual dysfunction (FSD) into three domains: female sexual interest/arousal disorder; female orgasmic disorder; and genito-pelvic pain/penetration disorder. Female sexual interest/arousal disorder occurs when a woman experiences persistent or recurrent inability to attain sexual arousal, or to maintain arousal until the completion of a sexual activity.(1) This disorder can also refer to an inadequate lubrication-swelling response that is normally present during arousal and sexual activity. Female orgasmic disorder occurs when a woman either cannot reach orgasm or has trouble reaching orgasm even after ample sexual stimulation. This disorder can be primary, or secondary due to ageing. Genito-pelvic pain/penetration disorder, commonly known as vaginismus, is diagnosed when a patient experiences persistent or recurrent difficulties with one or more of the following for a minimum duration of six months: (a) vaginal penetration during intercourse; (b) marked vulvovaginal or pelvic pain during vaginal intercourse or penetration attempts; (c) marked fear or anxiety about vulvovaginal or pelvic pain in anticipation of, during, or as a result of vaginal penetration; and (d) marked tensing or tightening of the pelvic floor muscles during attempted vaginal penetration.
The worldwide prevalence of FSD ranges from 27% to 70%.(2-4) FSD is associated with various demographic characteristics, including age and educational level. Sociocultural barriers, taboos and misconceptions make the estimation of prevalence difficult. The aetiology of FSD is believed to be multifactorial in nature, involving biological, physiological, anatomical, medical, affective, interpersonal, psychological and context-related factors. Chronic illnesses, such as infections, vascular disease, hypothyroidism, diabetes mellitus and neurologic disease, malignancies and medications can directly or indirectly affect sexual function. Therefore, a multidisciplinary evaluation involving trained specialists and psychologists, as well as a holistic treatment approach that addresses all these issues, is important. Assessment of FSD is best approached using a biopsychosocial approach and should include a sexual history and physical examination. Laboratory testing is usually not required to identify the causes. If left untreated, FSD can affect the quality of life, self-esteem and interpersonal relationships of the sufferer.
HOW RELEVANT IS THIS TO MY PRACTICE?
Combined oral contraceptives (COCs) are an affordable and reversible form of contraception. It is the third most popular contraceptive method, after female sterilisation and intrauterine devices, as it provides a high degree of contraceptive efficacy with an excellent safety profile and is a reversible method of birth control that is available throughout the world.(5) The use of COCs worldwide in women of reproductive age was reported to be around 14% in 2015.(5) While there are existing guidelines from the World Health Organization (WHO) and the United States’ Centers for Disease Control and Prevention (CDC) on the use of COCs,(6,7) they do not mention their effect on sexuality, even though there is evidence to suggest that older-generation COCs can cause female sexual dysfunction.(8)
Starting patients on combined oral contraceptive pills
Prior to starting COCs, the physician should evaluate the patient for contraindications and risks associated with COCs by conducting a thorough history and physical examination (
Table I
Patient assessment before initiation of combined oral contraceptives (COCs).
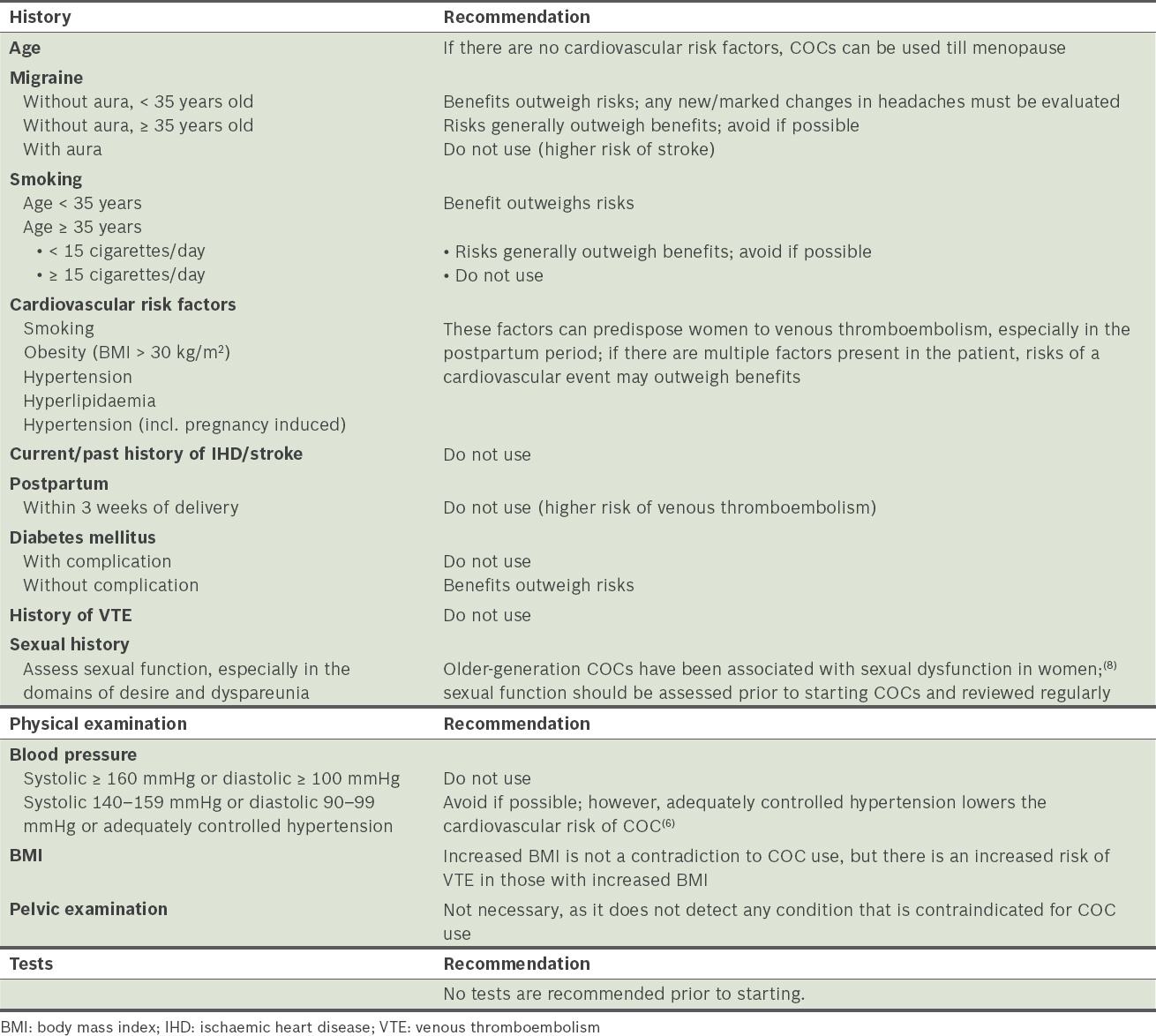
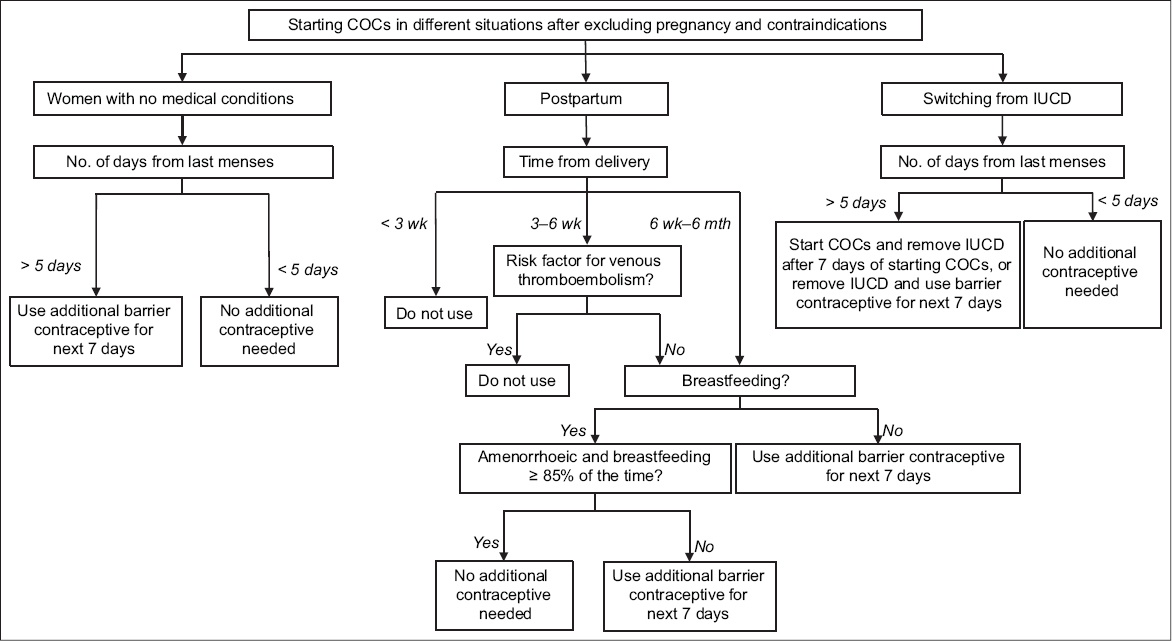
Fig. 1
Algorithm for initiation of combined oral contraceptives (COCs) in women after excluding pregnancy and other contraindications. IUCD: intrauterine contraceptive device
Important considerations when prescribing COCs to women of reproductive age
It is important to consider the effects of COCs on sexual function when prescribing them to women of reproductive age. WHO and CDC have established good practice guidance on how to assess women before prescribing COCs.(6,7) However, neither recommends the screening and assessment of sexual function, which we consider to be important. First, physicians should screen patients for pre-existing FSD, and inform them of the possible sexual side effects of COCs before prescribing them.(8) Second, COCs that contain drospirenone and ethinylestradiol or gestodene and ethinylestradiol are preferred choices for women who are concerned about sexual dysfunction, as they are less likely to cause FSD.(8) Third, patients who are taking COCs need regular review; during clinic visits, physicians should inquire about any sexual side effects, such as decreased sexual desire and dyspareunia. Fourth, in adolescents, a non-COC option for treating acne is preferred over COCs; however, if COCs are prescribed to this age group, the treatment duration should not exceed two years.(9)
Recommended follow-up
There is currently no recommended interval for follow-up after the initiation of COCs. However, in view of some of the effects of COCs, we recommend that patients should be followed up at one month, three months and six months after initiation of COCs, and yearly thereafter. During the follow-up visit, the physician should: (a) assess for compliance and pattern of withdrawal bleeding; (b) assess for any adverse reactions to the medications such as headache; (c) assess for sexual function, particularly in the domains of sexual desire and dyspareunia; (d) measure blood pressure; and (e) calculate the body mass index.
TAKE HOME MESSAGES
-
Thorough history-taking and appropriate physical examination are important to assess a patient’s suitability for use of COCs.
-
There is no indication to perform any tests before starting COCs.
-
COCs can cause or worsen FSD in reproductive women.
-
We recommend that clinicians incorporate taking of a sexual history before prescribing COCs and regular assessments of sexual function in patients taking COCs.
-
Women should be encouraged to discuss the sexual and emotional effects of COCs, and have an informed conversation with their healthcare provider.
Mrs Lim’s COC was switched to drospirenone 3 mg and ethinylestradiol 30 mcg. She was co-managed by a psychologist for her fear and anxiety and a physiotherapist for pelvic muscle stretching and exercises. After about three months, her libido improved and she reports that she no longer has discomfort during sexual intercourse.
SMJ-58-288.pdf


