Abstract
INTRODUCTION
Major depressive disorder (MDD) is one of the most common psychiatric disorders worldwide and has been associated with various sociodemographic risk factors, including age, gender and ethnicity. The present study aimed to establish whether gender-specific differences relating to the prevalence and correlates of MDD exist in the Singapore adult resident population.
METHODS
The Singapore Mental Health Study was a population-based, cross-sectional epidemiological study among Singapore citizens and permanent residents aged 18 years and above. Face-to-face interviews were completed with 6,616 respondents between December 2009 and December 2010. Psychiatric conditions were established using version 3.0 of the World Health Organization Composite International Diagnostic Interview (CIDI). In addition, data relating to chronic medical conditions was captured using a modified version of the CIDI checklist for chronic medical conditions.
RESULTS
The lifetime prevalence of MDD was higher among women (7.2%) than men (4.3%). MDD was more prevalent among men and women who were divorced/separated and widowed women, as compared to those who were single. Among men, MDD was more prevalent among Indian and other ethnicities as compared to Chinese. Of the 417 respondents with MDD, women had significantly higher odds of having generalised anxiety disorder but lower odds of having high blood pressure, as compared to men.
CONCLUSION
The study highlighted key gender-specific correlates of MDD. Given the comorbidities associated with MDD and other psychiatric disorders and/or physical illnesses, these correlates pose additional challenges for care providers.
INTRODUCTION
Epidemiological studies worldwide have consistently reported major depressive disorder (MDD) to be among the most common psychiatric disorders, with an estimated lifetime prevalence in the range of 12% to 16% in Western communities,(1-4) and much lower in Asia, ranging between 3% and 6%.(5-7) MDD can be chronic or recurrent, consequently affecting and impacting individuals for many months, years or even decades. MDD is also associated with significant comorbidity, poor health and mortality.
Certain sociodemographic risk factors, including age, gender and ethnicity, have frequently been associated with MDD. The prevalence of MDD is higher among women compared to men,(8-10) and is often 1.5–3 times higher among women than men.(11-13) Research has also shown that among women, depression is the leading cause of disease-related disability.(11) These findings have been reported in both clinical and general populations and remain evident, irrespective of where the research is conducted and how it is assessed. These gender differences are likely to be a result of a myriad of factors, including biological, social, demographic and/or psychological effects. Gender itself affects many aspects of psychopathology, including prevalence of disorders, expression of symptoms, course of illness, help-seeking behaviour and response to treatment.(14)
Singapore is located off the Malaysian peninsula in Southeast Asia and has a resident population (including Singapore citizens and permanent residents) of 3.8 million people.(15) The Singapore Mental Health Study (SMHS) was a population-based epidemiological study that aimed to establish the prevalence of mental disorders among Singapore residents aged ≥ 18 years. Findings showed that MDD was the most prevalent mental disorder among those examined in the SMHS, which reported a lifetime prevalence of 5.8% and a 12-month prevalence of 2.2%.(6)
Upon further analysis, the SMHS also found that the prevalence of MDD was significantly higher among women, Indians and those who were divorced/separated or widowed. Chronic physical comorbidities were also found to be present in approximately half of all respondents with MDD.(16) Given the high prevalence of MDD among the general adult Singapore population, combined with the significant treatment gap and likelihood of chronic physical comorbidities, the present study aimed to establish whether there were any gender-specific differences relating to the prevalence and correlates of MDD among the adult resident population in Singapore.
METHODS
The SMHS was a cross-sectional epidemiological survey among a representative household sample of Singapore citizens and permanent residents aged ≥ 18 years, who were fluent in English, Mandarin or Malay. Participants were randomly selected from an administrative database that maintains names and sociodemographic details, including age, gender, ethnicity and household addresses, of all Singapore residents. There were 2.7 million residents aged ≥ 18 years living in Singapore at the time when the sample was drawn from the sampling frame. A disproportionate stratified sample (based on age group and ethnicity) was used; the three main ethnic groups in Singapore (i.e. Chinese, Malay and Indian) were equally sampled, while older individuals (aged ≥ 65 years) were over-sampled. All participants provided written consent; for those < 21 years, consent was also obtained from a parent or guardian. Residents who were excluded comprised those who were incapable of completing an interview as a result of severe physical or mental conditions, language barriers or living outside the country during the survey period, and those who were not contactable due to an incomplete or incorrect address. Data collection was carried out between December 2009 and December 2010 following approval from the National Healthcare Group’s Domain Specific Review Board. During this time, face-to-face interviews were completed with 6,616 respondents, yielding a response rate of 75.9%.
Interviewers from an external survey firm conducted the interviews after undergoing extensive training conducted by research staff at the Institute of Mental Health (IMH), Singapore. Interviewers were taught about ethical aspects of the study, administration of survey measures, and logistical procedures relating to fieldwork and reporting during a three-week intensive training period. Upon passing a detailed evaluation, interviewers were initially closely supervised by IMH staff and field executives from the survey firm. To ensure high-quality data, quality assurance processes were implemented throughout the data collection phase and approximately 20% of each interviewer’s cases underwent detailed verification in order to determine any falsification of data. Additional information relating to the methods and procedures employed have been reported in another study.(17)
The presence of MDD and other psychiatric disorders was established using the World Health Organization Composite International Diagnostic Interview (CIDI) version 3.0.(18) CIDI 3.0 is a comprehensive, fully structured instrument that assesses mental disorders in terms of 12-month and lifetime prevalence, according to the definitions and criteria outlined by the Diagnostic and Statistical Manual of Mental Disorders, 4th edition (DSM-IV)(19) and the International Classification of Disease, 10th revision (ICD-10).(20) The SMHS included the following diagnostic modules: MDD; bipolar disorder; generalised anxiety disorder (GAD); obsessive-compulsive disorder (OCD); and alcohol use disorders (including alcohol abuse and alcohol dependence). Diagnostic hierarchy rules and organic exclusions were applied, where relevant.
Respondents were also asked a series of questions relating to treatment contact. To determine if treatment had ever been sought, respondents were asked whether they had ever ‘talked to a medical doctor or other professional’ about the disorder. The ‘treatment gap’ was defined as “the absolute difference between the true prevalence of a disorder and the treated proportion of individuals affected by the disorder”.(21)
The Sheehan Disability Scale (SDS)(22) was administered and captured functional impairment in three aspects – work/school, social and family life – in the worst month of the past year. Responses were scored on the visual analogue scale (range 0–10), and included the labels none (score 0), mild (score 1–3), moderate (score 4–6), severe (score 7–9) and very severe (score 10).
The depression module in the CIDI includes the Quick Inventory of Depressive Symptomatology – Self-Report (QIDS-SR)(23) which assesses symptom severity in patients with MDD during the worst month of the previous year. Scores from the QIDS-SR were converted into clinical severity scores and categories of the Hamilton Rating Scale for Depression(24) based on transformation rules. Categories included none (i.e. not clinically depressed), mild, moderate, severe and very severe. Research has shown very high concordance between the measures.(25)
A modified version of the CIDI 3.0 checklist for chronic medical conditions was also used. Respondents were read the following statement: “I’m going to read to you a list of health problems some people have. Has a doctor ever told you that you have any of the following…”. This was followed by a list of 15 chronic conditions that were considered prevalent in Singapore’s population. These were then reclassified into the following eight types of physical disorders: (a) respiratory disorders (asthma, chronic lung disease [e.g. chronic bronchitis] or emphysema); (b) diabetes mellitus; (c) hypertension and high blood pressure; (d) chronic pain (arthritis or rheumatism, back problems [including disk or spine] or migraine headaches); (e) cancer; (f) neurological disorders (epilepsy, convulsion or Parkinson’s disease); (g) cardiovascular disorders (stroke or major paralysis, heart attack, coronary heart disease, angina, congestive heart failure or other heart diseases); and (h) ulcer and chronic inflamed bowel (stomach ulcer, enteritis or colitis).
Sociodemographic information, including age, gender, ethnicity, education, marital status, income and employment history, was also collected for all respondents. For instruments that were unavailable in Mandarin or Malay, forward translation methods were used to translate these from the English versions.
Statistical analyses were carried out using the Statistical Analysis Software (SAS) System version 9.2 (SAS Institute, Cary, NC, USA). Data was weighted to adjust for oversampling and post-stratified by age and ethnicity distributions between the survey sample and the Singapore resident population in 2007. Descriptive analyses were performed to establish the prevalence of mental disorders and chronic medical conditions, and to describe the sociodemographic characteristics of the study population. We performed multiple logistic regression analyses to examine the odds of having lifetime mental disorders and chronic medical conditions among women when compared to men, after controlling for sociodemographic variables, which included age, ethnicity, marital status, education, employment and income. Analysis of variance and chi-square tests were used to compare the means and rates of continuous and categorical variables between the two groups. Standard errors and significance tests were estimated using the Taylor series linearisation method. Multivariate significance was evaluated using the Wald chi-square test based on design-corrected coefficient variance-covariance matrices. Statistical significance was evaluated at the 0.05 level using two-sided tests.
RESULTS
Of the 6,616 respondents who completed the study, there were slightly more women (51.5%) than men (48.5%). The majority of the respondents were Chinese (76.9%) followed by Malay (12.3%) and Indian (8.3%); 2.4% of respondents belonged to other ethnic groups (
Table I
Prevalence and sociodemographic correlates of major depressive disorder (MDD) by gender among overall sample (n = 6,616).
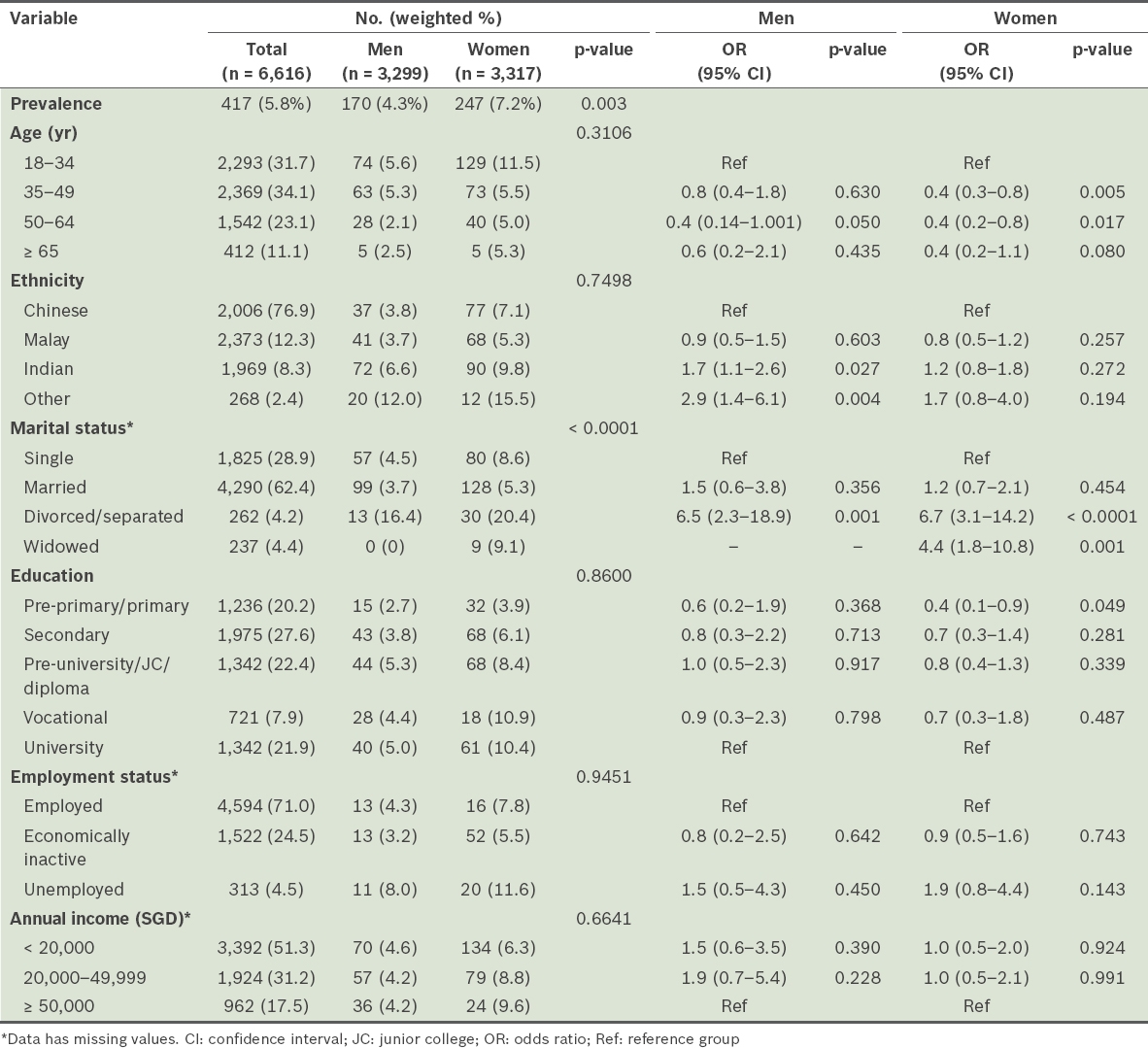
A total of 417 respondents with a lifetime diagnosis of MDD were included in the subsequent analysis. Tables
Table II
Prevalence of lifetime mental disorders among respondents with major depressive disorder (MDD) by gender and results of multiple logistic regression analysis (n = 417).
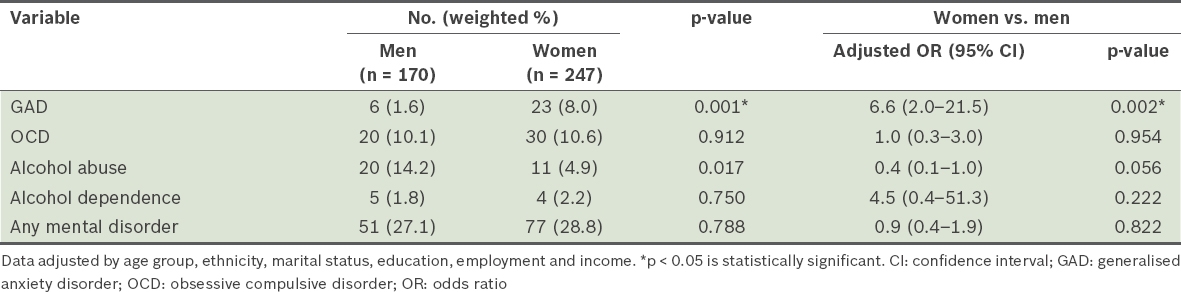
Table III
Prevalence of lifetime chronic physical conditions among respondents with major depressive disorder (MDD) by gender and results of multiple logistic regression analysis (n = 417).
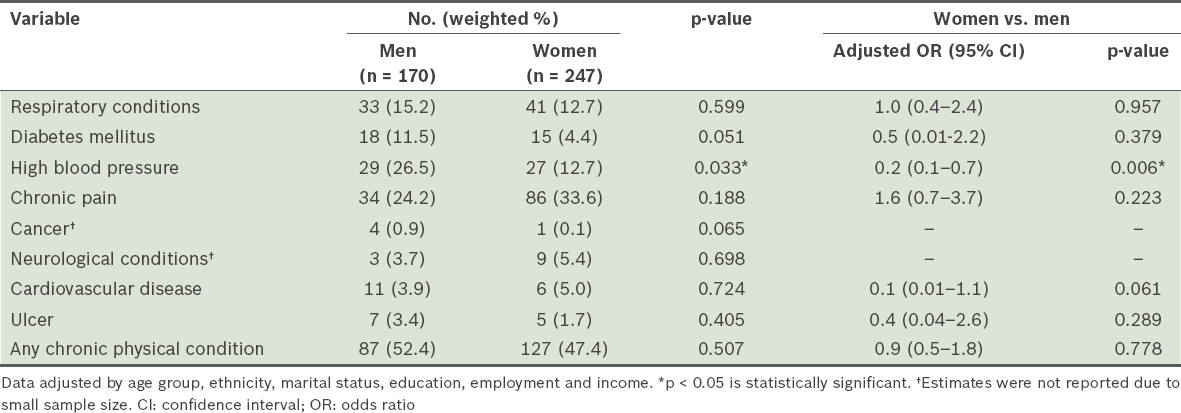
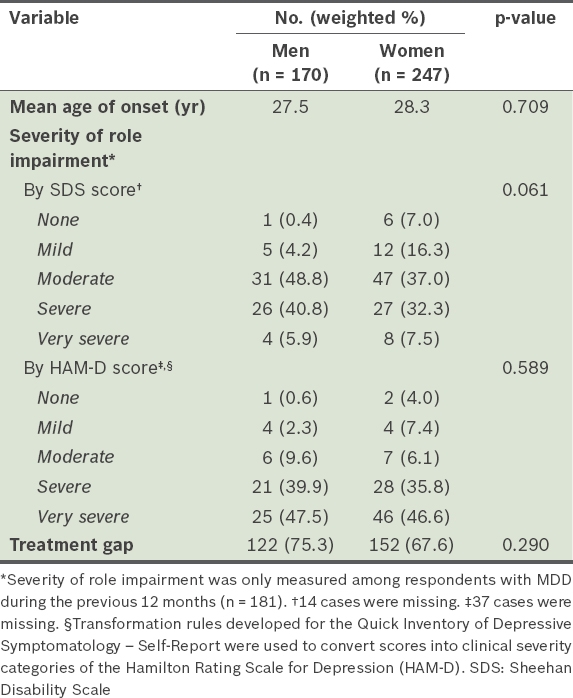
Table IV
Age of onset, severity and treatment gap among respondents with major depressive disorder (MDD) by gender.
DISCUSSION
A number of gender differences were observed among respondents with MDD. Firstly, the prevalence of MDD was higher among women (7.2%) compared to men (4.3%), a finding that has been consistently reported in psychiatric epidemiology. The prevalence of MDD among women in these studies is typically reported to be 1.5–3 times higher than that observed in men,(11) which is consistent with our findings. While the exact reason for such gender differences in relation to MDD prevalence is not known, it is likely to be a myriad of social, behavioural, psychological and biological factors that possibly interact with one another. More specifically, risk factors (e.g. biological susceptibility resulting from hormonal mechanisms, women being more likely and open to seeking help, and social and cultural influences) that have been deemed to lead to stress and added difficulties among women have been explored. However, these have yet to be determined with any certainty.(26)
While these gender gaps occur among patients with MDD and other mental disorders, it appears that the gap is narrowing in some countries.(27,28) Consequently, when explaining gender differences in mental disorders, there has been a shift toward the role of typical stressors, coping resources, and the opportunities available to men and women for expressing psychological distress.(8) This needs to be further explored, particularly as gender roles have changed over time in many parts of the world, including Singapore.
In the overall sample, women with MDD were less likely to be older (35–49 years and 50–64 years compared to 18–34 years). This is consistent with previous research, which has shown that younger age is a risk factor for MDD,(29) although this finding is not gender specific. MDD was also more likely among respondents belonging to the younger age group (age 18–34 years) in the overall sample;(16) when the sample was split by gender, it became evident that women were driving this association. Possible reasons that may account for these differences between the genders at a younger age include psychological characteristics; for instance, neuroticism may result in more vulnerable responses to life events.(30) While women are at greater risk of MDD at a younger age, compared to men, this may only partly account for their preponderance in rates of adult MDD.(26) In cases where age-related recall bias is less likely to be an influence, it is not surprising that the women with MDD are more likely to be younger, as MDD has been known to have an early age of onset irrespective of gender.(1,29,31)
Unsurprisingly, being divorced or separated was also a risk factor for MDD among both men and women when compared to single respondents, and this is likely explained by the nature of such a stressful life event. The cross-sectional nature of this study did not allow us to determine whether MDD caused divorce/separation or vice versa. However, it is thought to be bidirectional(32) and could also be explained by a number of social processes. For example, people with a mental illness may be less likely to get married or more likely to experience more marital difficulties, which may result in divorce or separation.(33) Widowed women also had higher odds of MDD than their single counterparts, as the death of a spouse may have adversely affected their mental health.(34) Contrary to some findings that marriage was a ‘buffer’ or protective factor for depression,(3,35) others have found that married women were more likely to have higher rates of depression compared to those who were divorced or separated;(36) however, we did not find this association among our sample.
Interestingly, we found that, among women, respondents with less education (pre-primary or primary school education only) had lower odds of MDD than those with university-level education. The association of education with MDD status and risk is debatable. Several studies have found that MDD risk did not differ by education,(3,9) while a recent study indicated a lower depression rate among those with more education, with the association between higher education and improved mental health being significantly more prominent among women compared to men.(35) While we are unable to ascertain the exact reason behind our findings, it is important to highlight that the number of men (n = 15) and women (n = 32) with MDD who had pre-primary or primary education in our study was quite small, and therefore this finding should be viewed with caution.
In our study, the risk of MDD was higher among men of Indian ethnicity when compared to Chinese men. Currently, we are unable to elucidate why Indian men are at higher risk of MDD. This could be due to a biological vulnerability that has yet to be explained, or environmental factors, including acculturation, resulting in the internalisation of disorders such as depression among ethnic minority groups.(37) In Singapore, Indians are a minority ethnic group compared to the Chinese and Malays. Notably, previous research, conducted largely in the United States, found a lower-than-expected prevalence of depression among minority ethnic groups compared to white Americans.(38-40) These ethnic or racial disparities may be a result of methodological procedures, including inconsistencies and discrepancies in how depression is measured, as well as how racial or ethnic groups are classified and the use of different controls as confounding factors. Caution is required when interpreting findings based on ethnic or racial groups, as comparisons are filled with challenges and complexities. Ethnic or racial categories are “at best approximations of societally defined groupings to which individuals are assigned based largely on skin colour, country of origin or … language or dialect spoken”,(41) and therefore may mask wide-ranging heterogeneity and complicate more granular differences.
Results from our study showed that among the subsample of respondents with MDD, there were a number of gender differences relating to other psychiatric comorbidities. For example, GAD was significantly more prevalent among women compared to men, and women with MDD were 6.2 times more likely to have a comorbid GAD diagnosis during their lifetime. Similar to MDD, psychiatric epidemiology has consistently found that an anxiety disorder is significantly more likely to occur in women during their lifetime, as compared to men.(42-44) It is important to note, however, that in our subsample of respondents with MDD, there was only a small number of men with GAD, which resulted in a wide CI.
Among those with MDD, men were significantly more likely to have high blood pressure compared to women (p < 0.006). Hypertension prevalence, as reported in a worldwide systematic review, tends to occur more frequently among men than women in most countries.(45) Various explanations have been proposed for gender differences in hypertension among respondents with MDD, including social factors. For example, research has shown that men are less likely to perceive themselves as being at risk of developing various health problems, and the two genders generally have different opinions on healthy behaviours.(46,47) Yang and Reckelhoff also attribute these gender differences to sex hormones, suggesting that premenopausal women are comparatively protected against hypertension in comparison to postmenopausal women and men.(48) The cross-sectional nature of this study, however, did not allow any causal relationships to be established.
In addition to the psychiatric and physical comorbidities observed among respondents with MDD, we also explored gender differences by age of onset, impairment severity and treatment gap. Women with lifetime MDD tended to have a slightly later age of onset of MDD, but less severe impairment based on SDS and HAM-D. Women also had a lower treatment gap compared to men. However, none of these differences were statistically significant. A cross-sectional epidemiological study, conducted by Gili et al, which explored gender differences in disability among those with MDD, also found no statistically significant difference in functioning between men and women, suggesting that the relationship between depression and functioning or impairment is not gender-dependent.(49)
Our findings should be viewed in the context of some limitations. First, depressive symptoms were assessed based on self-report and may be subject to various biases. Furthermore, it has been suggested that women may be more willing to admit their depressive symptoms or experiences to an interviewer than men.(50) Second, we were unable to establish the cause-and-effect relationship of mental and physical disorders due to the cross-sectional nature of the study. Third, people residing in nursing homes, prisons and hospitals were excluded from the survey. Lastly, due to respondent burden, we were unable to include all psychiatric disorders or physical illnesses that may co-occur with MDD, which might have resulted in an underestimation of MDD comorbidities.
These limitations notwithstanding, the strengths of our study are the use of a well-established instrument for collecting information, a large sample size and a high response rate that provides confidence in the results and improves generalisability. This study has highlighted key gender-specific predictors and risk factors for MDD. Given the comorbidities with MDD and other psychiatric disorders and/or physical illnesses, these correlates pose additional challenges for care providers, and also emphasise the importance of early detection and screening of conditions such as hypertension, particularly among men with MDD. Furthermore, the finding that MDD is more prevalent among women, particularly in the younger age group, suggests the need to involve and educate parents and teachers about the signs of depression. The importance of establishing early detection and screening systems in various settings, including educational institutions and general medical practices, is key and could include innovative measures, such as the Internet or telephone-based therapy.
ACKNOWLEDGEMENTS
The study was supported by funding from the Singapore Millennium Foundation and the Ministry of Health, Singapore.


