Dear Sir,
Although there is no causal treatment of mitochondrial disorders (MIDs) yet available,(1) treatment strategies to enhance respiratory chain (RC) functions, eliminate noxious compounds, shift the heteroplasmy rate, alter mitochondrial dynamics, transfer cytoplasm, or treat genes are increasingly applied.(1) One of the pharmacological compounds repeatedly reported for treating MIDs are glucocorticoids (GCs).(2) GCs may exhibit either beneficial or detrimental effects, or none at all. However, while the mechanism of action of GCs in the treatment of MIDs remains largely unknown, there are indications that GCs act not only in the nucleus but also in mitochondria.(3,4) Herein, we summarise and discuss the role of GCs in the treatment of human MIDs.
With regard to mode of action, there is increasing evidence that mitochondria are controlled by the action of GCs, as GC receptors are found in mitochondria and GC response elements also reside in mitochondrial DNA (mtDNA).(3) The expression of mtDNA genes can thus be regulated by either interaction with nuclear genes or direct action of GCs on the mtDNA and its RC, transfer RNA, and ribosomal RNA genes.(3) Additionally, GCs directly stimulate mitochondrial transcription via mitochondrial GC receptors, affecting RC biosynthesis.(4) GCs have also been shown to enhance mitochondrial biogenesis and modulate the intrinsic, but not the extrinsic, pathway of apoptosis,(5) and induce apoptosis, upregulate Bim, decrease mitochondrial membrane potential, release cytochrome c and cause DNA fragmentation.(6)
Regarding the clinical effect of GCs on MIDs published in the literature, there are only case reports or case series available (
Table I
Effects of glucocorticoids in mitochondrial disorders (MIDs).
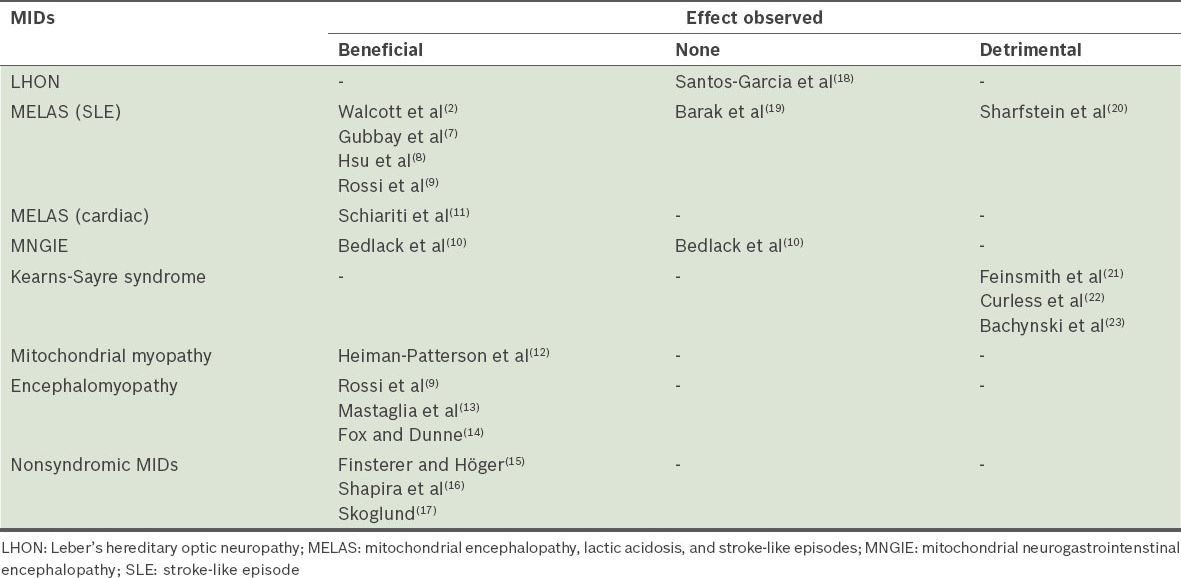
In conclusion, the evidence level for a beneficial effect of GCs in MIDs is low. Only case reports are available and no randomised controlled trials have been carried out to investigate the effect of GCs in the treatment of MIDs. Although single cases seem to profit from the use of GCs, others may experience disease deterioration. At present, there is limited evidence to support deciding which cases will benefit from the use of GCs, and in which cases the use of GCs will be detrimental. MELAS patients seem to profit from the use of GCs, whereas KSS patients seem to experience disadvantages.
Yours sincerely,


