Abstract
INTRODUCTION
Postoperative nausea and vomiting (PONV), and postoperative pain are common during the early postoperative period. In addition to these problems, elderly patients risk developing postoperative confusion. This study aimed to identify the risk factors associated with these problems, and the extent of these problems, in a Singapore inpatient surgical population.
METHODS
Over a period of six weeks, we surveyed 707 elective surgical inpatients aged ≥ 18 years who received general anaesthesia and/or regional anaesthesia.
RESULTS
The incidence of PONV was 31.8%(95% confidence interval [CI] 34.8–41.9). The incidence increased with increasing Apfel score (p < 0.001) and were higher in female patients (odds ratio [OR] 1.74, 95% CI 1.28–2.36), non-smokers (OR 1.72, 95% CI 1.04–2.88), patients with a history of PONV and/or motion sickness (OR 3.45, 95% CI 2.38–5.24), patients who received opioids (OR 1.39, 95% CI 1.03–1.88), and patients who received general anaesthesia (OR 1.76, 95% CI 1.11–2.79). Moderate to severe pain at rest and with movement were reported in 19.9% and 52.5% of patients, respectively. Among the patients who were predicted to experience mild pain, 29.5% reported moderate pain and 8.1% reported severe pain. The prevalence of postoperative confusion was 3.9% in the geriatric population.
CONCLUSION
Higher Apfel scores were associated with a higher risk of PONV and multimodal treatment for postoperative pain management was found to be insufficient. The incidence of postoperative confusion was low in this study.
INTRODUCTION
Postoperative nausea and vomiting (PONV), and postoperative pain are among the most common problems encountered during the early postoperative period. Recent studies report that the incidence of PONV ranges from 9% to 56%, while a meta-analysis of these studies found that the overall occurrence of PONV was 28.3%.(1) Similarly, a systematic review of published data on postoperative pain management found that 29.7% of patients reported moderate to severe pain at some point during the 24-hour period after surgery.(2)
Patients have ranked vomiting and incisional pain as the top fears that they have after anaesthesia and surgery.(3) Other than that, postoperative pain and its management have been shown to affect the risk of developing postoperative delirium in elderly patients.(4,5) Therefore, in the present study, we evaluated the risk factors for these problems, as well as the extent of these postoperative problems, in a Singapore inpatient surgical population.
METHODS
A survey of postoperative outcomes was conducted over a six-week period at a tertiary institution in Singapore. All elective surgical inpatients aged ≥ 18 years who received general anaesthesia and/or regional anaesthesia were eligible for inclusion in the present study. The exclusion criteria were as follows: (a) underwent monitored anaesthetic care without general or regional anaesthesia; (b) admitted to the intensive care unit, neurosurgery high-dependency unit or the psychiatric ward in the immediate postoperative period; (c) had cognitive impairment; (d) had difficulty with verbal communication; and (e) spoke languages other than English, Mandarin, Chinese dialects or Malay. The Domain Specific Review Board of the National Healthcare Group was informed and consulted about the department’s audit, and such quality improvement work was exempted from formal review. We obtained verbal consent from each patient prior to the start of the interview.
Two interviewers administered a standardised survey questionnaire (see
The severity of postoperative pain was assessed during the interview using a descriptive verbal rating scale (i.e. no pain, mild, moderate or severe pain). Such a scale was used because it is easy to administer in different languages and can be easily understood by patients. Five different pain scores for operative site pain were recorded: (a) pain at rest 24 hours after surgery; (b) pain during movement 24 hours after surgery; (c) highest pain score over the last 24 hours since surgery; (d) lowest pain score over the last 24 hours since surgery; and (e) average pain score over the last 24 hours since surgery. ‘Pain at rest’ refers to pain experienced when the patient is in bed without movement, while ‘pain during movement’ refers to dynamic pain (e.g. pain when sitting up, pain when turning in bed, pain when getting out of bed, pain during deep breathing, and pain when coughing or straining). The incidence of postoperative sore throat was also recorded.
The data on postoperative pain was evaluated according to the type of surgery the patients underwent and the anticipated level of postoperative pain. The anticipated levels of postoperative pain were decided based on reported data and our clinical experience.(7) Mild pain was anticipated for minor head and neck, minor limb, minor urological and minor lower abdominal surgeries (e.g. thyroidectomy, eye surgery, limb vascular surgery, arthroscopy, endoscopic urological procedures, hernia, cervix and vaginal surgery, and wound debridement). Moderate pain was anticipated for major head and neck, intermediate limb, minor thoracic and breast, minor upper abdominal, intermediate lower abdominal and minor spinal surgeries (e.g. oral and throat surgery, limb fracture open reduction and internal fixation, discectomy, mastectomy, cholecystectomy, appendectomy, lower segment Caesarean section and hysterectomy). Severe pain was anticipated for major limb, major thoracic, major upper and lower abdominal and major spinal surgeries (e.g. shoulder reconstruction surgery, lobectomy, colectomy, hepatectomy, gastrectomy, hip or knee replacement and spinal fusion surgery).
The patients’ premorbid neurological status and mental state were also recorded. We used the Confusion Assessment Method for the intensive care unit (CAM-ICU)(8) to assess postoperative confusion. To determine the presence or absence of emergence delirium, postanaesthesia care unit (PACU) clinical charts were reviewed. If the term ‘restless’, ‘agitated’, ‘confused’ or ‘delirious’ were documented by the PACU anaesthetist, the patient was taken to have emergence delirium. Data on the antiemetics and analgesia prescribed and administered were obtained from the intraoperative anaesthetic chart and the electronic inpatient medical record system.
Statistical analysis was performed using IBM SPSS Statistics software version 20.0 (IBM Corp, Armonk, NY, USA). Confidence intervals were obtained with calculators for the Newcombe score using the Wilson procedure with correction for continuity. Chi-square tests were used to determine statistical significance. Chi-square test for trend was used to evaluate the relationship of PONV and Apfel score. Univariate analysis was used to explore the risk factors that were thought to contribute to PONV (e.g. Apfel factors, type of anaesthesia, use of total intravenous anaesthesia [TIVA], use of nitrous oxide and age).
RESULTS
Although 727 patients were eligible for inclusion in the present study, 20 patients were excluded due to various reasons – 10 patients were excluded because a questionnaire in their language was not available, 5 patients refused participation and 5 were not present in their rooms despite three attempts to reach them. Among the 10 patients who were excluded because a questionnaire in their language was not available, 3 were of Indian ethnicity and 7 were of ethnicities other than Chinese, Malay or Indian.
A total of 707 patients were included in the present study; their profiles are presented in
Table I
Profile of the patients (n = 707).
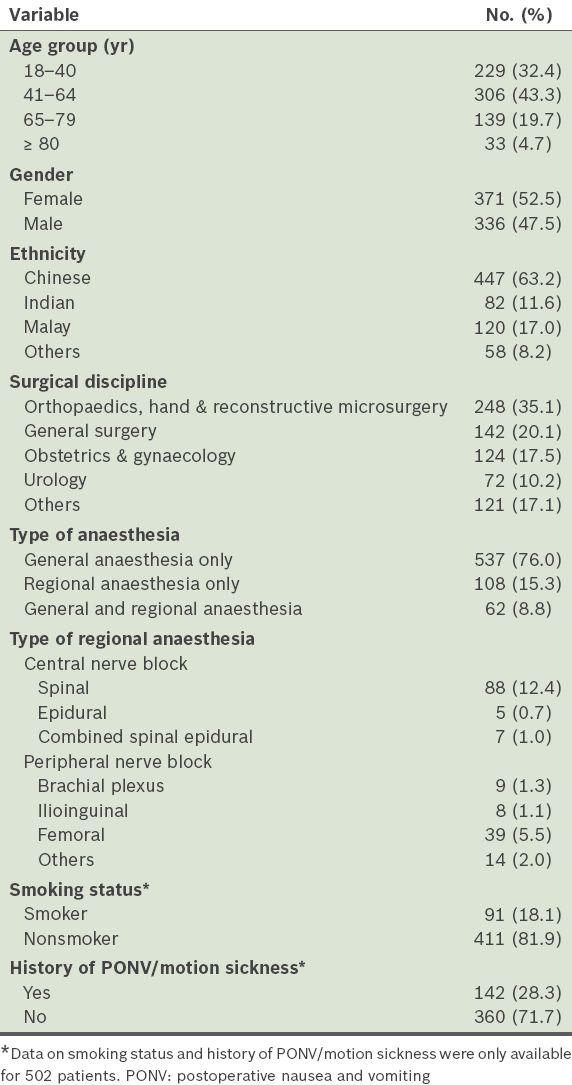
The incidences of postoperative nausea and postoperative vomiting were 31.8% (95% confidence interval [CI] 28.4–35.3) and 24.6% (95% CI 21.4–27.8), respectively. Among the patients, 18.0% (95% CI 15.1–20.8) experienced both nausea and vomiting, and the overall incidence of PONV, defined as nausea and/or vomiting experienced in the first 24 hours after surgery, was 38.3% (95% CI 34.8–41.9). The severity of nausea and the number of vomiting episodes are shown in
Table II
Proportion of patients with nausea and vomiting within the first 24 hours after anaesthesia and surgery (n = 707).
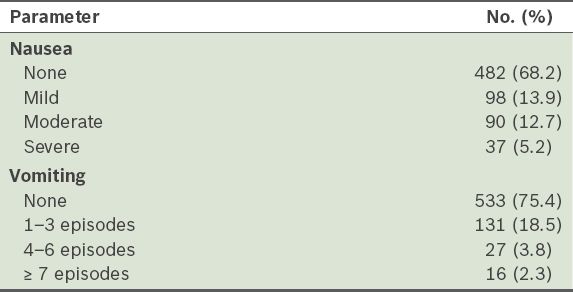
Table III
Proportion of patients with postoperative nausea and vomiting for each Apfel score.

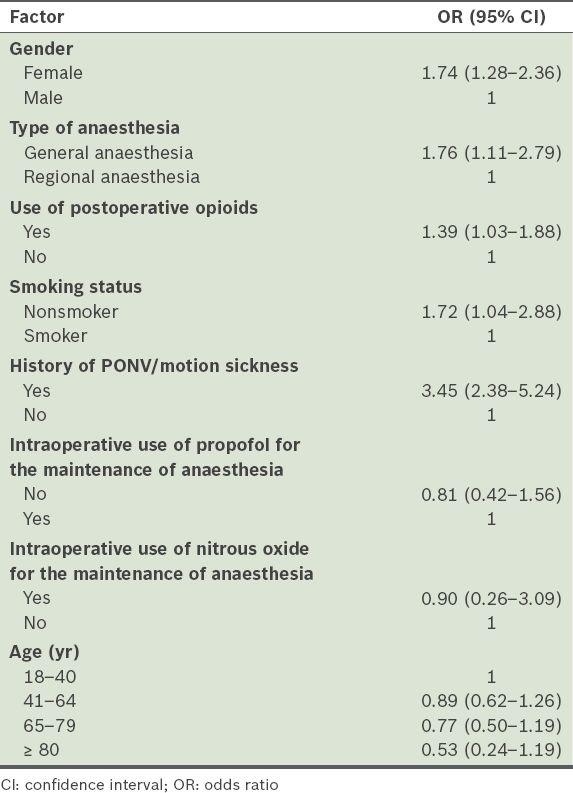
The impact of the risk factors on PONV is shown in
Table IV
Results of the univariate analysis performed to determine the factors affecting postoperative nausea and vomiting (PONV).
The prevalence of the severity of the postoperative pain experienced by the patients is summarised in
Table V
Prevalence of postoperative pain among the patients (n = 707).
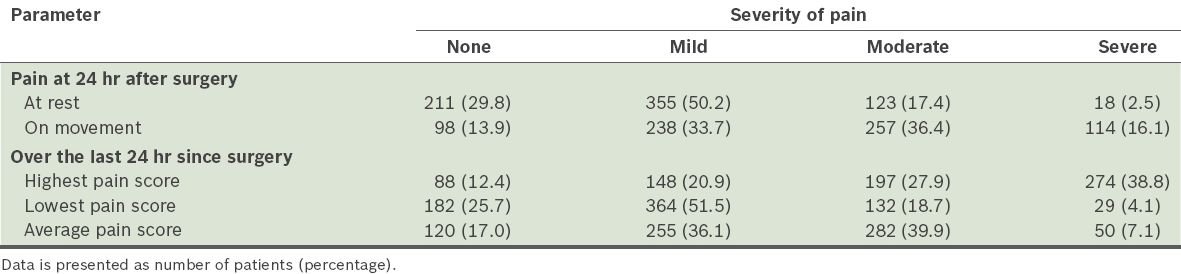
Table VI
Average pain experienced by patients over the 24 hours after surgery, according to the type of surgery (n = 707).
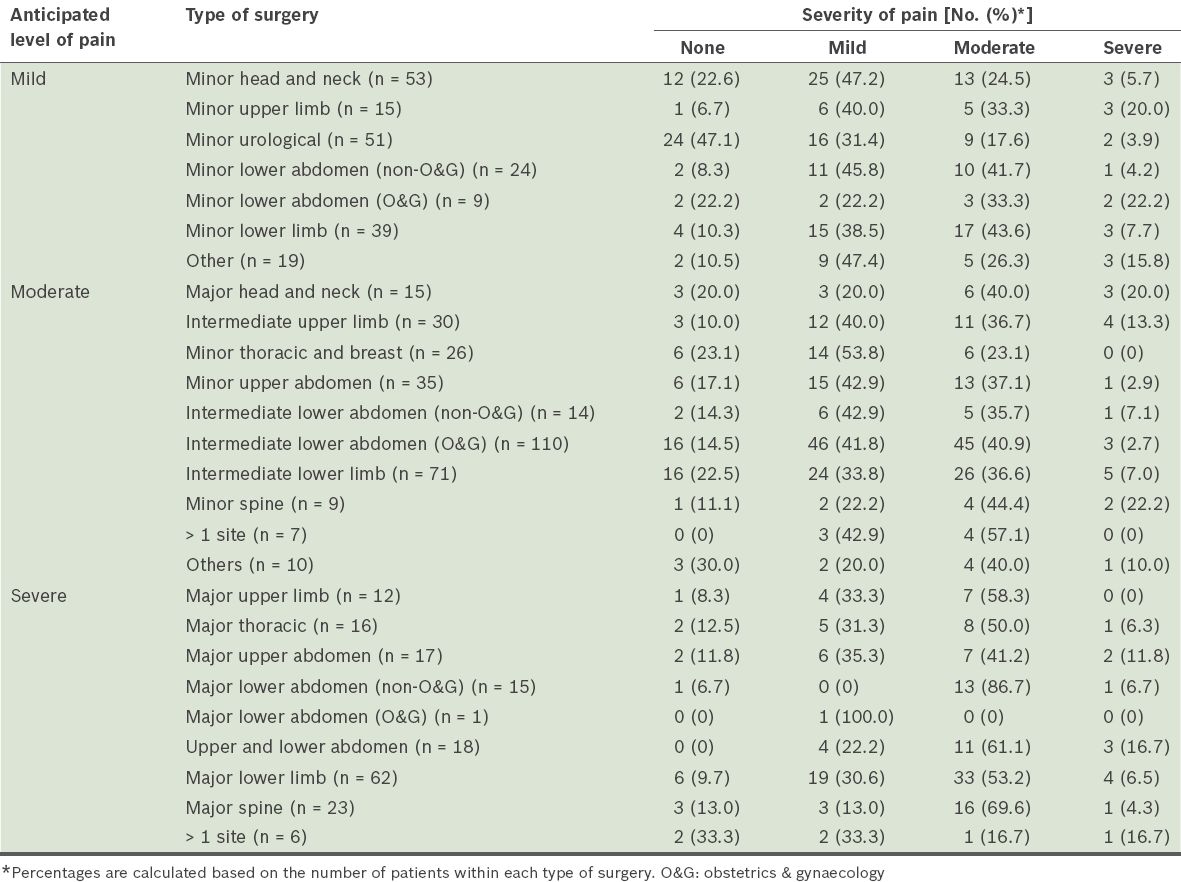
The analgesic prescriptions and administration for the different types of surgery are shown in
Table VII
Pharmacologic analgesic use according to anticipated level of pain.
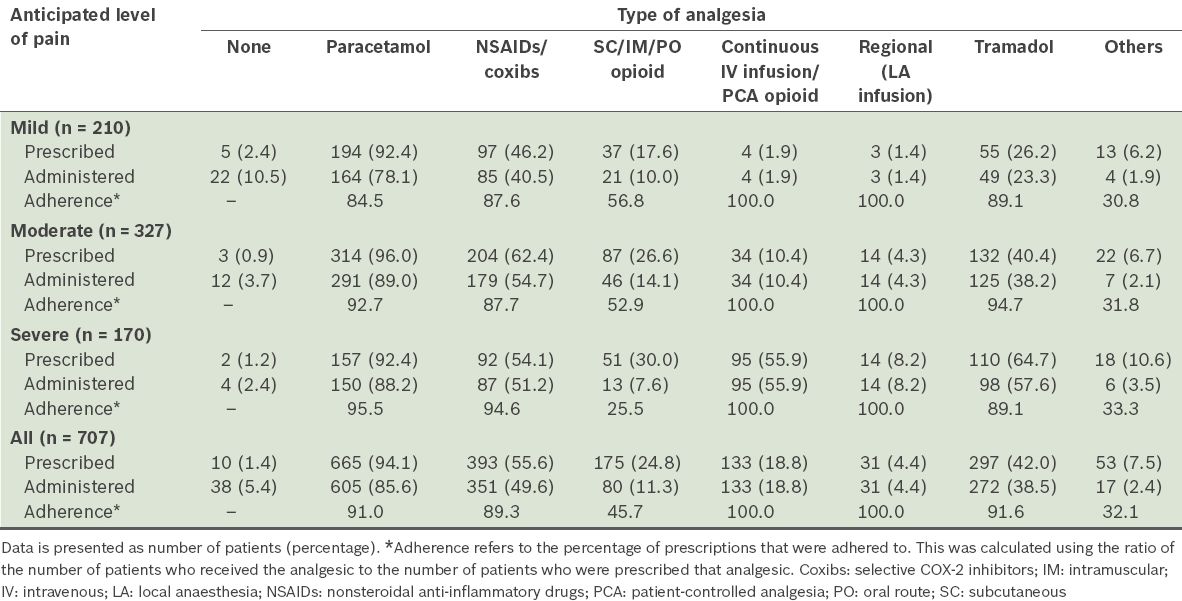
Table VIII
Incidence of postoperative sore throat.
In the present study, as 28 patients had dementia or other conditions that could cause confusion prior to surgery, CAM-ICU was not administered to them. Of the 679 patients who were well prior to surgery, 155 were aged > 65 years. Overall, the incidence of postoperative confusion at 24 hours was 1.0% (n = 7). Among the patients aged > 65 years, the incidence of postoperative confusion at 24 hours was 3.9% (n = 6). The overall incidence of emergence delirium was 0.6% (n = 4).
None of the patients in the present study were documented (in the case notes) to have an altered mental state by any of the ward doctors, nurses or allied health professionals, in the 24 hours after surgery, including the seven patients who were diagnosed using CAM-ICU. Atropine was not used in any of the seven patients who experienced postoperative confusion.
DISCUSSION
The present study showed a high incidence of PONV and moderate to severe postoperative pain in surgical inpatients. These two factors would have affected the quality of the patients’ recovery as well as their perioperative experience. The overall occurrence of PONV in the present study is comparable to the rates that were reported in a meta-analysis of 22 studies (n = 95,154).(1) In that meta-analysis, a large variation in the incidence of PONV (from 9% to 56%) was found.(1) There is much room for improvement in the effort to reduce the proportion of patients who experience PONV.
In the present study, it was of concern that a small proportion of patients reported severe nausea and frequent vomiting (≥ 7 times), despite receiving antiemetic prophylaxis and treatment. In a study by Apfel et al, in which the Apfel scoring system was used, the authors reported PONV rates of 10%, 21%, 39%, 61% and 78% for the Apfel scores of 0, 1, 2, 3, and 4, respectively.(6) In comparison, there was a higher occurrence of PONV in the patients with lower Apfel scores (i.e. 0 and 1) and a lower occurrence of PONV in those with higher Apfel scores (i.e. 2, 3 and 4) in the present study (
A large proportion of the patients in the present study had moderate to severe pain within the first 24 hours after surgery. This was despite routine multimodal analgesic use in the patients in whom moderate to severe postoperative pain was anticipated. The prescription of opioids in patients in whom moderate pain was anticipated was low and the actual administration was even lower. Prescriptions of subcutaneous, intramuscular and oral opioids were adhered to only 45.7% of the time despite the high pain scores. In our hospital, PCA is mainly used when severe pain is anticipated. In the present study, 18.8% of the patients were prescribed and administered PCA. This usage is similar to that of other studies.(9)
In the present study, the patients’ experience of moderate to severe pain is similar to that of other studies.(2,7,10) In addition, we found that non-opioid analgesics were most commonly used in the present study (
The Acute Pain Service in our hospital manages patients who are prescribed PCA morphine and regional analgesia infusions. Generally, patients who have undergone extensive and complex surgery are prescribed such analgesics. In the present study, 2.0% of these patients had severe pain at rest and 25.5% had severe pain with movement. This suggests that there could be a limit to the effectiveness of PCA morphine.
The results of the present study also suggest that it is difficult to accurately predict the extent of pain that the patients will experience and that it is common to underestimate the extent of the pain that will be experienced. Most patients who underwent surgeries that were predicted to result in mild postoperative pain were not prescribed opioids. Examples of such surgeries include minor gynaecological operations, upper limb peripheral vascular surgery, wound debridement and inguinal hernia repairs, where contrary to prediction, a substantial proportion are known to experience moderate or even severe postoperative pain. This was reported in a recent multicentre study.(11)
The methodology employed in the present study has inherent limitations. The use of data obtained from a single interview for a 24-hour period is limited by recall bias. However, as the documentation of pain assessment in routine clinical charts would have been done by several different nurses for each patient, it would be difficult to ensure that the assessments were done in a consistent manner for all the patients. While future studies could include the use of a trained assessor to obtain data contemporaneously, this was not feasible in the present study.
In the present study, multimodal or balanced analgesia was consistently used for patients who had undergone major surgeries in which severe postoperative pain was anticipated. Despite this, acute postoperative pain management was not optimal. Therefore, regular opioid prescription and administration may be needed to improve pain management. Procedure-specific postoperative pain management guidelines, regular staff training on postoperative pain management, and systematic information and education for the patient before surgery may improve postoperative pain management.(12)
Postoperative sore throat was found to be common with tracheal intubation and supraglottic airways in the present study. As such, we recommend that patients be informed of this risk preoperatively. While there were only four surgical inpatients whose anaesthesia involved the use of a facemask (i.e. tracheal tubes or supraglottic airways were not used), none of these patients experienced postoperative sore throat.
Postoperative confusion has mainly been studied in geriatric patients. Its reported incidence varies widely between 0% and 75.3%;(13) a review of 80 studies estimated the incidence to be about 38.6%.(13) This wide variation reflects differences in: (a) the diagnostic criteria for delirium; (b) the population studied; and (c) the method of postoperative surveillance for delirium.(13) In the present study, using CAM-ICU, we found that the incidence of postoperative confusion in patients aged ≥ 65 years was 3.9%. Interestingly, none of the patients who were found to have postoperative confusion using CAM-ICU were documented to have confusion in the ward. This suggests that screening tools such as CAM-ICU can be a more sensitive measure of postoperative confusion. The incidence of postoperative confusion in the present study is comparable to that of another study that also used CAM-ICU (the incidence of postoperative confusion in the latter study was 7.6%).(8) Removal of precipitating factors, as well as treatment, is required to prevent persistent and worsening confusion.(14)
In summary, the present study identified factors that increased the risk of PONV in a local patient population. Apfel risk scores were found to be associated with the risk of PONV; a significant trend of increasing PONV was observed with higher Apfel scores. Multimodal treatment, which is commonly used for postoperative pain management, was found to be insufficient. We also found that non-opioid analgesics were most commonly used for postoperative pain management. It is difficult to predict the extent of postoperative pain, and patients having minor surgery may have moderate or even severe pain. The CAM-ICU screening tool showed a low incidence of postoperative confusion in our study population.
Annex
-
Have you experienced any nausea since your operation?
◻ Yes ◻ No
-
(If Yes to Q1) How would you rate the severity of nausea?
◻ Mild ◻ Moderate ◻ Severe
-
Have you experienced any vomiting since your operation?
◻ Yes ◻ No
-
(If Yes to Q3) On how many occasions have you vomited since your operation?
◻ 1 ◻ 2 ◻ 3 ◻ 4
◻ 5 ◻ 6 ◻ > 7
-
How is your pain at rest now?
◻ Mild ◻ Moderate ◻ Severe
-
How is your pain with movement now?
◻ Mild ◻ Moderate ◻ Severe
-
What is your worst pain over the last 24 hours?
◻ Mild ◻ Moderate ◻ Severe
-
What is your least pain over the last 24 hours?
◻ Mild ◻ Moderate ◻ Severe
-
How is your average pain over the last 24 hours?
◻ Mild ◻ Moderate ◻ Severe
-
Do you have a sore throat?
◻ Yes ◻ No
Acute onset and fluctuating course Any acute change in mental status from the patient’s baseline? (Assessed by interviewer with reference to pre-op clinical charts) ◻ Yes ◻ No Inattention (assessed by interviewer) Difficulty focusing attention? ◻ Yes ◻ No (If present or abnormal) Did this behaviour fluctuate during the interview? (That is, tend to come and go or increase and decrease in severity) ◻ Yes ◻ No ◻ Not applicable Disorganised thinking Thinking disorganised or incoherent? (Yes if incorrect answers to ≥ 2 of the following 4 ‘Yes/No’ questions) ◻ Will a stone float on water? ◻ Are there fish in the sea? ◻ Does 1 kg weigh more than 2 kg? ◻ Can you use a hammer to pound a nail? Altered level of consciousness What is the patient’s level of consciousness? (Assessed by interviewer) ◻ Alert (normal) ◻ Vigilant (hyper-alert) ◻ Lethargic (drowsy but easily roused) ◻ Stuporous (difficult to rouse) ◻ Comatose (unrousable)


