Abstract
INTRODUCTION
Polypharmacy and inappropriate prescribing are associated with negative health outcomes in the elderly. Several prescribing tools have been developed to assess medication appropriateness. Explicit (criteria-based) tools often do not take into account patients’ preferences and comorbidities, and have little room for individualised clinical judgement.
METHODS
A cross-sectional observational study was conducted in 243 elderly patients admitted to the Geriatric Medicine service in a Singapore tertiary hospital over one month. We incorporated an implicit (judgement-based) tool developed by Scott et al into a mnemonic, ‘S-I-R-E’, to assess medication appropriateness: S = symptoms (‘Have symptoms resolved?’), I = indication (‘Is there a valid indication?’), R = risks (‘Do risks outweigh benefits?’) and E = end of life (‘Is there short life expectancy limiting clinical benefit?’).
RESULTS
Inappropriate prescribing was present in 27.6% of patients. The most common reason for inappropriateness of medications was lack of valid indication (62.2%), followed by high risk-benefit ratio (20.7%). The most common medications that lacked valid indication were supplements and proton pump inhibitors. Polypharmacy was found in 93% of patients and was significantly associated with inappropriate prescribing (p = 0.047).
CONCLUSION
Inappropriate prescribing and polpharmacy are highly prevalent in the hospitalised elderly. The ‘S-I-R-E’ mnemonic can be used as a memory aid and practical framework to guide appropriate prescribing in the elderly.
INTRODUCTION
Inappropriate prescribing is an increasingly common iatrogenic problem seen in healthcare today. Advances in medical research and drug development have increased life expectancy, but have resulted in the accumulation of multiple morbidities in our elderly. The burden of polypharmacy and inappropriate prescribing has been well-studied and is associated with negative health outcomes such as increased hospitalisation, institutionalisation, falls and impaired mobility, malnutrition and mortality in the elderly.(1) In the United States, it was found that 49% of hospitalised elderly received at least one inappropriately prescribed medication.(1) In Singapore, the prevalence of inappropriate prescriptions in nursing homes was reported to be as high as 70% in one study.(2)
The term ‘inappropriate prescribing’ has no established or agreed definition. In the literature, inappropriate prescribing entails the prescription of a drug when the net risk of the drug outweighs its clinical benefit. Drug risk includes adverse drug reactions that can lead to increased morbidity, mortality and healthcare utilisation in the patient.(3,4) Common causes for adverse drug reactions in the elderly include therapeutic duplication, drugs with no valid indication or limited benefits, significant drug-drug interactions and incorrect dosage of drugs. Data on the safety and efficacy of drugs in the geriatric population is limited, as the elderly are often excluded from clinical drug trials. This problem is further compounded by age-related changes, such as lower reserve capacities and altered homoeostatic mechanisms that affect the pharmacology of drugs. Hence, the elderly are most vulnerable to the harms of inappropriate prescribing.(5)
Explicit (criteria-based) tools have been developed to measure appropriateness and guide deprescribing, including the Beers criteria and STOPP-START criteria.(6,7) However, application of these tools in routine practice may not be practical in time-constrained settings. In addition, explicit tools often do not take into account a patient’s comorbidities and individual preferences, and also have limited transferability between countries due to variations in prescribing patterns and drug availability.(8,9) In recent years, more attention has been given to a more practical approach, using implicit (judgement-based) tools to assess medication appropriateness. This method systematically identifies medications in which the net harm of the medication outweighs the clinical benefit, assessed within the context of an individual patient’s care goals, level of functioning, life expectancy and preferences.(10) This approach supports patient-centred decision-making, a central pillar of good geriatric care.
To our knowledge, there has been no published data on the prevalence of inappropriate prescribing in the acute inpatient geriatric setting and the utility of a tool to guide deprescribing. This study aimed to explore the prevalence of inappropriate prescribing in our geriatric inpatient cohort, assessing medication appropriateness using an implicit deprescribing algorithm by Scott et al.(10)
METHODS
This was a cross-sectional observational study conducted in three acute geriatric wards in Changi General Hospital, Singapore, over a period of one month, from August 2016 to September 2016. Patients admitted from the emergency department to the acute geriatric wards would have met the hospital’s established admission criteria to the Geriatric Medicine service: (a) aged ≥ 80 years with a recognised geriatric syndrome; or (b) on active follow-up with a hospital geriatrician. Patients who were transferred from other inpatient specialist units following inpatient geriatric specialist consultation were also included.
Demographic, social and clinical data of included patients was collected by advanced practice nurses specialising in geriatric care. The Barthel Index of activities of daily living and Abbreviated Mental Test (AMT) were used to assess the functional and cognitive status of the patients, respectively. As part of routine care, ward pharmacists performed medication review and reconciliation for all patients admitted to the geriatric wards and any medication issues were raised to the treating team.
Medication appropriateness was assessed by ward pharmacists using the deprescribing algorithm published by Scott et al,(10) which we reorganised into an easy-to-remember mnemonic, ‘S-I-R-E’: S = symptoms (‘Have symptoms resolved?’), I = indication (‘Is there a valid indication?’), R = risks (‘Do risks outweigh benefits?’) and E = end of life (‘Is there short life expectancy limiting clinical benefit?’).(10) Each medication was assessed according to ‘S-I-R-E’ using clinical information from medical notes and available investigations. Clarification was sought from patients and the primary team when documentation was not clear. Polypharmacy was defined as the use of five or more medications, including both regular and as-required medications.(9)
The presence of symptoms was determined based on the medical documentation or clarification from the patient or medical team. Medication was deemed inappropriate if there were no active symptoms for which it was prescribed, such as medications that had been prescribed for symptomatic treatment of common transient conditions such as pain, itch, rash, giddiness and heartburn, but were continued even when the patient no longer had the symptom.
Indications for medications were assessed based on whether the medication was prescribed for a standard indication, based on guidelines from the British National Formulary.(11) Medications that were prescribed for reasons that did not meet established indications were deemed inappropriate. This also included medications that had outlived their clinical indication, such as the use of long-term proton pump inhibitors (PPIs) in patients with low risk of gastrointestinal bleeding, and supplements such as iron and folic acid that were continued despite replete levels.
Risks of medications were assessed individually based on relevant patient characteristics, such as premorbid function and mobility, presence of cognitive impairment, and patient-specific and general risks, then weighed against the potential perceived benefits for the patient. In deciding if a particular medication had an overall net risk, we also considered the current evidence and its limitations in terms of time to benefit and the study populations that had been included in trials. We also incorporated guidance from the Beers Criteria for Potentially Inappropriate Medication Use in Older Adults,(6) and from Holmes et al, who published a consensus list of medications with questionable benefit for patients with advanced dementia.(12) For example, the use of a statin may be considered inappropriate in a patient with severe dementia and a zero Barthel Index score and AMT score. Advice from physicians involved in the study was sought in cases where the perceived benefits were questionable.
Patients who were judged to be near end of life, as defined by the likelihood of having a life expectancy of less than a year as determined by the treating physician, had more rigorous medication reviews, particularly regarding medications used to reduce long-term morbidity and mortality. An example would be antihypertensives and statin therapy in a patient with advanced life-limiting diseases such as severe dementia or advanced malignancy.
Basic descriptive statistics were used for baseline demographics as well as prevalence and types of polypharmacy, and inappropriate medication use. Medications were classified into drug classes according to the British National Formulary 70.(11) Fisher’s exact test at a = 0.05 was used to determine the association between polypharmacy and the presence of inappropriate medications. Data was entered into Microsoft Excel 2010 (Microsoft Corporation, Redmond, WA, USA) spreadsheets and analysed using PASW Statistics version 18.0 (SPSS Inc, Chicago, IL, USA).
RESULTS
A total of 243 patients were included in our study. They had a median age of 85.0 ± 6.7 years. The majority of our sample population were Chinese (79.4%) and female (64.6%). The median AMT and Barthel scores of the cohort were 4.0 ± 3.6 and 13.5 ± 6.9, respectively. Most patients were community dwellers (90.1%) and cared for by family members (41.2%) or domestic helpers (33.3%). 37 (15.2%) patients were capable of independent care. A total of 2,616 medications were ordered, with a mean number of 10.8 ± 4.1 medications per patient (
Table I
Demographic and clinical characteristics of the patients (n = 243).
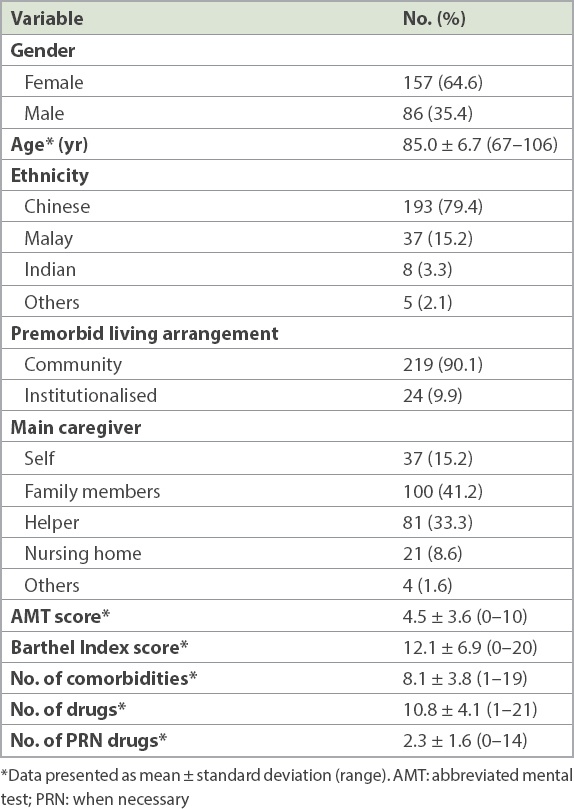
A total of 226 (93.0%) patients had been prescribed ≥ 5 medications and hence had polypharmacy. Out of the 243 enrolled patients, 67 (27.6%) had at least one inappropriate medication ordered during their inpatient stay (
Table II
Participants with polypharmacy and inappropriate drug use (n = 243).
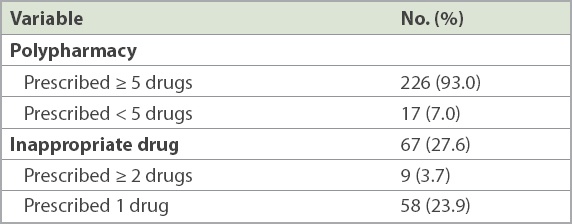
Table III
Drug classes associated with inappropriate drug use (n = 82).
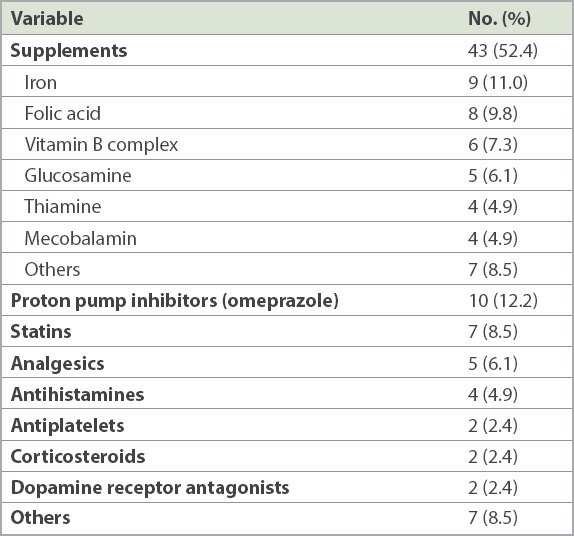
Omeprazole was the most prescribed inappropriate medication (12.2%), followed by iron (11.0%) and folic acid (9.8%) supplements (
Table IV
Reasons for inappropriate use of various drug classes (n = 82).
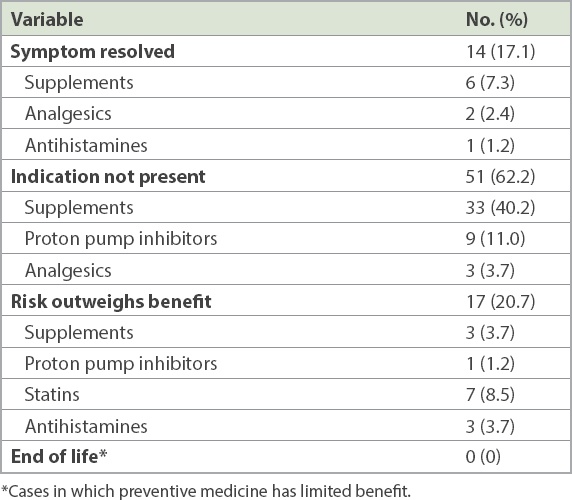
The primary team accepted pharmacist recommendations for discontinuation of 27 out of the 82 medications that were identified as inappropriate. Recommendations that were most frequently accepted involved medications used for the symptomatic treatment of symptoms that were no longer active (78.6%, 11 out of 14 drugs), followed by drugs with no indications (23.5%, 12 out of 51 drugs) and drugs with net risks (23.5%, four out of 17 drugs).
DISCUSSION
The prevalence of polypharmacy in our study population was 93.0%, comparable to rates in other studies.(13,14) Sehgal et al and Nobili et al reported a lower polypharmacy prevalence of 51.9% and 75.4%, respectively, but both studies defined polypharmacy according to the point of admission rather than during hospitalisation.(15,16) The high frequency of polypharmacy could be due to the frailty and advanced age of our inpatient geriatric cohort. We had a higher age cut-off of ≥ 80 years for patients admitted from the emergency department to our geriatric units, who formed the majority of patients in our wards. In addition, medication data was collected at any time after the first geriatrician review, and hence it was likely that the new medications to treat acute illnesses contributed to the higher frequency of polypharmacy. This finding was similar to reports from other studies that hospitalisation was associated with a higher prevalence of polypharmacy.(14,16) Inappropriate medications were prescribed in 27.6% of our study population, in keeping with studies done in other inpatient elderly cohorts.(13,17)
Supplements, such as iron, folic acid and vitamin B, were the most commonly prescribed class of drugs for which there was no valid indication or active symptoms. This high prevalence may be attributed to cultural and systemic factors. Prescription orders are often made without documenting the indication of a specific medication. This gap in information becomes a systemic problem, which is perpetuated by an electronic prescription system that allows repeat prescriptions to be carried forward with a simple click of the mouse. Hence, it is not uncommon for drugs to be repeated without a known indication. In addition, many physicians do not practise routine medication review when represcribing medications and often regard supplements as generally benign drugs. While supplements may seem harmless as compared to other chronic disease medications, many can cause gastrointestinal side effects such as constipation. Moreover, they add to a patient’s pill burden, which can affect overall medication compliance and quality of life.
PPIs contributed to 12.2% of the total inappropriate drug use due to no valid indication. This is a lower proportion than in other studies, which reported a prevalence of 22%–70%.(18-20) The high prevalence in these studies has been attributed to unlicensed indications and the lack of regular review to ensure continued appropriateness.(18,21,22) The prevalence of inappropriate PPI use in our data was also, unexpectedly, much lower than the reported prevalence of 43.2% in Singapore hospitals.(19) This could be explained by the increasing awareness among physicians about the association between long-term PPI use and Clostridium difficile infections, pneumonia and fractures.(20,23) An Ontario workgroup has developed an algorithm to aid clinicians to systematically assess the appropriateness of the prescription of PPIs, and the Department of Pharmacy, Changi General Hospital, Singapore, has also developed a similar hospital deprescribing guideline for PPIs.(23)
In our study, the inappropriate prescriptions of statins (8.5%) were for patients who were deemed to be physically and cognitively frail. Most were nursing home residents who were completely dependent on others for basic activities of daily living, had advanced dementia and were unlikely to enjoy the long-term preventative benefits of statins. The need to balance the risks and benefits of statin therapy in the elderly can be challenging, as this age group has the highest risk for cardiovascular diseases.(24-26) Physicians may not discontinue statins without a compelling reason, such as the occurrence of statin-induced myopathy. However, it is also important to be cognisant that the net benefits of statins may be attenuated in patients who are severely frail and that their life expectancy may be shorter than the time to benefit. In one trial, the deprescription of statins in the last year of life was not found to be associated with increased mortality and cardiovascular outcomes, but with improved quality of life.(27)
In a recent study of hospitalised elderly in Australia, the use of a deprescribing algorithm brought about a reduction of at least two inappropriate regular medicines in 84% of the 50 subjects included.(28) However, the use of deprescribing tools has been limited due to various reasons, including lack of time in busy healthcare settings, clinicians’ under-appreciation of the consequences of inappropriate prescribing, and lack of training and guidance in deprescribing.(29) Therefore, it is imperative to develop a simple and practical approach to assessment of the appropriateness of medications, to guide the deprescribing process. The ‘S-I-R-E’ mnemonic can be an easy-to-use tool to remind physicians to proactively assess medication appropriateness. This can be supplemented by educating junior and senior clinicians about commonly prescribed inappropriate drugs in the elderly, and the supporting evidence and literature.
Our study had several limitations. First, the use of a cross-sectional, observational design inevitably limits the ability to capture prescriptions that may change with time: a different prevalence or prescribing pattern may have been recorded prior to or following education on appropriate prescribing, which is provided by pharmacists as part of their routine medication review. A fixed time point for assessing appropriateness, such as prior to discharge, may have been more useful; however, there may be insufficient time for the necessary intervention and monitoring once any inappropriateness of medication has been ascertained. Secondly, the prescribing pattern seen in our study may not be reflective of the overall geriatric population cohort in the hospital, due to the higher age cut-off for admission to our geriatric units. It would be prudent to compare data in other medical units to ascertain any differences in inappropriate prescribing. Thirdly, there may be inter-rater variations between pharmacists when assessing for appropriateness of medication. However, this was minimised by having regular consultations with the physicians involved in the study whenever there was any clinical doubt of appropriateness. Lastly, we were unable to determine if inappropriate prescribing was associated with increased harm to our population, as our study was not designed to evaluate the association between inappropriate prescribing and frequency of adverse drug reactions during hospitalisation.
In conclusion, there is little doubt that inappropriate prescribing and polypharmacy in the hospitalised elderly are prevalent and can be reduced. Medication review and active deprescribing of inappropriate medications should be part of routine management for elderly patients. We propose the use of the ‘S-I-R-E’ mnemonic during medication review, especially in busy settings, to remind physicians to assess for medication appropriateness and deprescribe as appropriate.(10) Further research is still required to explore the effectiveness of deprescribing tools in reducing the negative effects of inappropriate prescribing and polypharmacy.


