Abstract
INTRODUCTION
Colorectal cancer (CRC) was the third most commonly diagnosed cancer worldwide in 2008 (1.23 million cases, 9.7%). CRC screening was shown to be effective in reducing 70% of CRC mortality. However, the screening rate for CRC remains poor.
METHODS
A cross-sectional survey was conducted among primary care physicians (PCPs) in public primary care clinics in Kuala Lumpur, Malaysia. A 30-item self-administered questionnaire was used to assess the knowledge and practice of CRC screening.
RESULTS
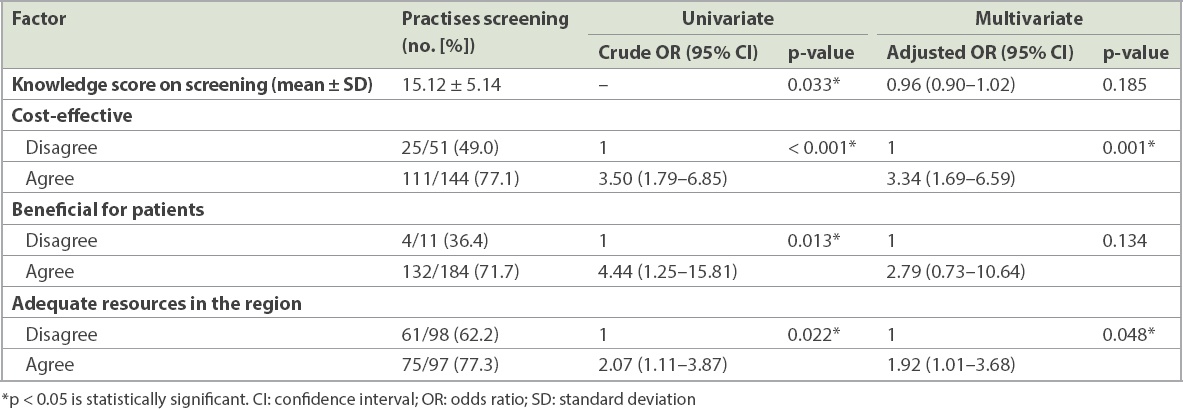
The response rate was 86.4% (n = 197/228). Less than half (39.1%) of the respondents answered correctly for all risk stratification scenarios. Mean knowledge score on CRC screening modalities was 48.7% ± 17.7%. The knowledge score was positively associated with having postgraduate educational qualification and usage of screening guidelines. Overall, 69.9% of PCPs reported that they practised screening. However, of these, only 25.9% of PCPs screened over 50% of all eligible patients. PCPs who agreed that screening was cost-effective (odds ratio [OR] 3.34, 95% confidence interval [CI] 1.69‒6.59) and those who agreed that they had adequate resources in their locality (OR 1.92, 95% CI 1.01‒3.68) were more likely to practise screening. Knowledge score was not associated with the practice of screening (p = 0.185).
CONCLUSION
Knowledge and practice of CRC screening was inadequate among PCPs. Knowledge of screening did not translate into its practice. PCPs’ perceptions about cost-effectiveness of screening and adequate resources were important determinants of the practice of screening.
INTRODUCTION
Over a million cases of colorectal cancer (CRC) are diagnosed annually worldwide. In terms of mortality, CRC causes more than 600,000 deaths annually. It was the third most commonly diagnosed cancer worldwide in 2008 (1.23 million cases, 9.7%), and is the second most common cancer in women and the third most common cancer in men.(1) In Malaysia, CRC is the second most common cancer, with an incidence of 13.2%, and the most common cancer among men (16.3%).(2)
CRC screening has been shown to be effective in reducing associated mortality and incidence. This is because the pathogenesis of CRC is well established. It has long been known that CRC arises from premalignant adenomatous polyps.(3) Therefore, the strategy for CRC screening is to detect early adenomatous polyps or early-stage adenocarcinoma for removal.(4) A large prospective study carried out in Japan reported a 70% reduction in CRC mortality with screening.(5) Similarly in the United States, there has been a steadily declining rate of CRC incidence due to extensive screening.(6)
Yet uptake of CRC screening remains low. A large multicentre study in Asia reported that countries in Southeast Asia had very low rates of CRC screening – Singapore (20.3%), Brunei (8.8%), Indonesia (1.5%) and Thailand (18.8%).(7) In this study, only 1.2% of Malaysian respondents were found to have been screened.(7) In the United States, the screening rate was about 50%.(8)
Malaysia practises a two-tier healthcare system, which includes the government and private sectors. In the government healthcare system, which is the focus of this study, CRC screening tests are available in hospitals and selected health clinics. Patients can be screened at minimal cost using the faecal occult blood test (FOBT) in health clinics and colonoscopy in tertiary hospitals. However, currently, there is no population-based CRC screening programme being implemented in Malaysia.(9,10)
Primary care physicians (PCPs) play a very important role in CRC screening. Many studies have shown that PCP recommendation is a strong predictor of screening participation by patients.(7,11,12) A local study reported that 84.6% of patients would undergo screening if advised to do so by their primary care providers.(12) The aim of this study was to improve screening rates for CRC by assessing the current level of knowledge and practice of CRC screening among PCPs in Malaysia.
METHODS
This was a cross-sectional questionnaire survey conducted from July to August 2014. All eligible PCPs working in public clinics in Kuala Lumpur, Malaysia, during the study period were invited to participate in the study. There were 13 government clinics and two academic primary care clinics in this geographical location.
The questionnaire for the study was adapted from three studies – two from the United States and another from Canada.(13-15) Some items were adapted from recommendations by the Singapore Ministry of Health Clinical Practice Guidelines for Cancer Screening 2010.(16) The questionnaire was divided into five sections (Sections A–E): demographic data and place of practice; risk stratification of CRC; knowledge on CRC screening modalities; practice of CRC screening; and perceived barriers and facilitators influencing decision-making on CRC screening (Appendix).
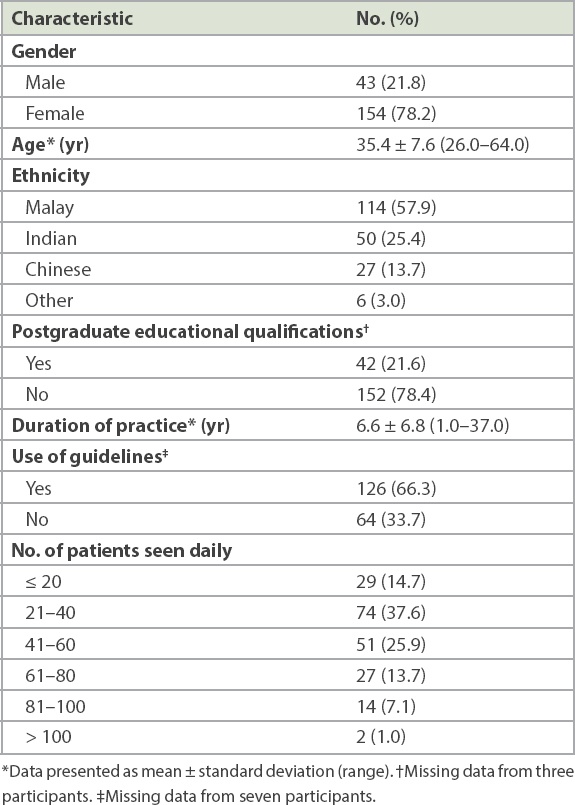
Section A pertained to the participant’s gender, age, ethnicity, postgraduate educational qualifications, years of practice, daily patient attendance, place of practice and usage of guidelines.
Section B was regarding risk stratification of CRC. Four clinical scenarios were given to the participant to risk stratify the patient into each scenario: scenario 1 (patient, aged 62 years, with history of inflammatory bowel disease); Scenario 2 (patient, aged 60 years, with family history of CRC in two relatives [one at age 45 years, another at age 50 years]); Scenario 3 (female, aged 60 years, no family history of CRC); and Scenario 4 (male, aged 60 years, no family history of CRC). For each scenario, participants were asked to categorise the patient as ‘low risk’, ‘average risk’, ‘high risk’ or ‘don’t know’.
Section C was regarding knowledge on CRC screening modalities. There were two parts to this section. The first part assessed the participant’s knowledge on CRC screening for average-risk patients. Areas that were assessed were recommended test, starting age and recommended frequency. The screening tests assessed were FOBT, colonoscopy, double-contrast barium enema, flexible sigmoidoscopy and serum carcinoembryonic antigen. The second part assessed the participant’s knowledge on CRC screening for high-risk patients. Participants were assessed on which conditions needed to be screened, recommended age at which to start offering screening, recommended frequency and recommended test for each condition. Patients were assessed for a family history of familial adenomatous polyposis, family history of hereditary non-polyposis colorectal cancer and history of inflammatory bowel disease.
Section D was regarding the practice of CRC screening. Participants were asked if they practised CRC screening, as well as the starting age for screening, proportion of eligible patients screened and screening test used in practice.
Section E concerned perceived barriers and facilitators influencing the decision for CRC screening. Participants were given six statements pertaining to CRC screening. The response was recorded in a Likert scale, ranging from ‘strongly agree’ and ‘agree’ to ‘disagree’, ‘strongly disagree’ and ‘not sure’.
The questionnaire underwent face and content validations by an expert panel comprising two primary care specialists – a gastroenterologist and a colorectal cancer surgeon. The draft study was piloted on ten PCPs from an academic primary care clinic, following which minor changes were made to produce the final version of the questionnaire. The estimated time needed to complete the questionnaire was 10–15 minutes.
All clinics were visited by the principal investigator at the start of the study. A medical officer in charge of the clinic coordinated the data collection. This representative was briefed in detail and face-to-face about the study. Informed consent was collected by the representative, who also distributed and collected the research forms (i.e. participant information sheet, consent form and questionnaire). The questionnaires and consent forms were then returned to the researcher.
Data was analysed using IBM SPSS Statistics version 20.0 (IBM Corp, Armonk, NY, USA). Data was summarised as mean and percentage values. The knowledge score was calculated based on responses received in Section C. For calculation of the knowledge score, a score of 1 was awarded for every correct response and no score was given for wrong or empty response. There was no negative marking. In the column for screening test for high-risk patients, no score was awarded if one of the choices ticked was a wrong response. This was to avoid any bias, as participants were given the option to tick multiple choices for this column. The minimum score possible was 0 and maximum score was 30. Logistic regression analysis using forward elimination was used to identify predictors of the practice of CRC screening.
This study was approved by the University Malaya Medical Centre Ethics Committee (reference no. 1045.8) and Ministry of Health Malaysia Medical Research and Ethics Committee (reference no. NMRR-13-1589-14913).
RESULTS
The response rate was 86.4% (197/228 participants). The characteristics of the participants are shown in
Table I
Characteristics of participants (n = 197).
In Section B, only 39.1% of PCPs risk stratified all four scenarios correctly, followed by 22.3% who risk stratified three scenarios correctly. Another 25.9% of PCPs risk stratified two scenarios correctly, while 11.7% risk stratified only one scenario correctly, and 2 (1.0%) participants responded wrongly to all four scenarios. Of these, one PCP answered ‘don’t know’ for all four scenarios. Among the scenarios that were correctly stratified, Scenario 3 received the lowest number of correct responses (47.0%), followed by Scenario 4 (70.0%) and Scenario 1 (75.0%). Scenario 2 received the highest rate of correct responses (95.0%).
Concerning the knowledge of PCPs on CRC screening modalities, for average-risk patients, most (90.4%) PCPs correctly recommended FOBT as a screening modality. Double-contrast barium enema and serum carcinoembryonic antigen were recommended by 17.9% and 18.8% of PCPs, respectively, even though these tests are not recommended in the guidelines. A majority could correctly identify the starting age for screening.
For high-risk patients, a high percentage of PCPs correctly recommended screening for patients with family history of familial adenomatous polyposis (95.4%), hereditary non-polyposis colorectal cancer (92.7%) and patients with inflammatory bowel disease (88.6%). Almost half of the PCPs recommended FOBT as a screening test although it is not recommended for any of the three conditions mentioned above.
As for the frequency of screening, only about 60.0% of PCPs gave the right recommendation for familial adenomatous polyposis and hereditary non-polyposis colorectal cancer. For inflammatory bowel disease, only 48.8% of PCPs recommended screening according to guidelines. However, a majority could not identify correctly the age at which to start offering screening.
In terms of CRC screening modalities, PCPs had a mean knowledge score of 14.60 ± 5.32 (48.7% ± 17.7%) out of a maximum possible score of 30 (100.0%). The lowest score recorded was 0 (0%) and the highest score was 28 (93.3%). When a general linear model was used to determine independent predictors that significantly affect the knowledge score, we found that usage of guidelines (p = 0.02) and postgraduate educational qualifications (p < 0.001) were significantly associated with the knowledge score of PCPs.
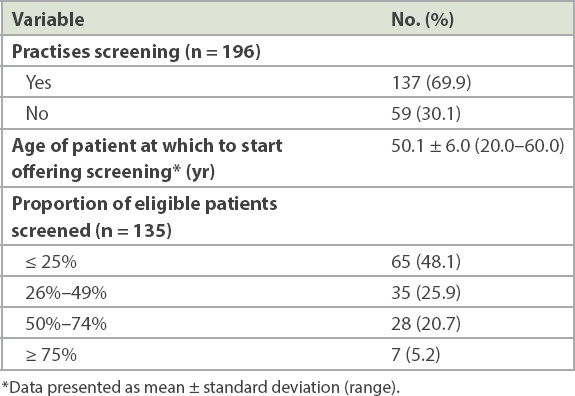
Overall, 69.9% of PCPs reported that they practised CRC screening for average-risk patients (
Table II
Practices with respect to colorectal cancer screening.
With regard to the perceived barriers and facilitators influencing the decision on CRC screening among PCPs, most PCPs perceived that they lacked knowledge about screening tests (55.9%) and recommendations for screening were inconsistent between the various guidelines (55.4%). However, two-thirds (74%) of PCPs felt that screening was cost-effective. A majority (94.4%) of PCPs perceived that screening was beneficial for their patients.
Concerning factors influencing PCPs to screen patients for CRC, univariate logistic regression analysis showed that knowledge was significantly associated with the practice of CRC screening (
Table III
Results of logistic regression analysis of factors influencing the practice of screening.
DISCUSSION
In our study, only one-third (39.1%) of PCPs were able to correctly risk stratify patients for all four scenarios presented for CRC risk stratification. Mean knowledge score on CRC screening modalities was 48.7% ± 17.7%. Knowledge score was positively associated with having a postgraduate educational qualification and the usage of guidelines for CRC screening. Although nearly 69.9% of PCPs reported that they practised CRC screening, only 25.9% of them screened over half of all eligible patients. The commonly used CRC screening tools were FOBT and colonoscopy. Serum carcinoembryonic antigen was also used although it is not recommended as a screening test in the guidelines. PCPs who agreed that screening was cost-effective and those who had enough resources in their locality were more likely to practise CRC screening.
With only 39.1% of PCPs being able to risk stratify all four scenarios correctly, the proportion of PCPs who were able to do this in our study was much lower than that in another study, where 72.7% of participants could respond correctly to all four scenarios.(15) Accurate risk stratification is important for PCPs to be able to provide the appropriate screening recommendation. A lower percentage of PCPs being able to risk stratify correctly is an area of concern, as the likelihood of them offering wrong advice or recommendations vis-à-vis CRC screening could, accordingly, be expected to be high. This could be due to CRC screening not being widely practised by PCPs, and thus their knowledge of risk stratification was lacking.
Most PCPs were able to give the right screening recommendations for average-risk patients. For high-risk patients, most PCPs correctly recommended screening for all three categories. However, most of the recommendations given were not guideline-consistent. FOBT was recommended by almost half of all PCPs although it is not recommended in the guidelines. This is a source of concern, as it points to wastage of resources due to inappropriate screening. Generally, this reflects a lack of knowledge among PCPs on screening for high-risk patients. A study done in the United States reported that PCPs had poorer knowledge, as compared to gastroenterologists, when it came to CRC screening for high-risk patients.(17) This could be due to a lack of exposure to these patient groups among PCPs, as most of such patients tend to be diagnosed by gastroenterologists. It is also likely that follow-up screenings for these patients would have been done by gastroenterologists.
The overall knowledge score was 48.7% ± 17.7% in our study, and PCPs with postgraduate educational qualifications and those using guidelines received significantly higher scores. These results were consistent with other studies that have shown that PCPs who adhered to guidelines and had training were more likely to give guideline-consistent screening recommendations.(18-20) A study in the United States that assessed knowledge of physicians on CRC screening in high-risk populations found that being unaware of screening guidelines was associated with significantly lower knowledge scores.(19) In a study from Italy, no association was found between knowledge score and gender, age, ethnicity and years of practice of physicians.(20)
In our study, 69.9% of PCPs practised CRC screening for their patients. In comparison, in the United States, almost 99% of PCPs practised CRC screening, while in Italy, about 80% of physicians practised the same.(20,21) Overall, the rate of screening was low in our study when compared to other countries. Even though our PCPs reportedly practised screening, not many patients were actually screened, with only 25.9% of PCPs screening more than 50% of eligible patients for CRC. Another Malaysian study found that only about 21% of healthcare providers screened more than 50% of their patients with FOBT.(22) One reason for this could be a lack of concerted effort on the dissemination of best practices for CRC screening, and of providing clear screening and follow-up guidelines to primary care providers. The consensus published by the Academy of Medicine Malaysia needs to be updated and highlighted to PCPs in order to improve the practice of CRC screening in the country.(23) Another possible reason for this finding could be that PCPs had overstated their practice in our survey.
PCPs who agreed that screening was cost-effective were more likely to practise CRC screening. This outcome was consistent with a similar study in Canada, where physicians who agreed that screening was cost-effective were 3.4 times more likely to practise screening.(14) A study that looked into factors associated with the practice of screening among PCPs in Hong Kong reported that cost was a major reason for not screening (62.4%).(24) To the contrary, a study in the United States had shown that CRC screening was, in fact, cost-effective. This study found that offering screening to patients aged 50 years and above would result in a gain of USD 11,900 per year of life.(25) Therefore, educating PCPs on the cost-effectiveness of CRC screening may increase the practice of screening. Nevertheless, the cost of screening may be variable in different countries and in different health systems. Therefore, this matter warrants consideration prior to educating PCPs on the cost-effectiveness of CRC screening in various countries.
PCPs who agreed that there were adequate resources for screening in their locality were more likely to practise CRC screening. In Malaysia, the unavailability of screening kits was cited as a major barrier to screening.(22) Currently, only selected government clinics have laboratory facilities and not all have FOBT kits. Only the larger hospitals and clinics selected for the CRC screening programme have screening tests at hand. Colonoscopy is only available in hospitals where there are gastroenterologists and surgeons who can perform the procedure. Most of the time, these practitioners are overwhelmed by the many referrals received and waiting times can be long, ranging from six months to one year. Therefore, increasing the resources available for screening is necessary to encourage more PCPs to practise CRC screening.
Another outcome observed in the present study was that the knowledge score of PCPs was not associated with the practice of screening. No significant association was found between knowledge score and screening practice after adjusting for confounders (p = 0.185). In fact, a study in Italy showed that 10% of physicians who had very high knowledge score did not practise screening.(20) Knowledge is important for ensuring that the screening done is consistent with guideline recommendations. However, just having knowledge may not be sufficient. A positive attitude towards screening is an important factor in the practice of screening, as reported by a study from Hong Kong, which found that PCPs with a positive attitude towards screening were two times more likely to practise screening.(24)
Our study was not without strengths and limitations. One of the strengths was the good response rate achieved among PCPs. The outcomes from this study have provided insights on the knowledge and current practices of CRC screening among PCPs in Malaysia. However, as this was a cross-sectional study, it only measured responses of PCPs at a certain point in time. This may not accurately reflect their actual medical practice. Another limitation was that this was a self-reporting survey and no comparisons were done to ascertain the actual practices of the PCPs. These results could thus be subject to recall bias and overestimation/underestimation. Also, our outcome only reflected PCPs practising in an urban setting. Further studies would be needed to determine the knowledge and practices of CRC screening among PCPs in rural areas.
Our study revealed that fewer than 40% of PCPs were able to risk stratify CRC risk correctly. Mean knowledge score on CRC screening was about 50% and was positively associated with having a postgraduate educational qualification and the usage of guidelines. The practice of screening was low in our study when compared to other countries. Educating PCPs with the latest evidence and guidelines will ensure that the right recommendations for screening are provided to patients. However, having knowledge does not necessarily translate into practice. Agreement that screening was cost-effective and having adequate resources for screening were shown to have a positive association with the practice of screening. Therefore, measures to further educate PCPs regarding the cost-effectiveness of screening and the provision of adequate resources need to be undertaken to further increase its practice in Malaysia. These measures can also be explored further when planning population-based CRC screening in the future.
APPENDIX
ACKNOWLEDGEMENTS
We would like to thank the Director General of Health, Malaysia, for his permission to publish this article and the staff at all the health clinics in Kuala Lumpur, Malaysia, for their support and participation in the study. This research was funded by the Postgraduate Research Fund (PPP) of University of Malaya, Malaysia (no. 2143/2014A).


