Abstract
The prevalence of non-alcoholic fatty liver disease (NAFLD) is increasing rapidly with the obesity and diabetes mellitus epidemics. It is rapidly becoming the most common cause of liver disease worldwide. NAFLD can progress to serious complications such as cirrhosis, hepatocellular carcinoma and death. Therefore, it is important to recognise this condition so that early intervention can be implemented. Lifestyle modifications and strict control of metabolic risk factors are the mainstay of treatment. As disease progression is slow in the majority of NAFLD patients, most can be managed well by primary care physicians. NAFLD patients with advanced liver fibrosis should be referred to specialist care for further assessment.
Mr Tan, a 48-year-old executive with diabetes mellitus, hypertension and dyslipidaemia, has been seeing you for the management of his chronic diseases for the past three years. His liver enzymes were found to be slightly elevated on routine monitoring of his chronic conditions. He rarely exercises due to his busy work schedule and has a body mass index of 29. Hepatitis B serology was negative. Ultrasonography of the liver reported fatty liver with no other significant finding.
HOW COMMON IS THIS IN MY PRACTICE?
Non-alcoholic fatty liver disease (NAFLD) is rapidly becoming the most common cause of liver disease worldwide. The prevalence of NAFLD is estimated to range from 5%–18% in Asians(1) and 20%–30% in the Western population.(2) Overall, its pooled global prevalence has been reported to be 24.4%.(3) However, in westernised Asian populations, including in Singapore, its prevalence appears to be higher; prevalence rates as high as 40% have been reported.(4,5) Notably, NAFLD does not only affect the obese population. It has been demonstrated that nearly 20% of non-obese ‘healthy’ Asians have radiological evidence of NAFLD.(6) It is of significant concern that the incidence and prevalence of NAFLD is expected to increase rapidly with the obesity and diabetes mellitus epidemics.(7)
WHAT IS NON-ALCOHOLIC FATTY LIVER DISEASE?
NAFLD is defined as an accumulation of more than 5% fat within the liver that is not attributable to alcohol or drugs. It is the hepatic manifestation of metabolic syndrome, which encompasses a spectrum of liver diseases ranging from simple steatosis, also known as non-alcoholic fatty liver (NAFL), to non-alcoholic steatohepatitis (NASH). NASH is associated with inflammatory cell infiltrate, which leads to injury and apoptosis of hepatocytes.(8)
Natural history of NAFLD
There remains considerable uncertainty about the natural history and prognosis of NAFLD. Despite its high prevalence, only a minority of NAFLD cases progress to cirrhosis and its associated complications.(9) Nevertheless, some subsets of NAFLD (10%–25%) do progress to serious complications such as liver failure, development of hepatocellular carcinoma and death. The most important predictor of long-term outcome in patients with NAFLD is the presence or absence of advanced liver fibrosis.(10) It is believed that liver fibrosis progression is less common in patients with simple steatosis and more frequent in NASH patients. Meta-analysis found that the average time needed for NAFLD patients with simple steatosis with no fibrosis to progress by one fibrosis stage was 14.3 years; if NASH was present, the time taken to progress by one fibrosis stage was shortened to 7.1 years.(11) However, the rate of fibrosis progression is not uniform in all NAFLD cases, as rapid progression can occur in some patients, including those with simple steatosis.
How to diagnose NAFLD
Ultrasonography, being safe and inexpensive, is an acceptable first-line screening procedure for NAFLD in clinical practice. It is important to ensure that other aetiologies of liver fat deposition, such as alcohol ingestion, are excluded before the diagnosis of NAFLD is made (
Table I
Common secondary causes of fatty liver.

In the tertiary care setting, liver biopsy is performed for high-risk NAFLD patients (i.e. NAFLD patients with advanced liver fibrosis). Liver biopsy remains the gold standard for direct diagnosis of NASH as well as evaluation of the degree of inflammation and stage of fibrosis. Although not all NAFLD patients should undergo liver biopsy, it should be considered for patients suspected to have advanced liver fibrosis and those with persistently elevated transaminase levels. Liver biopsy can stage their disease accurately and rule out other pathologies that may have caused the abnormally elevated transaminase levels.
WHAT CAN I DO IN MY PRACTICE?
Uncomplicated NAFL can be routinely managed and followed up in the primary healthcare setting. All NAFLD patients should undergo interventions aimed at promoting healthier lifestyles and strict control of metabolic risk factors.(12) Lifestyle modifications including weight loss, dietary changes and physical exercise are the first-line treatment for NAFLD patients. Weight loss of at least 3%–5% of body weight can improve steatosis and up to 10% can improve necroinflammation. To achieve this, exercise and a hypocaloric diet of about 30 kcal/kg (126 kJ/kg) to promote weight loss should be encouraged.(13) The primary care physician should provide dietary advice encouraging higher consumption of fibre and antioxidant-rich fruits and vegetables as well as lower consumption of carbohydrate and saturated fat. Patients should avoid fructose-enriched foods such as soft drinks, while coffee (ideally, without sugar) may be considered as an adjunct in NAFLD as it is associated with improved outcomes.(14) Exercise prescription is a crucial part of NAFLD management. It is recommended that NAFLD patients commit to at least 150 minutes of moderate-to-vigorous aerobic physical activities per week and avoid a sedentary lifestyle. The SMART (specific, measurable, achievable, realistic and time-framed) goal method is one practical tool that doctors can use to help patients to achieve their exercise targets.(15)
Aggressive control of metabolic risk factors such as dyslipidaemia, hypertension and diabetes mellitus in NAFLD patients is prudent, as ischaemic heart disease is a major cause of mortality in these patients. Optimising diabetes mellitus control with insulin sensitisers such as metformin or glitazones,(16) controlling hypertension with angiotensin-converting enzyme inhibitors or angiotensin receptor blockers,(17) and treating dyslipidaemia with statins and ezetimibe(18) have been shown to benefit NAFLD patients. Cessation of smoking is advocated, as it is associated with more severe NASH.(19)
Pharmacological therapy options for NAFLD remain limited. Although vitamin E, pioglitazone and pentoxifylline have been used with varying effectiveness, convincing evidence showing the long-term benefits of these pharmacological agents is still lacking. On the contrary, high-dose vitamin E supplementation has been shown to increase the risk of prostate cancer.(20) Therefore, it is generally recommended that only patients with biopsy-proven NASH should undergo pharmacological therapy with finite duration. Patients who require pharmacological intervention should ideally be managed in tertiary care.
WHEN SHOULD I REFER TO A SPECIALIST?
In the majority of NAFLD cases, progression of fibrosis stages is generally slow. Hence, primary care physicians can manage most of these patients by implementing lifestyle modifications and optimising metabolic risk factors, as described above. However, select groups of NAFLD patients, such as those with advanced liver fibrosis or cirrhosis, should be referred to specialist care. These high-risk patients may require further investigation, such as liver biopsy, to stage their disease and exclude other chronic liver diseases. They may also benefit from pharmacotherapy if NASH is confirmed on liver biopsy.
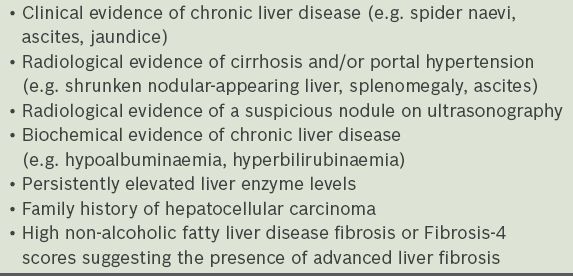
The most important predictor for poor outcome in NAFLD patients is the presence of advanced liver fibrosis. Therefore, high-risk NAFLD patients can often be identified by assessing their fibrosis stage using noninvasive methods, such as transient elastography (FibroScan®) or fibrosis scoring systems. The online NAFLD fibrosis score calculator(21) is a reliable risk stratification tool that uses age, the presence of hyperglycaemia, body mass index, platelet count, albumin level, and aspartate aminotransferase (AST)/alanine transaminase (ALT) ratio to predict advanced liver fibrosis in NAFLD patients. A score of < –1.455 would exclude advanced liver fibrosis with high accuracy and a negative predictive value of 93%.(21) The Fibrosis-4 (FIB-4) score is another simple score comprising of four easy-to-obtain variables: age, platelet count, ALT and AST. A FIB-4 index of < 1.45 could reliably exclude advanced liver fibrosis.(22) In the primary care setting, either the NAFLD fibrosis or FIB-4 score can be calculated; NAFLD patients with high FIB-4 or NAFLD fibrosis scores can be referred to specialist care for further evaluation, while those without high fibrosis scores should be followed up every two years to reassess their fibrosis status. In addition, NAFLD patients with evidence of cirrhosis, such as jaundice, coagulopathy or ascites, should also be referred to a specialist unit for further assessment.
Table II
Red flags that should prompt a referral to specialist care.
Mr Tan returned to your clinic about three months later for a follow-up appointment. He had taken your advice and begun exercising five days a week for 30 minutes each time. He had also changed his diet, decreasing his carbohydrate intake and avoiding foods containing fructose. His transaminase levels showed significant improvement since the last visit and his Fibrosis-4 score was < –1.455. You encouraged him to keep up the good work and checked that his metabolic risk factors such as diabetes mellitus, dyslipidaemia and hypertension were well-controlled.
TAKE HOME MESSAGES
-
NAFLD is rapidly becoming the most common cause of liver disease, increasing in prevalence with the obesity and diabetes mellitus epidemics.
-
Recognising NAFLD is important, as this condition can lead to cirrhosis and its associated complications.
-
Lifestyle modifications including weight loss, dietary changes and physical exercise are the mainstay of treatment.
-
Metabolic risk factors should be aggressively treated in NAFLD.
-
NAFLD patients with advanced liver fibrosis or cirrhosis should be referred to specialist care.
SMJ-57-371.pdf


