Abstract
INTRODUCTION
Narrow-band imaging with magnification endoscopy (NBI-ME) allows real-time visual assessment of the mucosal surface and vasculature of the gastrointestinal tract. This study aimed to determine the performance of NBI-ME combined with the water immersion technique (NBI-ME-WIT) in detecting villous atrophy.
METHODS
All patients who underwent gastroscopy were included. The duodenum was further examined with NBI-ME-WIT only after examination with white light endoscopy did not reveal a cause of anaemia or dyspepsia. Targeted biopsies were taken of visualised areas. NBI-ME-WIT findings were compared with the final histopathological analysis. We calculated the sensitivity (Sn), specificity (Sp), positive predictive value (PPV) and negative predictive value (NPV) of NBI-ME-WIT in detecting villous atrophy and the hypothetical cost saved by using a biopsy-avoiding approach.
RESULTS
124 patients (83 female) with a mean age of 46 (range 18–82) years were included. The most common indication for gastroscopy was abdominal pain (39%), followed by anaemia (35%), chronic diarrhoea/altered bowel habits (19%) and dyspepsia (6%). NBI-ME-WIT was able to detect all nine patients with villous atrophy – eight patchy and one total villous atrophy. The Sn, Sp, PPV and NPV of NBI-ME-WIT in detecting villous atrophy were 100.0%, 99.1%, 90.0% and 100.0%, respectively. Taking into account the cost of biopsy forceps (AUD 17) and pathology (AUD 140), this biopsy-avoidance strategy could have saved AUD 18,055 in these patients.
CONCLUSIONS
NBI-ME-WIT is a specific and sensitive tool to recognise and accurately diagnose villous atrophy. Biopsies can be avoided in patients with normal-sized villi, which may decrease the overall cost of the procedure.
INTRODUCTION
Coeliac disease (CD) is an immune-mediated condition of the small intestine that is characterised by mucosal inflammation, crypt hyperplasia and villous atrophy arising from exposure to dietary gluten in predisposed individuals, which resolves with avoidance of gluten in the diet. The prevalence of CD has been increasing in recent decades.(1,2) A study from Australia estimated that the disease affects 1.2%–1.9% of the population.(3) Recognising CD is challenging and at times results in a delay of diagnosis.(4,5) CD is usually suspected when symptomatology, such as iron-deficiency anaemia and chronic diarrhoea, is associated with malabsorption of nutrients. Following clinical suspicion, serology is generally performed to look for specific antibodies, including tissue transglutaminase immunoglobin A (IgA), anti-endomysial IgA and anti-deamidated gliadin peptide IgA antibodies. Although the antibody tests have high sensitivity (Sn) and specificity (Sp) for CD, a definitive diagnosis relies on histological examination of biopsies from the duodenum.(6)
Normal duodenal mucosa has finger-like villi and a regular network of capillaries. Reduced duodenal folds, scalloping of fold margins, a mosaic pattern of mucosa and grooves in the mucosa are the conventional signs seen on white light endoscopy (WLE) in patients with CD.(7-11) Histologically, villous atrophy is graded using the Marsh classification, which utilises the number of intraepithelial lymphocytes (IEL) as well as the presence of crypt hyperplasia and villus atrophy. These changes are denoted as Grades I, II and III (IIIa: mild atrophy; IIIb: marked atrophy; IIIc: total atrophy) reflecting the increasing severity of mucosal changes.(12) With WLE, the Sn of diagnosing mild or patchy villous atrophy is an abysmal 59%.(13) Narrow-band imaging (NBI), however, has the ability to differentiate normal and abnormal mucosal patterns in the oesophagus and stomach as well as in the small intestine. The evidence so far has shown NBI to be superior to conventional WLE in the evaluation of duodenal villi in CD and suspected malabsorption.(14,15) The benefits of NBI include facilitating targeted biopsy of abnormal mucosa, thereby reducing costs and complications, which may in turn mitigate the need to perform random biopsies.
There are only a few studies that examined the diagnostic ability of NBI with magnification endoscopy (NBI-ME) to detect villous changes specifically in CD. In one study of 44 patients by De Luca et al, NBI-ME detected villous atrophy in all 17 patients with established CD. In contrast, WLE was only able to identify seven of the patients. Hence, the Sn of NBI-ME was much better than that of WLE (100% vs. 41%).(15) An earlier study from Australia assessing villous atrophy using NBI-ME showed that this novel tool displayed Sn of 93.3% and Sp of 97.8% in patients with suspected CD.(16) These studies only enrolled patients with a high pretest probability of CD who had either malabsorption or positive serological tests.
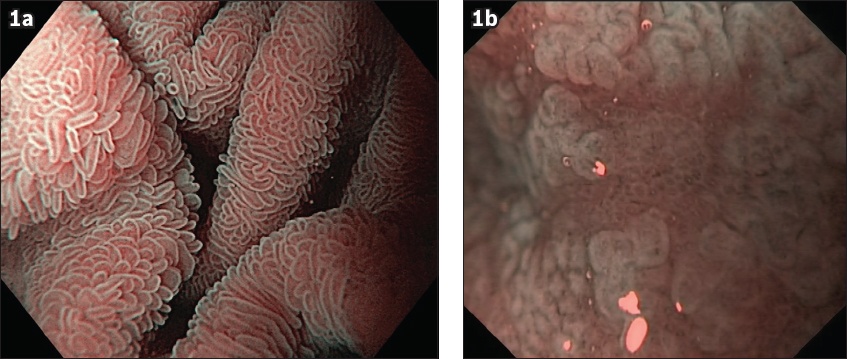
NBI-ME can be used with the water immersion technique (i.e. NBI-ME-WIT), enabling even greater enhancement of the mucosal/villi architecture (
Fig. 1
Water immersion with narrow-band imaging and magnification images show (a) normal villi (×60) and (b) partial-to-total villous atrophy (×60).
METHODS
This was a single-centre prospective study to assess the performance of NBI-ME-WIT in detecting villous atrophy. Written informed consent was obtained from each patient prior to upper GI endoscopy. All patients who underwent upper GI endoscopy between July 2008 and July 2015 were evaluated for eligibility.
Half an hour prior to the procedure, patients were given a drink of a mucolytic agent, N-acetylcysteine, and a defoaming agent, simethicone, mixed with water (total 50 mL). The procedures were performed either using a 160 Z (pre-year 2011) or a 190 HQ endoscope (Olympus Australia Pty Ltd, Melbourne, Victoria, Australia) and a high-definition television monitor (OEV181H; Olympus Australia Pty Ltd, Melbourne, Victoria, Australia). Both endoscopes had a magnification capability with a range of 60–115 times. A transparent distal attachment cap was used for all procedures.
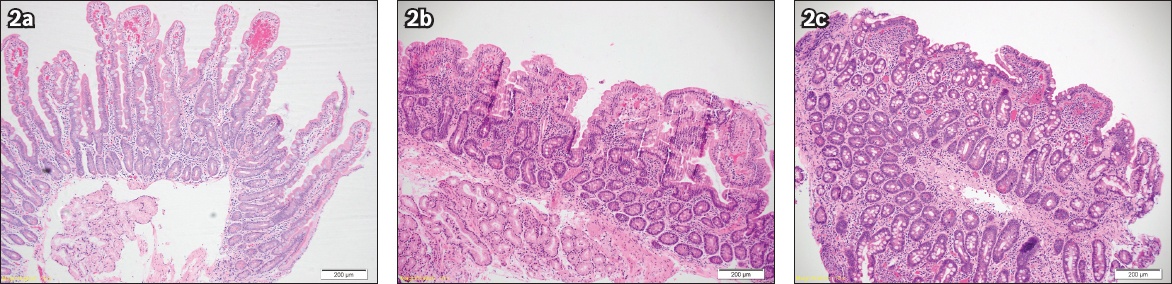
The duodenum was further examined with NBI-ME-WIT only after the examination with WLE in the oesophagus, stomach and duodenum did not reveal the cause of anaemia or dyspepsia. The duodenal lumen was filled with approximately 50 mL of water and views obtained with the endoscopic tip immersed under water. The transparent hood prevents water from escaping and ensures that a good fluid seal is obtained between the tip of the scope and the duodenum. If there was excessive peristaltic activity that interfered with obtaining optimal, crisp views, an antispasmodic agent, hyoscine-N-butylbromide (10–20 mg), was administered. Villous patterns were predicted based on a previous validated classification. Targeted duodenal biopsies were then taken irrespective of what was predicted on NBI-ME-WIT. The duodenal villous patterns observed using NBI-ME-WIT were compared with the final histopathological analysis (
Fig. 2
Histology images show (a) normal villi (Haemotoxylin & eosin [H&E], ×10); (b) partial villous atrophy (H&E, ×10); and (c) total villous atrophy (H&E, ×10).
Data was analysed to calculate the Sn, Sp, PPV and NPV of NBI-ME-WIT in detecting villous atrophy. Hypothetical cost saved by using a biopsy-avoiding approach was also calculated.
RESULTS
A total of 124 patients were evaluated. The process of patient enrolment is shown in
Table I
Baseline characteristics of the study population (n = 124).
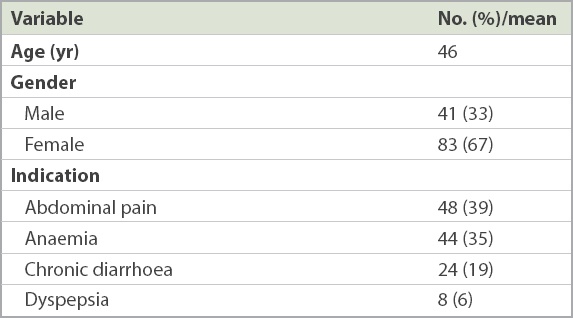
Fig. 3
Flowchart shows the study enrolment and outcome. IEL: intraepithelial lymphocytes; NBI-ME-WIT: narrow-band imaging with magnification endoscopy and water immersion technique
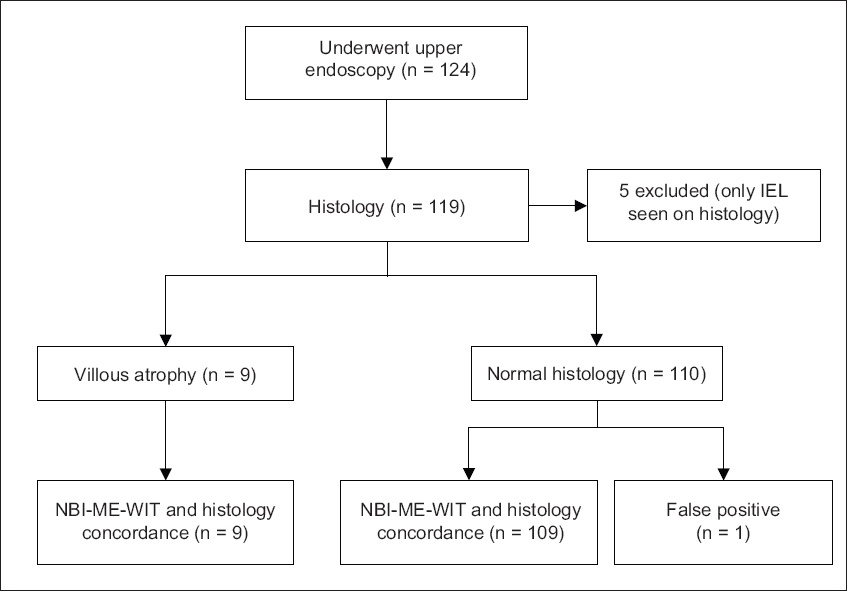
Villous atrophy was present in 9 (7%) patients; eight had patchy villous atrophy, while one had total villous atrophy. NBI-ME-WIT detected all nine patients. The Sn, Sp, PPV and NPV of NBI-ME-WIT in detecting villous atrophy were 100.0%, 99.1%, 90.0% and 100.0%, respectively. The final histological diagnosis in patients who had villous atrophy was duodenitis (n = 3, 33.3%), coeliac disease (n = 2, 22.2%), reactive changes (n = 2, 22.2%) and Crohn’s disease (n = 2, 22.2%). Raised IEL counts were found in five patients. A review of medical records and inquiry from the patients via a phone conversation revealed that none of them had been diagnosed with CD. Three were diagnosed with irritable bowel syndrome, one had spontaneous resolution of symptoms, and another patient had spontaneous partial improvement of symptoms with no diagnosis given. Importantly, none of these patients reported any symptoms of malabsorption or long-term sequelae.
Taking into account the cost of a single biopsy forceps (AUD 17) and Medicare Benefits Schedule (Australia) for pathology (AUD 140), a proposed biopsy-avoidance strategy could have saved AUD 18,055 in this cohort of patients.
DISCUSSION
Although CD prevalence has been rising over the last decades, diagnostic tools remain limited, leading to delays in diagnosis for this group of patients. Endoscopic markers using standard WLE have high Sn for diagnosing mucosal abnormalities, although it can vary from 50% to 94%.(10,18,19) This figure drops further for subclinical CD and patchy villous atrophy.(13,20-22) Image-enhanced endoscopic technologies such as NBI-ME-WIT may play a vital role in identifying abnormal changes and subsequently directing targeted biopsies.
NBI is a novel endoscopic imaging technique that enables characterisation of subtle mucosal changes. In NBI, the bandwidths of the blue and green filters used are narrowed, forming a narrowed wavelength of light. This light is maximally absorbed by haemoglobin, enhancing the visualisation of capillary microvasculature. It also has a relatively more superficial penetration compared to conventional white light, thus accentuating surface microstructure.
In this study, we showed that NBI-ME-WIT had high Sn (100%) and Sp (99.1%) for detecting villous morphology suggestive of CD. In our previous study, we examined the villous morphology using NBI and high magnification without water immersion on a sequence of videos. The overall Sn and Sp to correctly distinguish the presence or absence of villi in that study were 93.3% and 97.8%, respectively. The Sn and Sp in differentiating partial from total villous atrophy were 83.3% and 100%, respectively.(16) The study group was, however, small and comprised only 21 patients. In another recent study of 105 patients, of which 51 patients had CD, Goswami et al demonstrated that NBI-ME has Sn, Sp, PPV and NPV of 95.0%, 90.2%, 91.2%, and 94.2%, respectively, for diagnosing altered villous morphology.(14) The clinical benefits of the NBI-ME-WIT technique include its ability to facilitate targeted biopsy of abnormal mucosa, which may otherwise be missed on WLE. This technique also has the potential to decrease procedure time and reduce cost and complications if the need for a biopsy can be reduced.
The main strength of our study is that it is a novel case-finding study. We examined the capability of NBI-ME-WIT in identifying CD patients among any presenting patient who required an endoscopy after a cause of GI bleeding was ruled out (i.e. no ulcers/varices, dieulafoy lesions, erosions, etc). In the literature to date, no such study has been performed.
This study has a few limitations. All procedures were performed by a single expert endoscopist, which affects its generalisability. Another potential limitation includes the exclusion of patients with raised IEL counts. The prevalence of raised IEL count was reported to be as high as 5.4% in the general population.(23) Although raised IEL counts can be an early manifestation of CD (Marsh Grade I), this finding is not specific for CD, which is only one of many possible causes. Other causes of raised IEL counts include Helicobacter pylori infection and the use of medications such as nonsteroidal anti-inflammatory drugs, as well as small bowel bacterial overgrowth and systemic autoimmune disorders. Regardless of the cause, a raised IEL count can cause diarrhoea and weight loss. If it is associated with CD, clinical signs of anaemia and dermatological manifestations can often be seen. Natural history, long-term consequences and the role of IEL in CD diagnosis remain uncertain.
In conclusion, NBI-ME-WIT is a specific and sensitive tool to recognise and accurately diagnose villous atrophy. Biopsies can be avoided in patients with normal-sized villi, which may decrease the overall cost of the procedure.


