Abstract
Acute myocardial infarction is one of the conditions frequently managed in the emergency department. There are many complications associated with right ventricular infarction, and the incidence of right ventricular infarction associated with inferior myocardial infarction is as high as 51% based on electrocardiographic findings. We herein report the case of a 45-year-old Chinese man with inferior myocardial infarction complicated by right ventricular failure. He had hypoxaemia refractory to supplemental oxygen due to an acute right-to-left shunting through a patent foramen ovale (PFO). He underwent coronary angioplasty and closure of the PFO. It is crucial for the attending physician to consider the presence of a right-to-left shunt when there is persistent uncorrectable hypoxaemia despite maximal oxygen supplementation in the setting of right ventricular infarction, as there are clinical implications and certain clinical managing principles that should be applied.
INTRODUCTION
Acute myocardial infarction is a condition frequently managed in the emergency department, accounting up to 14.7% of visits for chest pain.(1) The incidence of right ventricular (RV) infarction associated with inferior myocardial infarction is as high as 51% based on electrocardiographic findings(2) and it can occur in isolation in less than 3% of infarcts.(3) RV infarction has many complications, ranging from arrhythmia to hypotension, and there have been described cases of persistent hypoxaemia from the development of an acute right-to-left shunting through a previously dormant patent foramen ovale (PFO). Herein, we report the case of a patient with inferior myocardial infarction complicated by RV failure and had refractory hypoxaemia due to an acute right-to-left shunting through a PFO.
CASE REPORT
A 45-year-old Chinese man presented to our emergency department (ED) with a two-day history of exertional shortness of breath, left-sided chest pain and abdominal bloating. He was a smoker and had a medical history of hypertension, hyperlipidaemia, and inferior and RV ST-elevation myocardial infarction (STEMI) ten years ago status post-percutaneous coronary intervention (PCI) to the distal right coronary artery (RCA). Coronary angiography done three years after PCI showed a patent distal RCA stent. He subsequently defaulted his medications.
On arrival to our ED, his vital signs were as follows: blood pressure (BP) 88/59 mmHg; respiratory rate 22 breaths per min; and oxygen saturation by pulse oximetry 90% on non-rebreather mask. There were decreased breath sounds bibasally with occasional basal crepitations. The jugular venous pressure was not elevated, and the rest of the physical examination was unremarkable.
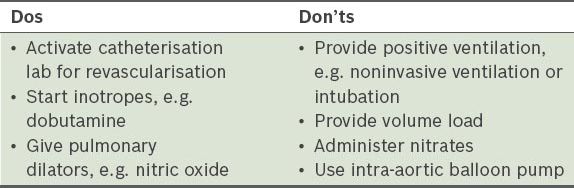
Electrocardiography (ECG) showed evolved inferior STEMI (Figs.
Fig. 1
ECG taken on the patient’s arrival at the emergency department shows evolved inferior ST-elevation myocardial infarction.
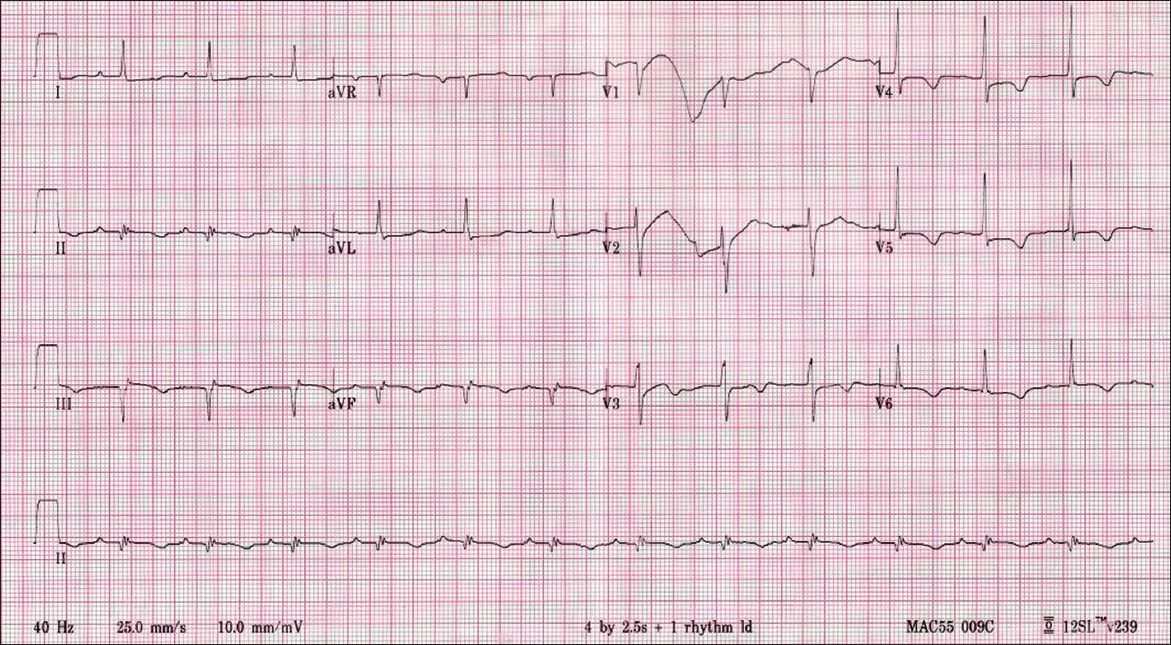
Fig. 2
Right-sided ECG taken on the patient’s arrival at the emergency department shows evolved inferior ST-elevation myocardial infarction.
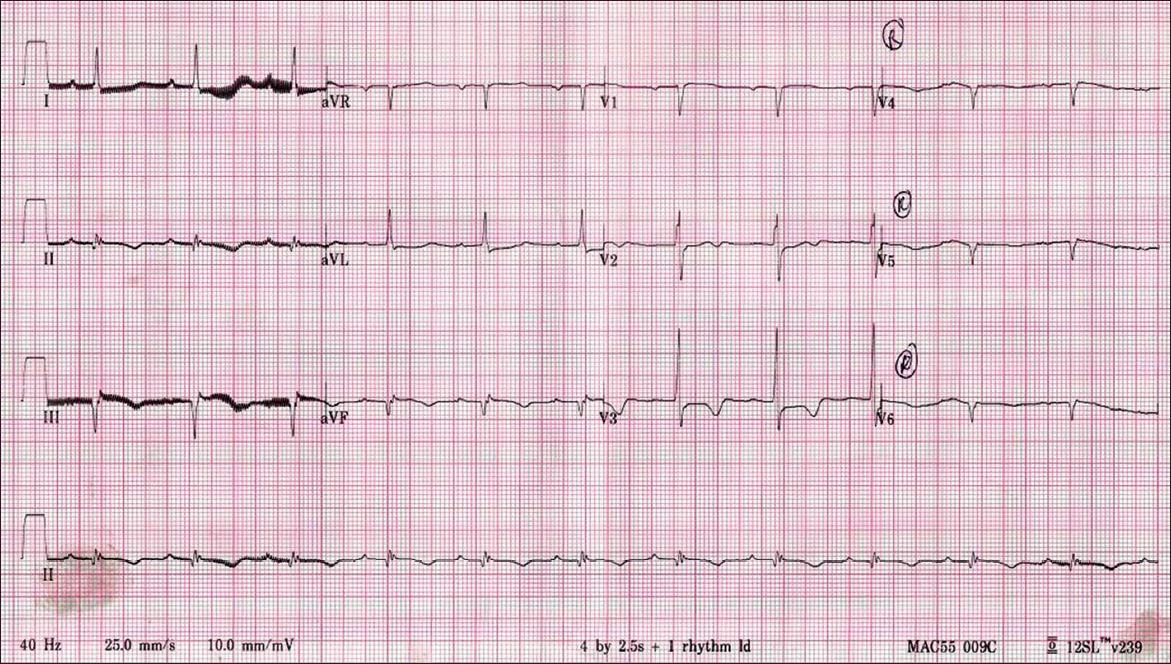
Fig. 3
Previous ECG taken in 2004.
Fig. 4
Radiograph taken on the patient’s arrival at the emergency department.
Coronary angioplasty showed double vessel disease with 50% stenosis of proximal left anterior descending artery (pLAD) and 100% stenosis of proximal RCA (pRCA). Seeing as the patient was hypoxic and hypotensive, balloon angioplasty of the pRCA lesion was performed, with < 30% residual stenosis post-procedure. He subsequently underwent a transthoracic echocardiography (TTE), which showed a PFO with severe biventricular dysfunction with an ejection fraction of 15%. Right-to-left intra-atrial shunting through the PFO was confirmed on saline contrast study (i.e. ‘bubble study’) (
Fig. 5
Echocardiogram shows right-to-left shunted saline bubbles inside the left atrium (arrow).
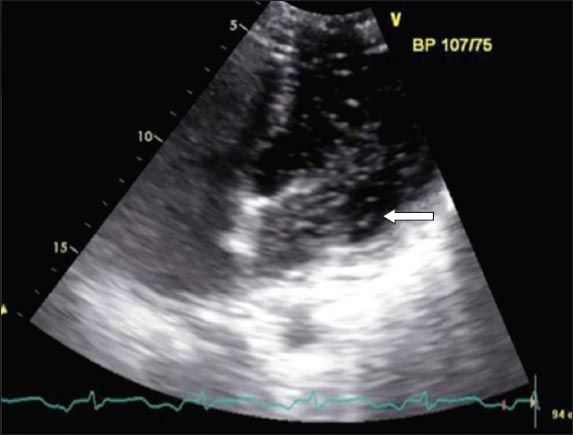
Right-heart catheterisation showed right and left atrial mean pressures of 25 mmHg and 21 mmHg, respectively. The decision was made to close the PFO with a 9 mm Amplatzer occluder rather than a PFO device due to the wide gap. Final transoesophageal echocardiography (TOE) with agitated saline bubble contrast study, however, still showed the right-to-left shunting within the first three cardiac cycles, but the patient’s oxygen saturation improved from 88% to 95% on room air. Repeat TOE three days later showed marked improvement of RV function with no residual shunt. He was discharged well on Day 8 of admission.
DISCUSSION
Hypoxaemia is a recognised complication in acute myocardial infarction; it can occur due to pulmonary oedema from congestive cardiac failure in left ventricular infarct, pulmonary embolism or even due to an acute right-to-left shunting through a previously dormant PFO.(4) A dormant PFO has been described in up to 25%–30% of the general population,(5-7) whereas fusion is complete at birth in the rest of the population.(7)
Patients with PFO remain asymptomatic, as the defect is a flap-like lesion and does not permit left-to-right cardiac shunting normally(8) when the atrial pressure is higher on the left than the right. During atrial contraction in ordinary circumstances, the right atrial pressure rises to 4–6 mmHg whereas the left atrial pressure rises to about 7–8 mmHg.(9) In RV infarction, the pressure in the right atrium increases to above that of the left atrium during inspiration when the right atrium is in diastole,(2,4) and therefore results in a right-to-left inter-atrial shunting. The resultant effect is systemic hypoxaemia refractory to oxygen supplementation. This finding is consistent with that in our report – the patient’s right atrial mean pressure (25 mmHg) exceeds the left atrial mean pressure (21 mmHg).
Our patient has refractory hypoxia not corrected by high-flow oxygen in the absence of significant pulmonary pathology (clear chest radiography and no pulmonary embolism on CT pulmonary angiography [CTPA]). This should lead the attending physician to suspect the possibility of a shunt or haemoglobinopathy, such as methaemoglobinaemia, which is even rarer. The diagnosis of PFO resulting in right-to-left shunting could be clinched at bedside by performing echocardiography via a subcostal view that demonstrates colour flow across the inter-atrial septum. The natural tendency to correct the hypotension would be to give small fluid boluses, but in the context of PFO with a right-to-left shunt, this could result in more shunting and worsening of the hypoxia. In such circumstances, it is best to start intravenous inotropes to improve the haemodynamics.
Hence, in RV infarction, it is essential to consider the presence of a right-to-left shunt when there is persistent uncorrectable hypoxaemia, especially if pulmonary embolism has been excluded with imaging. This consideration is crucial, as certain clinical managing principles need to be applied. Any measure that results in an increase in right-sided cardiac pressures and decreases afterload (which will cause a decrease in the left atrial pressure) will aggravate the right-to-left shunting.(4,10) To avoid an increase in the right-sided cardiac pressure, the physician should avoid positive-end expiratory pressures(4,10) and a volume load(10) whenever possible. If the diagnosis is clear, inotropic agents or even cardiac pacing, if there is an atrioventricular block, may be more effective in cardiogenic shock.(10) In patients with severe refractory hypoxia and cardiogenic shock, the physician may still need to institute positive pressure ventilation (via endotracheal intubation) and start inotropes before sending the patient to the catheterisation lab. Reducing afterload, and hence, left atrial pressure, which then increases shunt flow, is also contraindicated. Intra-aortic balloon pump insertion and nitrates should be avoided.(4,10)
Achieving revascularisation of the culprit lesion in acute myocardial ischaemia is the mainstay of treatment,(11) as reperfusion improves the function of the right ventricle. Hence, the attending physician should recognise the need to activate the cardiac interventionist at the earliest possible time. Other measures to improve RV function, such as starting inotropic agent like dobutamine(11) or using a pulmonary dilating agent like nitric oxide, may also be considered.(10,11) Performing a CTPA in an acute STEMI with hypoxia within the first few hours of chest pain may not be appropriate, as it delays PCI. However, in our patient, the symptoms were already two days old. Hence, we decided to proceed with CTPA to rule out pulmonary embolism as a cause of refractory hypoxia.
Therefore, in the setting of inferior myocardial infarction with RV involvement, it is important for the attending physician to consider the possibility of the presence of a PFO with right-to-left shunting if there is persistent hypoxaemia refractory to oxygen supplementation, so as to avoid medications or interventions that could worsen the shunt (
Table I
A summary of dos and don’ts in an acute right-to-left shunt in right ventricular ST-elevation myocardial infarction.