Abstract
INTRODUCTION
Fetoscopic laser photocoagulation (FLP), a treatment option for twin-to-twin transfusion syndrome (TTTS) in monochorionic twin pregnancies, is currently the treatment of choice at our centre. We previously reported on our experience of FLP from June 2011 to March 2014. This paper audits our fetal surgery performance since then.
METHODS
15 consecutive patients who underwent FLP for Stage II–III TTTS before 26 weeks of gestation from June 2011 to January 2017 were retrospectively reviewed, consisting of five cases from our initial experience and ten subsequent cases. Perioperative, perinatal and neonatal outcomes were analysed.
RESULTS
Of 15 pregnancies, 10 (66.7%) and 5 (33.3%) were for Stage II and III TTTS respectively, with FLP performed at an earlier Quintero stage in the later cohort. Overall mean gestational ages at presentation, laser and delivery were comparable between the cohorts at 19.7 (15.4–24.3) weeks, 20.3 (16.3–25.0) weeks and 31.2 (27.6–37.0) weeks, respectively. 2 (13.3%) cases had intra-amniotic bleeding and 1 (6.7%) had iatrogenic septostomy. 1 (6.7%) case had persistent TTTS requiring repeat FLP, and another (6.7%) had preterm premature rupture of membranes at seven weeks post procedure. The overall perinatal survival rate was 21 (75.0%) out of 28 infants. One mother underwent termination of pregnancy for social reasons at 1.4 weeks post procedure. Double survival occurred in 8 (57.1%) out of 14 pregnancies, while 13 (92.9%) had at least one survivor.
CONCLUSION
FLP requires a highly specialised team and tertiary neonatal facility. Continual training improves maternal and perinatal outcomes, ensuring comparable standards with international centres.
INTRODUCTION
Monochorionic (MC) twins account for 20% of spontaneous twin pregnancies.(1) Sharing a placental mass and fetoplacental circulation, MC twins are interdependent, predisposing them to specific serious complications based on intertwin discordance, including fetal size, amniotic fluid volume, fetoplacental haemodynamics and structural defects. Management of these specific complications requires great expertise, as the risks of neurological damage and death, respectively, are 15% and 26% in the surviving twin after a single fetal death. Twin-to-twin transfusion syndrome (TTTS) is the most common serious complication, occurring in approximately 15% of MC twin pregnancies. Untreated TTTS carries a perinatal loss of 70%–90%.(2) It usually presents after 16 weeks of gestation and rarely after 26 weeks.
TTTS results from chronic imbalanced, unidirectional blood flow from artery to vein through placental arteriovenous anastomoses between the two fetal circulations sharing similar placental masses.(3,4) The donor hence manifests signs of hypovolaemia and growth discordance, with oliguria, oligohydramnios and abnormal umbilical artery (UA) Doppler studies. The recipient twin, conversely, shows signs of being overloaded: polyhydramnios, cardiomegaly with abnormal venous Doppler studies (reverse flow in the ductus venosus [DV]) or pulsatile flow in the umbilical vein and hydrops.
Fetoscopic laser photocoagulation (FLP) as a treatment for TTTS was first reported in the early 1990s.(5) Increased experience has led to refinement of the technique with good outcomes,(5,6) and it is now superior to serial amnioreduction in TTTS diagnosed at less than 26 weeks of gestation.(7,8) TTTS was first introduced at KK Women’s and Children’s Hospital (KKH), Singapore, in June 2011. FLP has since been our choice of treatment for Stage II–III TTTS in MC twin pregnancies presenting between 16 and 26 weeks of gestation. We previously reported our experience with five cases that underwent FLP at our centre from June 2011 to March 2014.(9) This study reviews our overall perioperative, perinatal and neonatal outcomes, and more importantly, audits our fetal surgery performance since then.
METHODS
We conducted a retrospective single-centre review of all MC diamniotic twin pregnancies that were (a) complicated by TTTS of at least Stage II; (b) presented before 26 weeks of gestation; and (c) underwent FLP in our unit from June 2011 to January 2017. A total of 15 cases were reviewed, five cases from June 2011 to March 2014(9) and ten cases subsequently. TTTS was diagnosed using the basic ultrasonography (US) criteria and staged according to the Quintero criteria,(10) as follows – Stage I: ultrasonography finding of fluid discrepancy between the two fetuses (polyhydramnios in the recipient [i.e. deepest vertical pool (DVP) > 8 cm at ≤ 20 weeks’ gestation or > 10 cm at > 20 weeks’ gestation] and oligohydramnios in the donor [DVP ≤ 2 cm]); Stage II: absence of visualisation of the donor’s fetal bladder; Stage III: abnormal Doppler findings; Stage IV: the presence of hydrops; and Stage V: the fetal demise of one or both twins.
Patients were informed of their diagnosis and management options, consisting of FLP, amnioreduction or cord coagulation. Procedure information, particularly FLP and its associated risks including fetal loss, preterm membrane rupture, miscarriage and neurological deficit in the surviving twin(s), was conveyed and an informed choice was made by the patient. Sonographic data on placental localisation and extent, umbilical cord insertion sites, amniotic fluid volume, umbilical vessel, DV and middle cerebral artery (MCA) blood flow as well as cardiac function was collected. The vascular equator, where vessels from either twin anastomose, giving rise to arteriovenous, arterio-arterial (AA) and venovenous anastomoses, was mapped using colour flow Doppler studies. In particular, AA anastomoses were identified.
Informed consent was taken and prophylactic antibiotics administered 30 minutes before the procedure. All procedures were performed by a team of trained operators in the Antenatal Diagnostic Centre at our institution. Instruments included the 2-mm fetoscope (HOPKINS® II Straight Forward Telescope 0° 26008AA; Karl Storz, Tuttlingen, Germany) that was used previously, and the current 3.3-mm fetoscope (Miniature Straight Forward Telescope 0° 11506 AA; Karl Storz, Tuttlingen, Germany), diode laser with a 600-μm laser fibre (Dornier Medilas D MultiBeam; Dornier MedTech, Wessling, Germany), vascular access trocar and cannula (Terumo Radifocus 11F; Terumo Corporation, Tokyo, Japan).
Under US guidance, an avascular area was located perpendicular to the stuck twin and the site of fetoscopic entry was planned based on the placental location and placental cord insertions of both twins. The patient was positioned accordingly in a supine or lateral decubitus position depending on the placental location. Local anaesthetic (subcutaneous infiltration of 1% lignocaine) was given in all cases at the planned entry site on the maternal abdomen. This was followed by a 3-mm skin incision. The trocar and cannula were inserted into the amniotic cavity of the recipient twin using the Seldinger method perpendicular to the vascular equator. In cases of an anterior or fundal placenta, a 30° scope was used to overcome poor visualisation of vessels. FLP was performed using the Solomon technique:(11) after anastomotic vessels along the vascular equator were selectively identified and coagulated using the laser at 30 W, surface coagulation of the placenta between the ablated anastomotic sites was carried out, creating a physical separation of the donor’s and the recipient’s vascular territories on the placenta surface and ablating the superficial AA anastomoses. An anastomosis was identified if the vessel originated from one cord and ended in a cotyledon from which the other emerging vessel ran towards the cord of the other twin. Vessels were not coagulated if the emerging vessel led back to the same twin. At the end of the procedure, amnioreduction was performed in the recipient sac to attain normal liquor volume (DVP < 8 cm). Sonographic assessments of fetal viability and cervical length were also done at the end of the procedure.
After the procedure, patients were admitted for one day for observation and given antibiotics and progesterone. Analgesia and tocolytics were given on demand. Doppler studies (UA, DV and MCA) of both twins as well as fetal viability were routinely performed the next day before discharge.
Cases were closely followed up in the Fetal Medicine Clinic at least once or twice a week initially and later fortnightly for evidence of resolution, persistence or recurrence of TTTS as well as other complications. Persistent TTTS was defined as the persistence of polyhydramnios-oligohydramnios sequence in at least one week following FLP, although no general consensus exists on its precise definition. Parameters measured each visit included DVP, visualisation of fetal bladder, standard deviation score of abdominal circumference and Doppler studies.
Patient demographics, operative details, perioperative and postoperative complications, as well as pregnancy and perinatal outcomes were prospectively collected and retrospectively reviewed. Overall survival rate was calculated using the number of surviving fetuses divided by the total number of fetuses. The double fetal survival rate was the proportion of pregnancies in which both fetuses survived. The at-least-one-twin survival rate was the proportion of pregnancies in which one or both fetuses survived. Obstetric, fetal and neonatal data was obtained from medical records in all cases.
Neonatal death was defined as the number of deaths in infants who were born alive at ≥ 24 weeks of gestation but who died within 28 days. Weight discordance was assessed in surviving pairs of twins and was calculated in the following manner: ([birth weight of the larger twin − birth weight of the smaller twin]/birth weight of the larger twin) × 100%. Significant weight discordance was defined as > 25%.
RESULTS
A total of 28 cases of MC twin pregnancies with Stage II–IV TTTS presented to our unit from June 2011 to January 2017. Of these, three cases went to an overseas institute for further management and another three went to another centre for a second opinion. A further seven cases were unsuitable candidates for FLP, as three cases presented after 26 weeks of gestation and underwent serial amnioreduction, and four cases underwent cord coagulation. In the cord coagulation cases, FLP was technically not possible in two patients due to anatomical or positional causes, including unfavourable placenta positions (anterior/fundal) with close cord insertions and a stuck twin overlying the site of the vascular equator. In the other two cases, it was the patient’s choice: in one, the donor twin had severe intrauterine growth restriction and in the other, the recipient twin had severe fetal hydrops. The remaining 15 cases underwent FLP, five cases from June 2011 to March 2014(9) and ten cases from November 2014 to January 2017.
Overall, 66.7% of the cases had Stage II and 33.3% had Stage III TTTS.
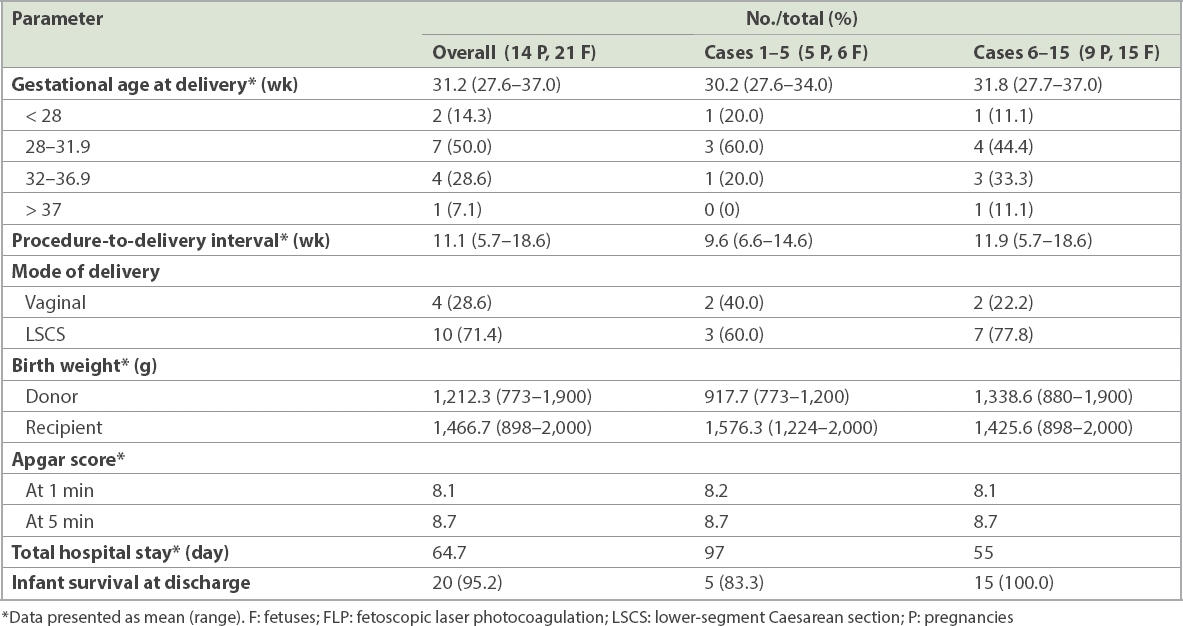
Table I
Antenatal characteristics of 15 cases of monochorionic twin pregnancies that underwent fetoscopic laser photocoagulation in our centre.
Two cases (one case each from the earlier and later cohorts) had intra-amniotic bleeding during the procedure, which resolved spontaneously. Needle insertion in the uterine wall resulted in haematoma formation and bleeding in the first case, while the second case bled from accidental vessel puncture on the placental surface. Iatrogenic septostomy, inadvertent perforation of the twins’ dividing membranes, occurred in Case 14. There were no cases of intraprocedure fluid leak.
Complications were reported in three cases during post procedure follow-up. Case 4 had persistent TTTS despite FLP at 20.3 weeks and amnioreduction at 23.1 weeks (Day 19 post FLP), requiring a repeat FLP at 24.3 weeks (Day 28 post initial FLP). Case 9 had preterm premature rupture of membranes (PPROM) at 24.6 weeks (Day 48 post FLP). Case 10 was noted to have a shortened cervical length of 0.2 cm with cervical dilatation of 0.6 cm, necessitating placement of an emergency cerclage at 21.7 weeks (Day 2 post FLP). There were no cases of placental abruption or twin anaemia polycythemia sequence (TAPS), or any maternal complications such as amniotic fluid embolism, disseminated intravascular coagulation, stay in the intensive care unit and Mirror syndrome – a rare and potentially life-threatening obstetric complication characterised by the development of maternal oedema and preeclampsia in association with fetal hydrops.
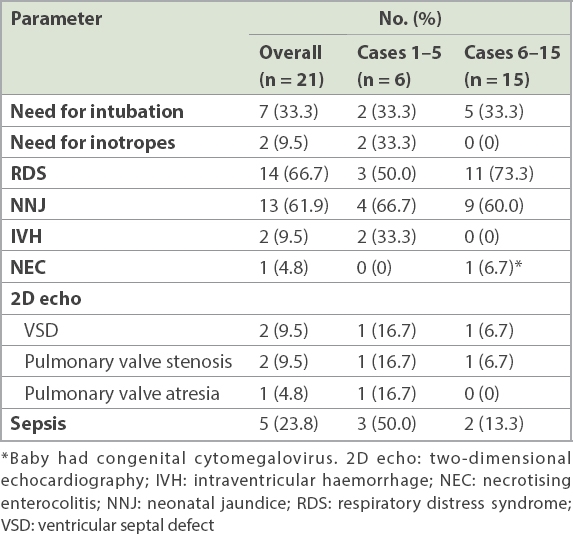
Table II
Post-FLP complications in 15 cases of monochorionic twin pregnancies that underwent FLP in our centre.
Since July 2015, our unit has routinely performed magnetic resonance (MR) imaging of the fetal brain for all cases at four weeks post procedure. During the study period, normal results were seen in all six cases that underwent fetal MR imaging of the brain, with all fetuses showing no evidence of neurological damage at birth.
One recipient and two cases of donor demise were recorded at one day post procedure. Both donors were noted before FLP to be severely growth restricted, with one of them exhibiting absent end-diastolic flow. The recipient was noted to be severely growth restricted prior to FLP, with absent reversed end-diastolic flow and cardiomegaly with poor cardiac contractions. These pregnancies were followed up closely in our MC twin clinic.
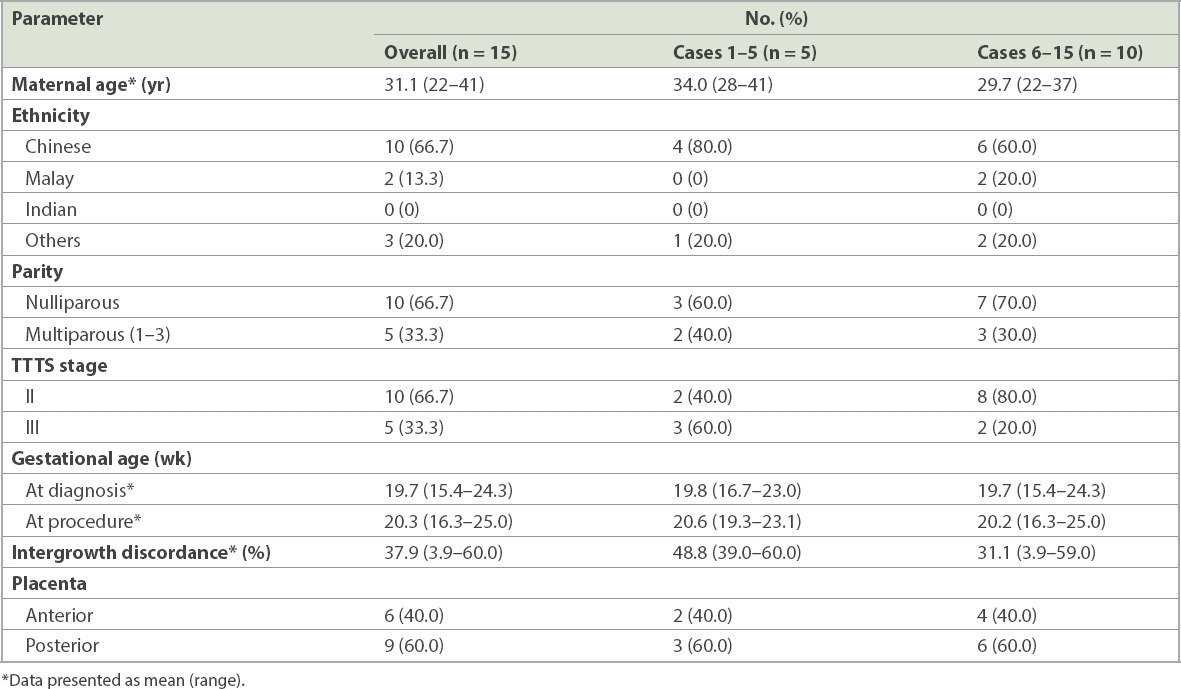
Analysis of the Doppler images (
Fig. 1
Doppler studies show pre- and postprocedure (a) umbilical artery pulsatility index (UAPI); (b) ductus venosus pulsatility index (DVPI); (c) middle cerebral artery pulsatility index (MCAPI) and (d) middle cerebral artery peak systolic velocity (MCAPSV) in both the recipient and donor.
One woman in the later cohort subsequently underwent a mid-trimester termination of pregnancy at 24 weeks of gestation (at postprocedure 1.4 weeks) due to social reasons. Perinatal survival rates are thus reported based on 14 pregnancies and 28 fetuses (
Table III
Perinatal survival rates in 14 cases of monochorionic twin pregnancies that underwent FLP in our centre.
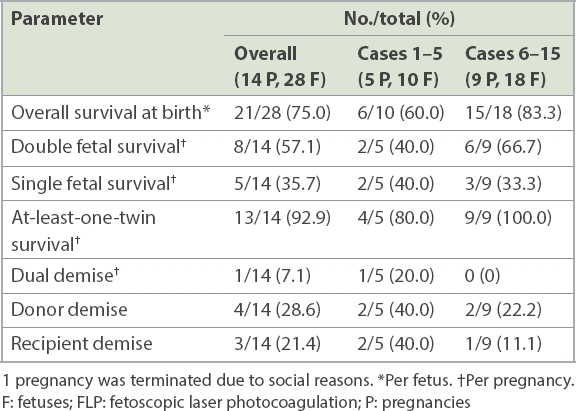
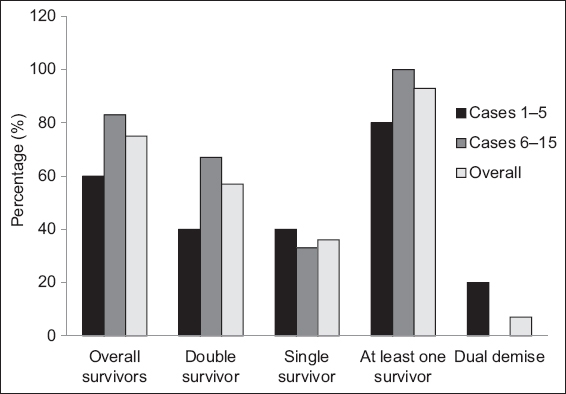
Fig. 2
Bar chart shows a comparison of perinatal outcomes between the earlier cohort (Cases 1–5) and the later cohort (Cases 6–15) of patients who underwent fetoscopic laser photocoagulation.
Perinatal outcomes are described in
Table IV
Overall perinatal outcomes in 14 cases of monochorionic twin pregnancies that underwent FLP in our centre.
When compared with the earlier cohort, the overall, double and at-least-one fetal survival rates were higher in the later cohort, with no dual demise (
Comparatively, the birth weight of the donor twin was greater in the later cohort (1,338.6 g vs. 917.7 g). Although the recipient’s birth weight was lower in the later cohort, the birth weight difference between the donor and recipient was significantly smaller than that of the earlier cohort (6.1% vs. 41.8%). The total length of the neonatal hospital stay was shorter in the later cohort compared to the earlier cohort (55 days vs. 97 days).
There were 21 survivors at birth, of whom 18 (85.7%) required a newborn intensive care unit stay. Among the 3 (14.3%) infants who did not, one was delivered at 36.7 weeks (single survivor) and two at 37 weeks (double survivor). Neonatal death occurred in 1 (4.8%) live-born infant, an ex-donor twin from the earlier cohort on Day 17 of life, due to multiple complications of severe prematurity and sepsis.
Table V
Overall neonatal outcomes for all survivors at birth (n = 21).
DISCUSSION
With better knowledge on TTTS over the past three decades, various interventions have been developed to improve pregnancy outcomes and reduce neonatal morbidity and mortality. These include serial amnioreduction, intertwin septostomy, cord coagulation and FLP. In particular, with the evolution of laser techniques from non-selective to selective sequential as well as the Solomon technique, perinatal survival has significantly increased.(12)
Since the first paper on FLP,(13) different surgical techniques have been developed for carrying it out. Non-selective laser coagulation of placental vessels was initially described, in which any vessel crossing the intertwin membranes (i.e. membranous equator) was coagulated,(5) followed by selective laser coagulation of placental vessels (SLCPV), in which anastomoses crossing between the twins (i.e. vascular equator) were coagulated.(14) Most recently, the Solomon technique, first described by the Leiden group in the Netherlands,(15) involves SLCPV with subsequent surface coagulation of the placenta between the ablated anastomotic sites to create a physical separation of the donor’s and recipient’s vascular territories on the placental surface. It has been reported to reduce residual anastomoses, potentially reducing complications such as recurrent TTTS or TAPS with no increase in adverse outcomes.(11)
Several studies have compared the success rates of FLP with other treatments of TTTS. The Eurofetus study demonstrated that pregnancies complicated by Stage II or III TTTS treated with FLP had a higher likelihood of survival of at least one twin to 28 days and 6 months of age compared to serial amnioreduction.(7) Similarly, a meta-analysis of ten articles, eight of which classified TTTS via Quintero staging, found that fetuses with Stage II–III TTTS undergoing laser ablation were twice as likely to survive and had an 80% reduction in neurologic morbidity compared with fetuses undergoing serial amnioreduction.(8) Selective fetocide with cord coagulation remains an option, with 80%–90% survival of the remaining twin and no neurological abnormalities at birth reported.(16) With the introduction of FLP, however, cord coagulation remains possibly a last resort for preterminal TTTS.(9)
In pregnancies where TTTS was diagnosed after 26 weeks, management options included delivery or amniodrainage. Most published series of TTTS cases treated with laser surgeries are limited to gestation ages under 26 weeks;(7,17) FLP is usually not advocated as the treatment of choice in late presentations of TTTS, possibly due to its higher rates of complications such as PPROM compared to amniodrainage.(18)
Performing FLP in MC twins with an anterior placenta is known to be technically more challenging than in those with a posterior placenta due to a higher risk of injury and bleeding. For these patients, our approach included a detailed pre-fetoscopy US mapping of fetal positions, placental locations and cord insertions. A curved fetoscope coupled with a more lateral approach for fetoscope insertion was undertaken to clearly visualise the placental anastomosis.
The severity of TTTS at diagnosis and gestation age at delivery remain the main factors in determining the perinatal outcomes of such pregnancies.(19) Early diagnosis and timely intervention are thus imperative to achieve the best outcomes. In our study, the overall mean gestational age at diagnosis and procedure was 19.7 weeks and 20.3 weeks respectively, comparable to the mean gestational age at procedure in the international data, which was 20.9 ± 1.9 weeks.(12) When the earlier and later cohorts were compared, 80.0% of patients who underwent FLP in the later cohort had Stage II TTTS compared to 40% in the earlier cohort. This can be attributed to the earlier detection of TTTS through greater awareness and screening, together with the fact that our centre is the largest tertiary maternal unit in the country.
In our study, most donors showed a reduction in UAPI, MCAPI and MCAPSV, while the majority of the recipients showed increased resistance in UA blood flow (UAPI) and an increase in MCAPI and MCAPSV after the procedure. Our results are similar to those of various papers(20-22) in which fetal vasoconstriction maintained blood pressure and minimised changes in cerebral blood flow secondary to the drop in systemic blood flow in recipients after FLP, leading to an increase in MCAPI.
DVPI has been noted to increase in donors following FLP, a transient rise associated with benign hydropic findings due to transient relative hypervolaemia, congestive heart failure and hypertension.(21,22) In the present study, there was an increase in DVPI in the majority of the recipients. This was surprising, as a reduction would be expected following FLP.(23) Flow pulsatility in the DV is usually a reflection of pressure changes in the inferior vena cava and right atrium.(24) Pressure is usually elevated together with other signs of right heart overload in the recipients prior to FLP. Our findings may be explained by the limited data available (six out of 14 cases).
The most common serious complication following FLP is PPROM, which has an incidence of as high as 27%(25) and carries significant neonatal morbidity and mortality due to complications from preterm labour, prematurity and chorioamnionitis.(26) In our series, 1 (6.7%) out of 15 cases had PPROM on postprocedure Day 48, at 24.6 weeks of gestation. Other complications reported in the literature include TAPS,(27) recurrence of TTTS in 29.6% of pregnancies post FLP(28) and adverse long-term neurodevelopmental outcomes in 6%–18% of survivors.(29) In our series, Case 4 had persistent TTTS requiring repeat FLP at 24.3 weeks (Day 28 post initial FLP) and Case 10 required an emergency cerclage at 21.7 weeks (Day 2 post procedure).
Following FLP, MC twins with TTTS are at an increased risk of neurodevelopmental impairment, with a 6.1% and 11.1% rate of neurologic morbidity at birth and at 6–48 months of age, respectively.(30,31) The risks, however, do not differ significantly from the baseline risks of MC twin pregnancies without TTTS matched for gestational age at delivery, suggesting that neurological damage may be secondary to complications of prematurity rather than a direct result of TTTS or FLP.(32) Prenatal MR imaging has been an established method in the diagnosis of brain damage after FLP.(17) In our study, there was good correlation between the results of fetal MR imaging of the brain and neurological outcome at birth for all our patients, with no intrauterine death or evidence of neurological damage reported in the surviving twin after delivery. This can likely be attributed to the success of our laser procedure in achieving division of all significant vascular communications between the twins.
The mean gestational age at delivery was 31.2 weeks in our study and was comparable between both cohorts (Cases 1–5: 30.2 weeks, Cases 6–15: 31.8 weeks). This is significantly earlier than the 32.4 ± 1.3 weeks reported in the literature(12) and was likely due to our more cautious approach to management of these complicated twin pregnancies with a lower threshold for delivery. Six women were delivered due to worsening Doppler findings and severe intrauterine growth restriction, and five due to preterm labour. Despite the earlier mean gestational age at delivery of the later cohort, infants in this group had a shorter hospital stay (55 days vs. 97 days) compared to those in the earlier cohort, reflecting the improved knowledge and experience of our neonatal unit, the largest one in Singapore, in managing preterm babies. Although neonatal morbidities were comparable in both the earlier and later cohorts, there was a higher rate of respiratory distress syndrome in the later cohort (73.3% vs. 50.0%). This may be attributed to the larger number of emergency Caesarean sections that were performed for these pregnancies for causes unrelated to TTTS, leading to inadequate time for steroid injection.
The double, single and at-least-one fetal survival rates in our study were 57.1%, 35.7% and 92.9% respectively. In a systematic review of 34 studies with 3,868 MC twin pregnancies published over the last 25 years,(12) the mean perinatal survival of both twins, one twin and at least one twin was 52% ± 14.8%, 29% ± 10.5% and 81% ± 8.3%, respectively. The overall survival rate of fetuses was 69.1%, which is comparable to the rate of 75% in our study.
Comparing the two cohorts, better perinatal outcomes (overall, double and at least one survivor) were observed in the later cohort compared to the earlier cohort, with 100% survival of at least one fetus in the later cohort. Although gestational age at diagnosis and procedure were similar in both cohorts, gestational age at delivery was more advanced in the later cohort (31.8 weeks vs. 30.2 weeks), translating to a longer procedure-to-delivery interval (11.9 weeks vs. 9.6 weeks). Possible reasons for this were a more experienced team and better facilities and equipment. In July 2015, from Case 5 onwards, our institution opened the Fetal Surgery Suite, a place dedicated to fetal procedures, and we started using the 3.3-mm fetoscope (Miniature Straight Forward Telescope 0° 11506 AA; Karl Storz).
Stage I TTTS has always presented clinicians with a management dilemma. Intervention may lead to procedure-related complications, while expectant management risks potential deterioration. Various management options have been described, from expectant to amnioreduction, septostomy and laser photocoagulation of placental anastomoses. Most clinicians managed these patients conservatively,(33) only recommending treatment if the patient lived far from hospital, was symptomatic for polyhydramnios or had a short cervix (< 1.5 cm). O’Donoghue et al(34) suggested that in the majority of Stage I TTTS cases, the disease remained stable (28.3%) or regressed (41.3%). Perinatal survival rates in those that regressed, remain stable and progressed were 90%, 69% and 46%, respectively. Huber et al(35) reported similar findings – only 14% of Stage I TTTS detected at 20 weeks progressed to a more severe stage requiring intervention. No difference in survival rates was reported in Stage I TTTS between cases who were treated with FLP and those who were managed conservatively.(36) Stage I TTTS is managed expectantly in our centre, with FLP being offered to women with Stage II–III TTTS.
Our study has several limitations. The 15 cases that underwent FLP were not compared with patients who underwent other procedures that are also being used in our institution, namely amnioreduction and cord coagulation. Long-term neurological and developmental outcomes were also not assessed in our case series, as the focus was on neonatal outcomes. Due to the retrospective nature of our study, the following data was not included, unlike in prospective data collection: number of anastomosis lasered, operation time and site of port insertion.
As FLP is a highly specialised procedure for which experience in the technique translates into an increase in survival rates,(37,38) continual training and a transparent audit system (e.g. publication of outcomes) need to be in place to point out areas for improvement. Although our centre sees a lower volume of TTTS requiring intervention compared to larger overseas institutions, we constantly strive to improve our skills and provide dedicated, comprehensive care to these high-risk pregnancies through the use of training models and various fetoscopy training workshops (
Fig. 3
Photograph shows the use of a training model at our institution to practise the skills required for fetoscopic laser photocoagulation.
Until recently, there were no formal training programmes for this procedure.(39) Historically, most providers observed 5–10 procedures at an established centre before they began to offer the treatment at their own centre. In certain situations, a new physician or a trainee would join the established group and gradually transform from observing to assisting and, finally, to acting as the primary surgeon. To shorten the learning curve, Papanna et al(40) suggested that operators could be trained by an experienced operator rather than only through observation, and progress from easier to more challenging cases. A typical new trainee might observe and assist in 15–20 cases of FLP and then perform about 20 additional cases as a primary surgeon before achieving competency. The findings by Papanna et al are important because they could be useful in establishing guidelines for training future fetoscopic interventionists and developing methods for regular audits of the performance of individual operators or centres. A performance assessment tool for FLP, developed by Peeters et al,(41) included a validated checklist of the essential steps in FLP and also evaluated its use to ensure the competency of future fetal surgeons before they performed the procedure unsupervised, illustrating that operators may reach a level of competence after at least 25 FLP procedures. Previous studies have reported learning curves for FLP by grouping initial series and comparing it to later series. Similar to this paper, they illustrated improved outcomes in the later series.(37) However, these studies do not describe operator-specific outcomes or the number of cases that an operator would need to reach competency to perform FLP.
In conclusion, FLP, although an effective treatment for TTTS, requires a team skilled in carrying out interventional therapies for the treatment of potential complications as well as tertiary neonatal facilities. Knowledge about the condition is also vital to ensuring timely referrals and interventions. This series shows that outcomes of FLP for TTTS in our centre were comparable to those of the international published literature and have remained stable after an initial learning curve.


