Abstract
INTRODUCTION
Anorexia nervosa (AN) and eating disorders not otherwise specified (EDNOS) are on the rise in Singapore. Abnormal liver function tests have been reported for up to 12.2% of patients with AN. These patients are also known to present with comorbid psychiatric disorders. This study aims to investigate the correlation between body mass index (BMI) and the severity of abnormal liver function tests, and between BMI and the presence of comorbid psychiatric disorders.
METHODS
A retrospective cohort analysis of 373 patients diagnosed with AN or EDNOS at a tertiary hospital was performed. The clinical course of transaminitis and comorbid psychiatric disorders was correlated with the patient’s BMI.
RESULTS
Patients with a BMI of ≥ 16.6 kg/m2 at their first consult had a significantly lower risk of having comorbid psychiatric disorders (χ2 = 32.08, p < 0.001). These patients were five times less likely to have comorbid psychiatric disorders as compared to patients from the other BMI groups (odds ratio [OR] 0.21). On the other hand, patients with a BMI of < 14.6 kg/m2 had a significantly higher risk of having transaminitis (χ2 = 72.5, p < 0.001). They were 11.1 times more likely to develop transaminitis as compared to patients with a BMI of ≥ 14.6 kg/m2 (OR 11.05).
CONCLUSION
Severity of BMI can be used by clinicians as an indicator to assess for secondary psychiatric comorbidities and/or transaminitis during the first consultation. This could help reduce the morbidity and mortality rates in patients with AN or EDNOS.
INTRODUCTION
Eating disorders such as anorexia nervosa (AN) and eating disorders not otherwise specified (EDNOS) are real, treatable medical conditions that are on the rise. An EDNOS is defined as any eating disorder that does not fulfil the criteria for AN as stated in the 4th edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-IV). AN frequently coexists with other medical or psychiatric illnesses that can complicate the treatment process and place the individual at a higher risk of morbidity and mortality. People with AN have been shown to be 18 times more likely to die early compared to people of a similar age within the general population.(1) The high morbidity may be related to the medical complications associated with AN, the comorbid psychiatric disorders and/or the eating disorder alone.
The DSM-IV criteria for AN(2) are as follows:
-
A refusal to maintain body weight at or above a minimally normal weight for the age and height of the individual (e.g. weight loss leading to maintenance of body weight that is < 85% of that expected, or failure to have expected weight gain during a period of growth, leading to body weight < 85% of that expected).
-
An intense fear of gaining weight or becoming fat, even though the individual is underweight.
-
Disturbance in the way in which one’s body weight or shape is experienced, undue influence of body weight or shape upon self-evaluation, or denial of the seriousness of the current low body weight.
-
In postmenarcheal women, amenorrhoea, i.e. the absence of at least three consecutive menstrual cycles. (A woman is considered to have amenorrhoea if her periods occur only after administration of hormones, e.g. oestrogen.)
Individuals suffering from AN can be categorised to have either (a) restrictive behaviour or (b) binge-eating or purging behaviour (i.e. self-induced vomiting, or the misuse of laxatives, diuretics or enemas). In the restrictive type, the person has not regularly engaged in binge-eating or purging behaviour during the current episode of AN. In the binge-eating/purging type, the person has regularly engaged in binge-eating or purging behaviour during the current episode of AN.(2)
The impact of eating disorders is multifaceted. Apart from the psychological aspects of the disease, many patients with eating disorders suffer from serious medical complications. Abnormal liver function tests, often demonstrating elevated transaminases, have been reported for up to 12.2% of patients in the community with AN and for up to 60% of hospitalised patients.(3,4) Although rare, there have been case reports of fatalities due to liver failure among patients with AN.(5,6)
The severity of hepatocellular injury has been reported to be inversely related to the body mass index (BMI) of patients; it can usually be reversed with nutritional intervention.(7) Case studies have shown that a brief elevation of hepatic enzymes occurs secondary to liver ischaemia in cases of AN.(8-10) The mechanisms for this complication are unclear. Common histological findings include liver cell glycogen depletion (under electron microscopy examination) and increased serum alanine aminotransferase in the absence of significant hepatocyte necrosis. Starvation-induced autophagy in the human liver, amongst other mechanisms, may be involved in the death of liver cells in AN cases. Many patients with AN also have hepatic steatosis (on imaging of the liver), which is thought to be related to protein-energy malnutrition.(8,11) The acute rise in liver enzymes, which results from ischaemic hepatitis secondary to liver hypoperfusion, is typically resolved through therapeutic intervention comprising fluid support and gradual nutritional support.(12,13)
There are few studies in the literature that support the correlation between BMI and medical and/or psychiatric complications.(14,15) Comorbid depressive symptoms are the most common psychiatric disorders that are associated with eating disorders.(16-19) A study by Lee et al on a population of patients with AN in Singapore found that depression was the most common comorbid psychiatric condition, affecting 25.4% of the patients.(19) There may be a correlation between the severity of depression and the extent of AN.(20) Other psychiatric disorders commonly seen among patients with AN include anxiety and obsessive-compulsive disorders.(21-23) Personality change is also postulated to have a correlation with the degree of severity of AN.(24) Aspects of personality change may differ between individuals, and can range from irritability to being argumentative or obstinate. These changes are often noticed by the patients’ family members as their condition deteriorates and weight loss becomes more severe.
The principal aim of the present study is to correlate the presence of transaminitis with the severity of AN and EDNOS, through the measurement of BMI. The study also attempts to correlate comorbid psychiatric disorders with BMI in this population of patients with AN or EDNOS.
METHODS
This was a retrospective cohort study of patients with eating disorders who were under the care of the Department of Psychiatry, Singapore General Hospital, from 1 January 2003 to 31 December 2010. The study period was chosen because the Eating Disorder Service at the Singapore General Hospital was only set up in 2003; an arbitrary cut-off date (i.e. 31 December 2010) had to be set to facilitate the completion of the thesis. The study was approved by the SingHealth Centralised Institutional Review Board, which operates in accordance with the International Conference on Harmonisation’s guidelines.(25) The power of the study was calculated with the help of a statistician.
Inpatients and outpatients who met the following criteria were included in the present study: (a) aged 12–40 years; (b) fulfilled the first three DSM-IV criteria for AN or diagnosed with EDNOS (they did not need to fulfil the criteria for amenorrhoea); and (c) had liver function tests done. At the Singapore General Hospital, all patients with eating disorders have to adhere to a treatment protocol, which requires a series of blood investigations, including liver function tests, during the first consultation. This age range was set because patients aged < 12 years would be treated in a hospital specialising in paediatric care (i.e. such patients would not be transferred to the Singapore General Hospital) and the epidemiology of first-onset eating disorders shows that they usually occur in the younger age groups; the catchment for eating disorder patients aged > 40 years was small. The following were excluded from the study: (a) patients with existing liver diseases; (b) patients who had a drug overdose (both intentional and accidental) during the first consultation; and (c) patients with a history of substance abuse (e.g. alcohol and/or opiate abuse), as their pre-existing medical conditions may have resulted in liver function abnormalities.
Only information from the patients’ first consultation was used in the analysis, as the clinical presentation often changes with disease progression. The other rationale behind this choice was to enable the results to be used to facilitate future decision-making on treatment options for patients with eating disorders during their first consultation. Data mining was done by two independent doctors from April 2012 to March 2013. The doctors were trained together to ensure consistency in data collection.
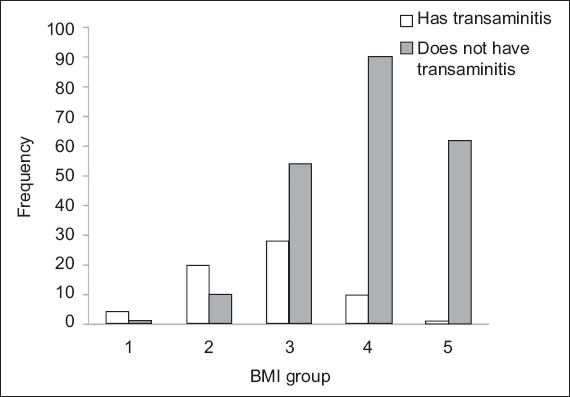
A total of 373 patients were identified for the study, but only 281 were included; 92 patients were excluded, as 89 of them had either bulimia nervosa or EDNOS with a BMI > 18.5 kg/m2, and three exceeded the age limit of 40 years. The case notes of eligible patients were reviewed and information on the demographics, BMI, presence and absence of liver function abnormalities, and presence and absence of comorbid psychiatric disorders were collected. The 281 patients were divided into five groups, according to their BMIs, in order to find out which BMI range was associated with the highest risk of developing transaminitis and having a comorbid psychiatric condition. The BMI groups were as follows: (a) Group 1: patients with a BMI ≤ 10.5 kg/m2; (b) Group 2: patients with a BMI of 10.6–12.5 kg/m2; (c) Group 3: patients with a BMI of 12.6–14.5 kg/m2; (d) Group 4: patients with a BMI of 14.6–16.5 kg/m2; (e) Group 5: patients with a BMI 16.6–18.5 kg/m2. As our study population consisted of patients with AN subtypes, the upper limit of their BMI was set at 18.5 kg/m2 and there was no restriction on the lower limit. Details of the 281 patients are shown in
Table I
Demographic and clinical characteristics of the five body mass index groups (n = 281).
Liver function abnormalities were determined by checking for elevated aspartate aminotransferase, alanine aminotransferase and alkaline phosphatase in the liver panel performed during the initial consult. If the patient had any result that was above the stipulated range for the respective liver enzymes, the patient would be considered to have liver function abnormalities or transaminitis. As slimming pills may be a confounder to transaminitis and since it may be hard to delineate transaminitis from severe starvation, patients who were on slimming pills (n = 14) were excluded from the transaminitis analysis. The comorbid psychiatric disorders that were studied included major depressive disorder, generalised anxiety disorder and obsessive-compulsive disorder (as defined in the DSM-IV criteria). We also analysed the personality changes that were reported by family members, such as irritability, being argumentative, obstinacy and depressive features not amounting to major depressive disorders; these personality changes were categorised together under comorbid psychiatric disorders.
Statistical analysis was done using IBM SPSS Statistics for Windows version 21.0 (IBM Corp, Armonk, NY, USA). Parametric variables were analysed using t-tests and chi-square tests, while nonparametric variables were analysed using Kruskal-Wallis tests and Mann-Whitney U tests, for continuous and categorical variables, respectively. Statistical significance was set at 0.05. The data analyst was blinded to the data collection.
RESULTS
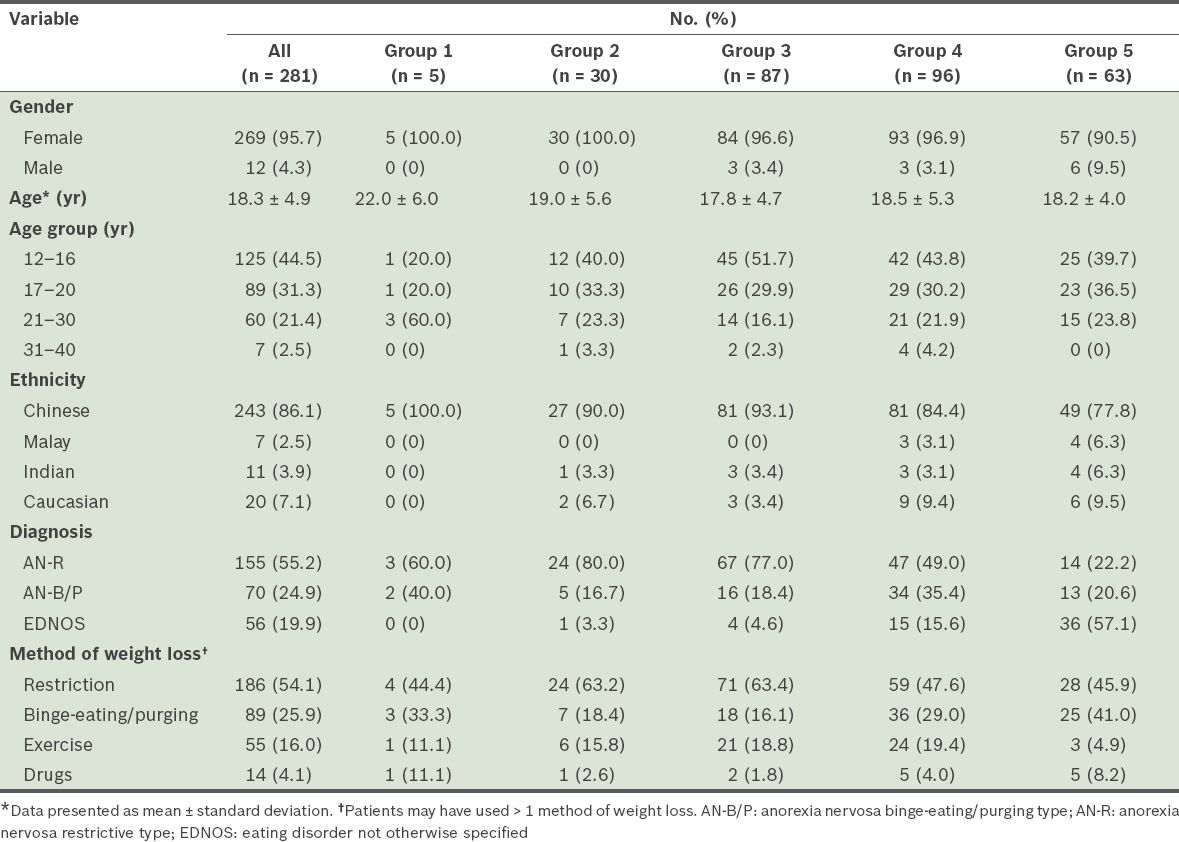
The presence of comorbid psychiatric disorders in the various BMI groups was compared using chi-square tests. This was done to see if there was a relationship between BMI and the presence of psychiatric comorbidities.
Fig. 1
Bar graph shows the frequency of patients with and without psychiatric comorbidities in each BMI group. Group 1: ≤ 10.5 kg/m2; Group 2: 10.6–12.5 kg/m2; Group 3: 12.6–14.5 kg/m2; Group 4: 14.6–16.5 kg/m2; Group 5: 16.6–18.5 kg/m2.
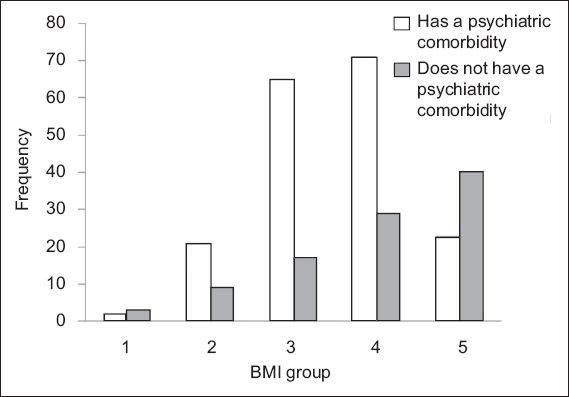
To evaluate if there were any associations between BMI and the presence of transaminitis, the presence of transaminitis in the various BMI groups was compared.
Fig. 2
Bar graph shows the frequency of patients with and without transaminitis in each BMI group. Group 1: ≤ 10.5 kg/m2; Group 2: 10.6–12.5 kg/m2; Group 3: 12.6–14.5 kg/m2; Group 4: 14.6–16.5 kg/m2; Group 5: 16.6–18.5 kg/m2.
DISCUSSION
When a patient with an eating disorder first presents to the Singapore General Hospital’s Eating Disorder Service clinic, some measurements, such as BMI, can be taken fairly easily by either the nurses or the patients themselves. These measurements can be obtained even before the patient enters the consultation room as there are weighing machines that can be used at no charge at the waiting area.
Comorbid psychiatric disorders can be difficult to identify during the first consult as patients may not be forthcoming and there may not be any family members present to provide a collaborative history. If undiagnosed, these patients with eating disorders will retain their psychological distress until the next review. There is also no guarantee that they will be cooperative in giving relevant information regarding any psychiatric distress experienced during the follow-up sessions.
From the results of the present study, we found that patients with BMIs 16.6–18.5 kg/m2 had a significantly lower risk of having comorbid psychiatric disorders as compared to those with BMIs < 16.6 kg/m2 (OR 0.183). The bulk of the psychiatric comorbidities were found among the patients from BMI Group 3 (i.e. BMI 12.6–14.5 kg/m2); this group of patients was found to be at a significantly higher risk of having psychiatric comorbidities as compared to the patients from the other groups (OR 3.33). Thus, it is important to identify patients with eating disorders who have BMIs < 16.6 kg/m2 as this group is more likely to have comorbid psychiatric disorders. These comorbid disorders, especially depression, can compromise the treatment of eating disorders as they may result in a loss of appetite. These comorbidities also result in these patients with eating disorders becoming even sicker than they already are, compromising the recovery process.(26) Therefore, it is important that patients with eating disorders who have BMIs < 16.6 kg/m2 are promptly screened for comorbid psychiatric disorders and treated early so that the vicious cycle does not impede their recovery.
This finding is one that could revolutionalise first consults at eating disorder clinics. Clinicians can use a patient’s BMI as a gauge to check on the history provided. Patients who have BMIs < 16.6 kg/m2, particularly those with BMIs 12.6–14.5 kg/m2, should be screened more closely for the presence of comorbid psychiatric disorders. Using the same principles as those used to screen for comorbid psychiatric disorders, BMI can also be used as an adjunct in determining the risk of transaminitis. Early identification of patients who have transaminitis is important as refeeding syndrome may set in following nutritional rehabilitation and this can cause more insult to the hepatocytes. There is a need for early referral to the gastroenterology department to seek a professional opinion on how these patients with eating disorders and liver dysfunctions can be co-managed.
Based on the findings of the present study, patients with BMIs < 14.6 kg/m2 were 11.1 times more likely to develop transaminitis as compared to patients with BMIs ≥ 14.6 kg/m2 (OR 11.05). When the individual BMI groups were compared against the other groups, it was found that patients in BMI Groups 1, 2 and 3 were more likely to develop transaminitis relative to the patients in the other BMI groups (OR 14.64, 9.63 and 2.42, respectively). In contrast, patients from BMI Group 4 (i.e. 14.6–16.5 kg/m2) and Group 5 (i.e. 16.6–18.5 kg/m2) were less likely to develop transaminitis relative to the patients from the other groups (OR 0.27 and 0.04, respectively). Thus, a BMI cut-off of 14.5 kg/m2 can be used to indicate the need to test for the presence of transaminitis in a patient with eating disorders. If clinicians are managing patients with eating disorders who have BMIs < 14.6 kg/m2, nutritional rehabilitation should not be started until all liver function tests have been performed and there is evidence that the patient is not suffering from transaminitis. Even if these patients test negative for transaminitis at the first visit, they should be closely monitored during treatment as they have an increased risk of transaminitis.
The present study was not without limitations. As it is a retrospective cohort study, it was difficult to draw a direct causal link between BMI and comorbid psychiatric disorders, and between BMI and transaminitis. However, the evidence that was obtained from the analysis was adequate to infer the possible links. The present study was also limited by the quality of the data collected as it is a retrospective study that made use of an available registry on patients with eating disorders.
To conclude, patients with AN or EDNOS were found to be at an increased risk of having comorbid psychiatric disorders and transaminitis. The findings of the present study provide evidence that patients with AN or EDNOS who have a BMI < 16.6 kg/m2 and < 14.6 kg/m2 are at a higher risk of having comorbid psychiatric disorders and transaminitis, respectively. The clinical course of liver function abnormalities and comorbid psychiatric disorders appeared to be correlated with improvements in the patient’s BMI. Specifically, patients with BMIs < 16.6 kg/m2 and comorbid psychiatric disorders were found to be more likely to recover from their comorbid psychiatric disorder with nutritional rehabilitation.
ACKNOWLEDGEMENTS
We would like to thank Dr Andrew Green for his assistance in the data collection of the present study, and Dr Jason Chang and Dr Tay Hiang Keat from the Department of Gastroenterology, Singapore General Hospital, for their patience in answering our queries on liver function abnormalities. We also thank Miss Pearlene Lim, research assistant from the Department of Psychiatry, Singapore General Hospital, for helping with the data analysis.


