Dear Sir,
A 45-year-old Caucasian woman with a history of Type 1 insulin-dependent diabetes mellitus presented with fever, nausea and malaise. She was treated with empirical antibiotics but returned with complaints of dizziness after one week. Physical examination showed a blood pressure of 107/52 mmHg and signs of mild fluid overload.
Electrocardiography (ECG) showed junctional bradycardia at 33 beats per minute (bpm). Chest radiograph showed increased interstitial shadowing with bilateral pleural effusions, consistent with mild cardiac failure. Serum high-sensitive troponin T level was mildly elevated at 45.1 (normal range [NR] ≤ 14) pg/mL and N-terminal pro-B-type natriuretic peptide (NT-proBNP) level was 1,044 (NR 0–125) pg/mL. The rest of the laboratory results were as follows: haemoglobin level 11.1 g/dL, white blood cell count 9.59 × 109/L (with mild lymphopenia), platelet count 458 × 109/L, urea level 8.0 mmol/L, creatinine level 84 μmol/L, albumin level 40 g/dL, aspartate aminotransferase level 284 U/L and alanine aminotransferase level 167 U/L. Her C-reactive protein level was 31.1 mg/L and procalcitonin level was less than 0.06 ng/mL.
The patient was initially administered the empiric antiviral Tamiflu (oseltamivir) and antibiotics doxycycline and Augmentin (co-amoxiclav). She was also treated with frusemide for diuresis. Continuous ECG monitoring showed infrequent sinus pauses of up to 8.6 seconds (
Fig. 1
Continuous ECG monitoring strip shows one episode of sinus pause. The longest pause detected in our patient was 8.6 seconds.

Echocardiography revealed a mildly dilated left atrium with diffusely thickened left atrial wall measuring 6 mm (
Fig. 2
Echocardiogram shows a left atrium (LA) wall that was (a) diffusedly thickened (arrows) before corticosteroids and (b) of normal thickness six months after treatment. The left ventricle (LV) wall was of normal thickness at initial presentation. Ao: aorta
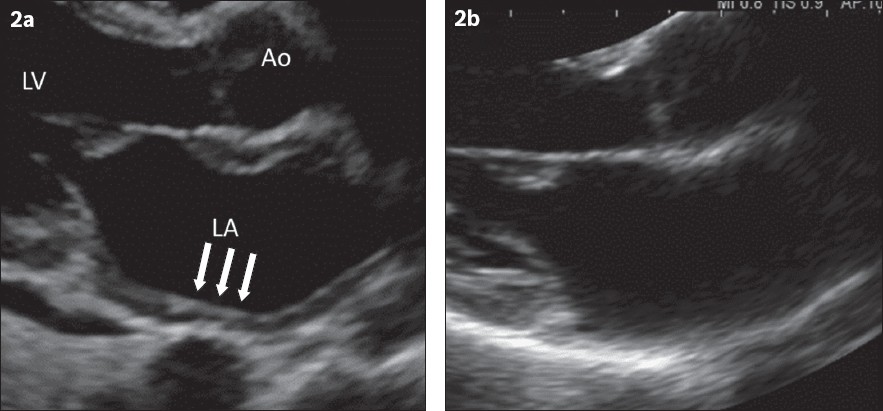
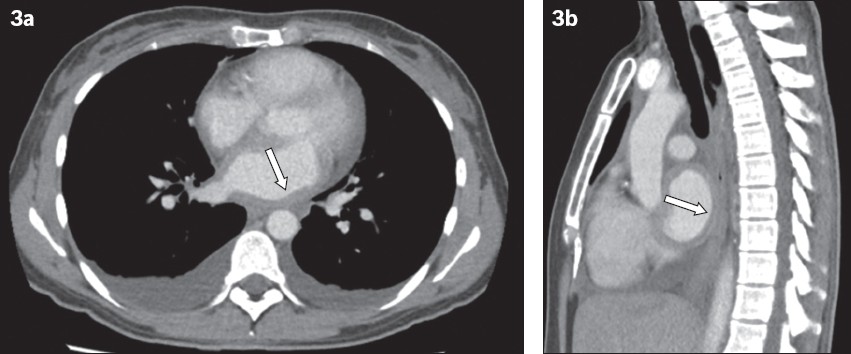
Fig. 3
CT images of the chest in (a) transverse view and (b) sagittal view show diffused thickening of the left atrium (arrows) and bilateral pleural effusions.
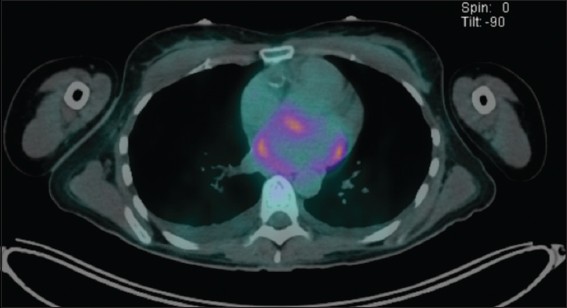
Fluorodeoxyglucose (FDG) positron emission tomography (PET)-CT imaging for FDG avid foci showed heterogenous uptake along the wall of the left atrium, including the left atrial appendage (
Fig. 4
PET (positron emission tomography)-CT image shows heterogenous fluorodeoxyglucose uptake along the wall of the left atrium, including the left atrial appendage.
These findings were consistent with the diagnosis of isolated left atrial myocarditis. The patient was advised to undergo a biopsy of her atrial tissue for histological diagnosis before commencing immunosuppressive therapy; however, she declined. She was started on high-dose oral prednisolone at 1 mg/kg/day on Day 9 of hospital admission. The sinus pauses completely resolved, but she developed atrial fibrillation and atrial flutter with rapid ventricular conduction between 100 bpm and 150 bpm, requiring nebivolol for rate control. No significant ventricular arrhythmias were noted throughout the admission. After 18 days of atrial fibrillation and flutter, the patient spontaneously reverted to sinus rhythm. Troponin levels normalised after starting oral steroids. Follow-up cardiac magnetic resonance (MR) imaging prior to discharge showed mild late gadolinium contrast enhancement (LGE) in parts of the left atrial wall measuring 4–5 mm in thickness, indicative of patchy inflammation or fibrosis in the left atrium. No inflammation or fibrosis was noted in the right atrium or ventricular walls.
The patient experienced infrequent episodes of short-lasting palpitations after discharge, with no further episodes of fever or dizziness. Serial 24-hour ambulatory ECG monitoring showed short runs of non-sustained atrial tachycardia, which reduced in frequency over time. Repeat echocardiography also showed resolution of the left atrial wall thickening and pericardial effusion (
Atrial involvement can occur concurrently with ventricular myocarditis in patients with myocarditis of different aetiologies.(1) However, isolated atrial myocarditis is an uncommon presentation of acute myocarditis. The most commonly reported aetiology of isolated atrial myocarditis is giant cell myocarditis.(2) Other reported causes include sarcoidosis and lymphocytic myocarditis.(3) The clinical features and outcomes of previously reported cases of atrial myocarditis are summarised in
Table I
Summary of clinical features and outcomes of reported cases of isolated atrial myocarditis.
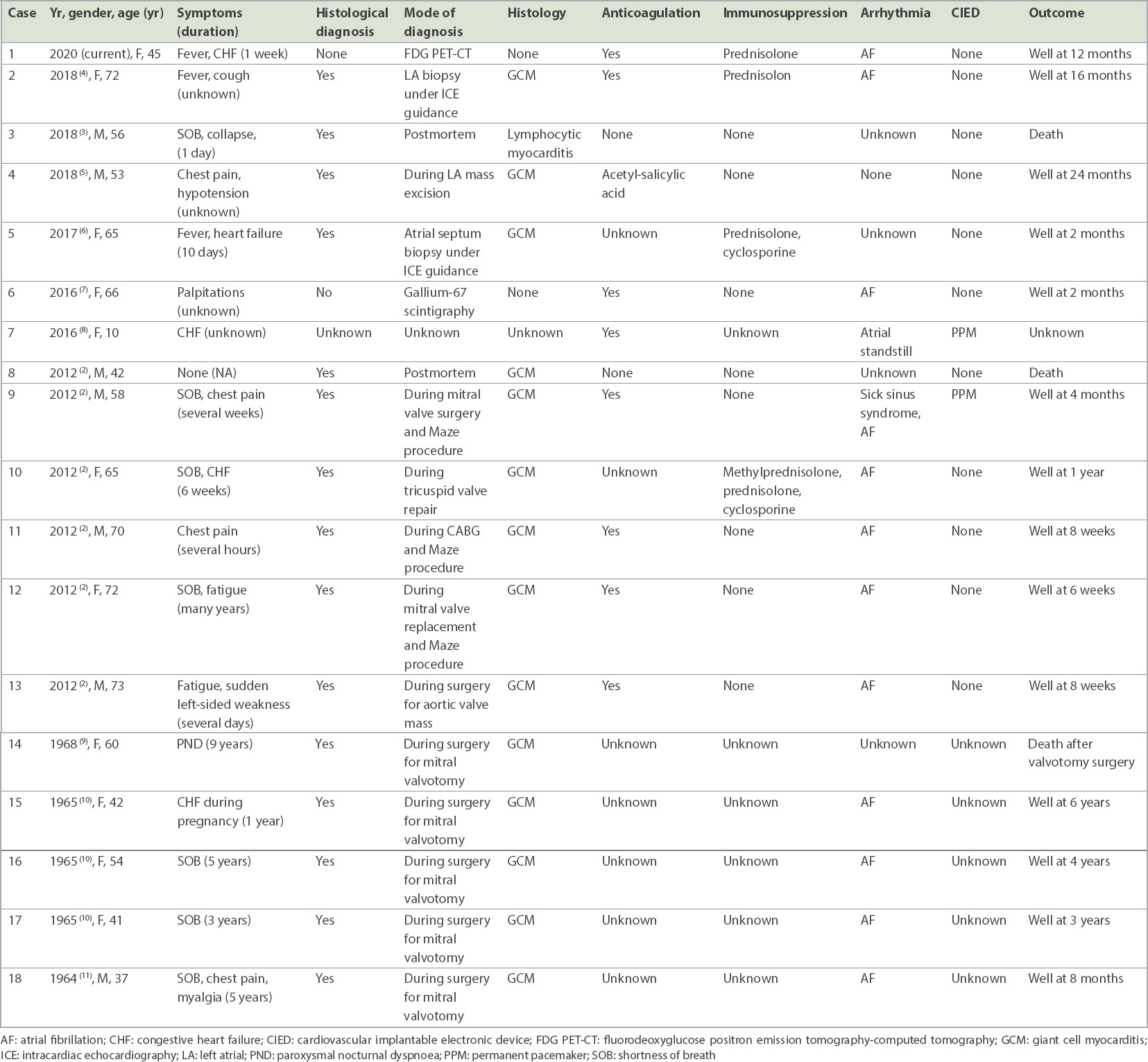
Most patients with isolated atrial myocarditis present with non-specific symptoms such as fever, breathlessness and mild fluid overload, or are incidentally diagnosed when they undergo atrial fibrillation ablation or cardiac surgery, or on postmortem.(2,4) Atrial myocarditis is usually associated with atrial arrhythmias, most commonly atrial fibrillation.(2) Atrial dysfunction increases the risk of thrombosis and cerebrovascular embolic events, which may be mitigated by anticoagulation. In some cases, persistent atrial standstill can also occur, requiring permanent pacemaker implantation.(8) Our patient was found to have significant sinus pauses with infrequent episodes of fleeting dizziness during admission. In acute management of sinus node dysfunction, temporary pacing is indicated in patients with severe symptoms and hemodynamic compromise.(12) However, the patient declined temporary insertion of transvenous pacemaker. Eventually, the sinus pauses resolved, converted to atrial fibrillation and flutter, and subsequently reverted to sinus rhythm following steroid therapy.
A definitive diagnosis requires histological confirmation via an atrial wall biopsy. Histological diagnosis is also important to guide the choice and duration of immunosuppression. Endomyocardial biopsy typically involves biopsy of the interventricular septum from the right ventricle via a transjugular venous approach. In our patient, this would require biopsy of the thinner interatrial septum under transoesophageal or intracardiac echocardiography guidance, a higher-risk procedure, which the patient declined. An alternative approach would be via surgical resection of the left atrial appendage.
Diagnosis of atrial myocarditis using transoesophageal echocardiography, gallium-67 scintigraphy, FDG PET-CT or cardiac MR imaging has been described.(2,6,7) In our patient, a markedly thickened left atrial wall could be observed on echocardiography early in the disease. This finding corroborated with subsequent chest CT, FDG PET-CT and cardiac MR imaging. Cardiac MR imaging and FDG PET-CT have complementary roles in the assessment of suspected myocardial inflammatory disease. We opted for FDG PET-CT as the initial imaging for diagnosis, assessment of overall disease involvement and localising extra-cardiac involvement that was amenable to biopsy. Unfortunately, the FDG PET-CT only revealed isolated increased left atrial wall FDG uptake, without any conclusive evidence of extracardiac involvement. No abnormal FDG uptake was observed at the right atrial or both ventricular walls. Subsequently, cardiac MR imaging was performed 20 days after FDG PET-CT, which showed patchy LGE at the left atrial wall without any definite abnormality in the right atrium, interventricular septum, right ventricle or left ventricle. Both FDG PET-CT and cardiac MR imaging revealed an isolated left atrial abnormality, which was related to an inflammatory process. The manifestation of sinus pauses indicated the involvement of the right atrium, which is not uncommon in previously reported cases. However, the degree of inflammation and extent of involvement of the right atrium may have been too subtle to be detected on both imaging modalities in our patient.
Immunosuppressive therapy in patients presenting with viral myocarditis remains controversial and is, therefore, not routinely used.(13) Combination immunosuppression with drugs such as cyclosporine or azathioprine is, however, recommended if the underlying aetiology is giant cell myocarditis.(14) Nevertheless, the duration of immunosuppressant therapy for isolated atrial myocarditis is presently unclear. The prognosis of atrial giant cell myocarditis appears to be much better than that observed in ventricular giant cell myocarditis, while the natural course and pathogenesis also seem to be substantially different.(2) In the Mayo Clinic case series of atrial giant cell myocarditis, only one out of six patients received long-term immunosuppression. In our patient, who had Type 1 diabetes mellitus, a high dose of a potent corticosteroid such as methylprednisolone would also risk derangement of her blood sugar control, thereby increasing the risk of secondary bacterial infection. We were also concerned about the risks of starting combination immunosuppression for her without histological confirmation. Finally, in view of the cardiac rhythm disturbance and atrial involvement, we successfully treated our patient by starting with a pulse of oral prednisolone on Day 9 of admission, with gradual tapering over six months.
There is no recommended method of monitoring response to therapy. The serum troponin level of our patient was only mildly elevated on initial presentation and normalised quickly with steroid therapy. NT-proBNP appeared to be a useful biomarker to track her response over time. BNP is produced mainly by the ventricles but also, to a smaller degree, from the atria. NT-proBNP level was likely elevated owing to the atrial inflammation. It could also be attributable to stiff left atrial syndrome resulting from reduced left atrial compliance, pulmonary venous hypertension and manifestation of fluid overload.(15)
In summary, isolated atrial myocarditis is an uncommon presentation of acute myocarditis and is associated with supraventricular tachyarrhythmias and bradyarrhythmias. Based on the limited available literature, its clinical course and prognosis also appear different and possibly more favourable than those of ventricular myocarditis. In the absence of definitive histological diagnosis, imaging with echocardiography, FDG PET-CT or cardiac MR imaging may be useful to aid diagnosis. Our patient responded well to steroid therapy, and we report a good short-to-intermediate-term prognosis. The benefits of long-term immunosuppressive therapy remain unclear and warrant further investigation.


