Abstract
INTRODUCTION
ADAMTS13 (a disintegrin-like and metalloproteinase with a thrombospondin Type 1 motif, member 13) plays a fundamental role in the regulation of haemostasis and thrombosis. Its deficiency leads to an accumulation of ultra-large von Willebrand multimers, inducing spontaneous platelet aggregation, thrombosis in the microvasculature, and thrombotic thrombocytopenic purpura (TTP), a condition with 90% mortality when left untreated. Prompt quantification of ADAMTS13 antigen, activity and autoantibody plays a crucial role in the diagnosis and management of TTP and can help differentiate it from other thrombotic microangiopathies (TMAs). Reference ranges for ADAMTS13 are generally derived from Caucasian patients. Given that polymorphism in the ADAMTS13 gene can be associated with variable ADAMTS13 levels, we aimed to establish the first reference range in Singapore and provide a crucial laboratory test for institutions here and elsewhere.
METHODS
150 healthy voluntary donors (75 men, 75 women) aged 21–60 years, with an ethnic mix mirroring Singapore’s population profile, were recruited. ADAMTS13 antigen, activity and autoantibody levels were measured using the fluorescence resonance energy transfer-vWF73 and enzyme-linked immunosorbent assay methodologies.
RESULTS
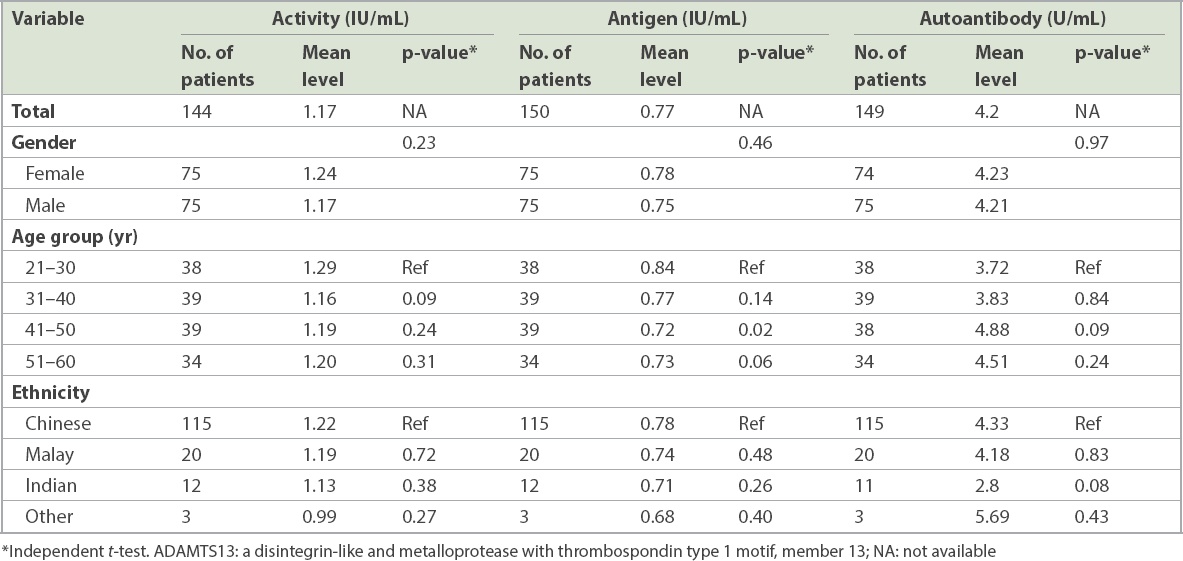
Levels (activity 0.65–1.79 IU/mL, antigen 0.36–1.17 IU/mL, autoantibody 1.4–12.5 U/mL) were not statistically different between the genders and various age groups.
CONCLUSION
TTP and TMAs are encountered in a wide range of specialties. The availability of new assays in Singapore will aid clinicians in the timely management of these conditions. Standardising reference ranges established for Singapore against World Health Organization standards allows harmonisation of measurements between laboratories and for future research collaborations.
INTRODUCTION
ADAMTS13 (a disintegrin-like and metalloproteinase with a thrombospondin Type 1 motif, member 13) is an essential protease that regulates the size of ultra-large von Willebrand factor (UL-vWF) by cleavage at the Tyr1605-Met1606 bond in the A2 domain.(1) Under conditions of shear stress in the presence of ADAMTS13 deficiency, UL-vWF multimers accumulate, which, in turn, induce spontaneous platelet aggregation and thrombi formation in the microvasculature, especially in vital organs, leading to thrombotic thrombocytopenic purpura (TTP).
Up to 20% of patients with TTP present with neurological complications,(2) 11% of patients have acute and/or chronic renal failure,(3) and patients can experience sudden cardiac death secondary to myocardial infarction, heart failure and arrhythmias.(4) Without treatment, TTP is associated with a mortality rate of about 90%, with a majority of deaths occurring within the first 24 hours of presentation.
TTP is a rare disorder, with a reported annual incidence of six per million population.(5) Congenital TTP (or Upshaw-Schulman syndrome) is due to an inherited deficiency of ADAMTS13, while acquired TTP results from a deficiency of ADAMTS13 secondary to anti-ADAMTS13 antibodies. The ability to quantify ADAMTS13 activity and autoantibody levels is critical for differentiating inherited TTP from acquired TTP. It would also help to differentiate TTP from other thrombotic microangiopathies such as haemolytic uraemic syndrome (HUS), atypical HUS, disseminated intravascular coagulation, HELLP (haemolysis, elevated liver enzymes, low platelet) syndrome/pre-eclampsia, malignant hypertension,(6) Kasabach-Merritt syndrome, paroxysmal nocturnal haemoglobinuria and catastrophic antiphospholipid syndrome. The absence of ADAMTS13 autoantibodies in a patient with < 10% ADAMTS13 activity differentiates congenital TTP from acquired TTP, as 94%–97% of patients suffer from idiopathic TTP and have severe ADAMTS13 deficiency but will have circulating anti-ADAMTS13 antibodies in the plasma.(7) This undoubtedly affects the modalities of patient management, as congenital TTP is treated with regular infusion of plasma and, potentially, the novel therapy BAX 930/SHP655, a recombinant ADAMTS13,(8,9) which is still in clinical trials. On the other hand, the treatment for acquired TTP requires plasma exchange, corticosteroids and/or rituximab (anti-CD20 monoclonal antibody).
Up to 75%–90% of patients with TTP would have very low ADAMTS13 activity level < 10%, a reading that has 90% sensitivity and specificity. In two more recent studies, ADAMTS13 activity cut-off < 10% was found in more than 97.5%(10) and 100%(11) of patients with TTP. The median ADAMTS13 activity of patients with thrombotic microangiopathies in the United Kingdom TTP registry, on the other hand, ranges from 56% to 66%.(5) However, it should be stated that up to 14% of patients with HUS(12) and up to 10% of patients with severe Child-Pugh C liver cirrhosis can have ADAMTS13 activity level < 10%.(13)
ADAMTS13 activity and autoantibody levels can provide prognostic information in acute TTP. Patients with activity level < 10% are more likely to respond to plasma exchange, have higher remission rate (> 80% vs. 20%–75% in three different studies(14-16)) and lower mortality rate (16% vs. 45%(14)).(17) A prospective study conducted by Ferrari et al found the persistence of undetectable activity in remission to be predictive of relapse within 18 months, with a positive predictive value of 38.5%. In contrast, the negative predictive value for relapse of measurable ADAMTS13 activity in remission was 94.7%.(18,19)
Despite appropriate treatment, the mortality of patients with TTP still ranges between 15% and 20%.(5) Around 30% (in certain studies, up to 60%(20)) of patients who achieve remission would also relapse.(20) Persistence of ADAMTS13 autoantibodies during remission predicts recurrence,(21,22) with a threefold increase in relapse rate compared with those without detectable autoantibodies.(21) However, the timing of relapse is unpredictable, as demonstrated in one study, where a wide range was noted (duration 0.3–9.5 years).(23) Hence, although the ADAMTS13 assay cannot inform clinicians about the timing to commence pre-emptive treatment, it would strongly support the need for closer monitoring than in patients without persistent autoantibodies and low activity during clinical remission. The persistence of low ADAMTS13 activity level and elevated autoantibodies during acute TTP treatment, on the other hand, would suggest the need to intensify plasma exchange and/or institute additional therapy, such as rituximab.(24)
There is no universally agreed normal or reference range for ADAMTS13 activity. Variations exist depending on the assay methodology, age, gender and genetic variants in different ethnic populations.(25-27) In general, normal ADAMTS13 activity for healthy adults ranges between 50% and 160%. For example, the Mayo Clinic, USA, accepts an activity level ≥ 70% to be normal.(28) However, in parts of Europe,(29) the lower limit of reference range for ADAMTS13 activity can be as low as 40%. Single-nucleotide polymorphisms, for example P.Ala732Val, can be associated with a decrease of over 20% in ADAMTS13 activity. The P.Pro475Ser polymorphism, which is restricted to the Eastern Asian population, is heterozygous in approximately 10% of the Japanese population and associated with a reduction in ADAMTS13 activity level.(30) Feys et al reported that normal Chinese donors have significantly lower ADAMTS13 antigen levels than Caucasian donors.(31) It was not recommended to adopt the manufacturer’s ADAMTS13 reference ranges for our laboratory, as the manufacturer used a different fluorescence reader and wavelengths (BioTek FL × 800 TBI, BioTek Instruments Inc, Winooski, VT, USA), with excitation at 340 nm and emission at 450 nm. Our study, meanwhile, used Tecan Infinite F200 Pro microplate reader (Tecan Austria GmbH, Grodig, Austria), with excitation at 340 nm and emission at 465 nm. Together with the variation of ADAMTS13 among different ethnic groups, there was a need to establish the first ADAMTS13 reference range for adults in Singapore.
METHODS
This prospective study recruited 150 healthy adult volunteers in Singapore. Ethics approval from the domain specific review board and informed consent of all participants were obtained prior to the study.
Blood (2 × 2.7 mL citrate tube and 1 × 3 mL EDTA [ethylenediaminetetraacetic acid] tubes) samples were collected from each volunteer. The equipment consisted of the Tecan Infinite F200 Pro microplate reader for fluorescent resonance energy transfer [FRET]-vWF73 and enzyme-linked immunosorbent assay [ELISA], Technozym® Fluorogenic ELISA kit, Technozym® ADAMTS-13 INH ELISA kit and the World Health Organization (WHO) First International Standard ADAMTS13 plasma (NIBSC code 12/252). Platelet-poor citrated plasma was prepared by centrifugation; the ADAMTS13 antigen and activity were measured using the FRET methodology, while human autoantibodies (immunoglobulin G [IgG]) in serum or plasma against ADAMTS13 were measured using ELISA. Full blood count was performed using the EDTA specimens. Platelet count was checked to be within the local reference interval.
For antigen and activity measurements, patients’ samples, controls and calibrators were incubated with monoclonal anti-ADAMTS13 antibody coated on the wells of a microplate. The ADAMTS13 present was bound to the antibody. After washing, a fluorogenic substrate vWF-fragment (FRETS-vWF73) was added for activity measurement. Fluorescence generated by the cleavage of fluorogenic substrate was measured kinetically at 360 nm/460 nm for 15 minutes using the Tecan Infinite Series microplate reader. After measuring the ADAMTS13 activity, the microplate was washed. The conjugate, which contained anti-ADAMTS13 POX, was then added. This conjugate would then bind to the ADAMTS13 present. Excess conjugate was washed off and antigen substrate was added for colour development. Stop solution was then added to end the reaction after 15 minutes and the endpoint was read at 360 nm/460 nm. Patients’ results were calculated from the reference curve generated. All calibrator values were calibrated against and traceable to the WHO First International Standard ADAMTS-13 plasma (12/252).
For autoantibody measurement, diluted samples, controls and calibrators were incubated with recombinant ADAMTS13 protease coated on the wells of a microplate. If autoantibodies were present in the sample, they would be bound to ADAMTS13 protease during incubation. After washing, antihuman IgG POX was added. This antibody binds to the ADAMTS13 autoantibodies. After incubation and washing, chromogenic substrate was added, which would react with the horseradish peroxidase of the antihuman IgG POX. After ten minutes of incubation, the reaction was stopped by adding stop solution. The intensity of colour was measured with Tecan Infinite Series microplate reader at 450 nm. A reference curve with point-to-point fit was set up to read the patient’s result, where the X-axis represented concentration of ADAMTS13 IgG (U/mL) and Y-axis represented extinction at 450 nm. Statistical software, including Microsoft Excel and Analyse-it, were used.
RESULTS
The 150 (75 men, 75 non-pregnant women) healthy adult volunteers recruited were aged 21–60 years. The ethnic mix of donors mirrored the ethnic population profile of Singapore (115 Chinese, 20 Malay, 12 Indian, 3 Other). The donors were categorised into different age groups: 21–30 years; 31–40 years; 41–50 years; and 51–60 years. There was an equal number of male and female donors in each age group.
As the reference range for ADAMTS13 activity was 0.65–1.79 IU/mL, six of our 150 donors whose results exceeded the measurement range were excluded. Similarly, as the reference range for ADAMTS13 antigen was 0.36–1.17 IU/mL, two donors whose results exceeded the measurement range were excluded. Likewise, as the reference range for ADAMTS13 autoantibodies was 1.4–12.5 U/mL, one donor who was an outlier according to Dixon’s rule was excluded. Samples with levels > 15 U/mL were considered as positive for autoantibodies. Anti-ADAMTS13 antibody below the detection limits was reported as < 2 U/mL.
There were no statistical differences, according to independent t-tests, for ADAMTS13 antigen (
Fig. 1
Charts show ADAMTS13 activity and antigen levels according to (a) age group and (b) ethnicity. ADAMTS13: a disintegrin-like and metalloprotease with thrombospondin Type 1 motif, member 13
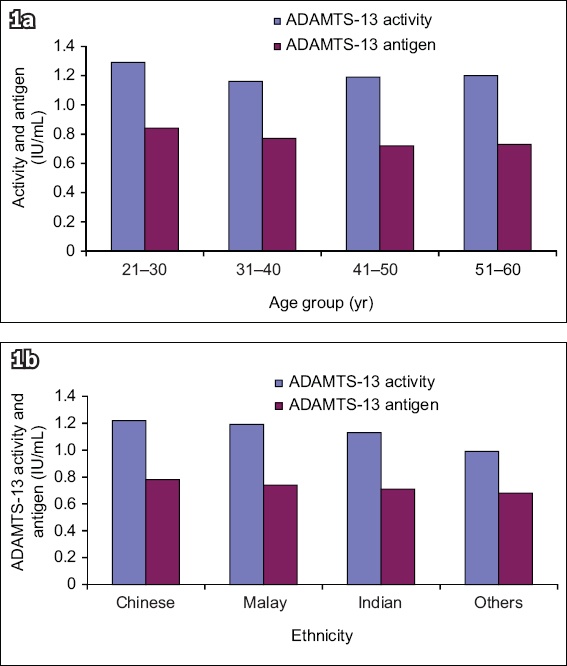
Fig. 2
Charts show ADAMTS13 autoantibody levels according to (a) age group and (b) ethnicity. ADAMTS13: a disintegrin-like and metalloprotease with thrombospondin Type 1 motif, member 13
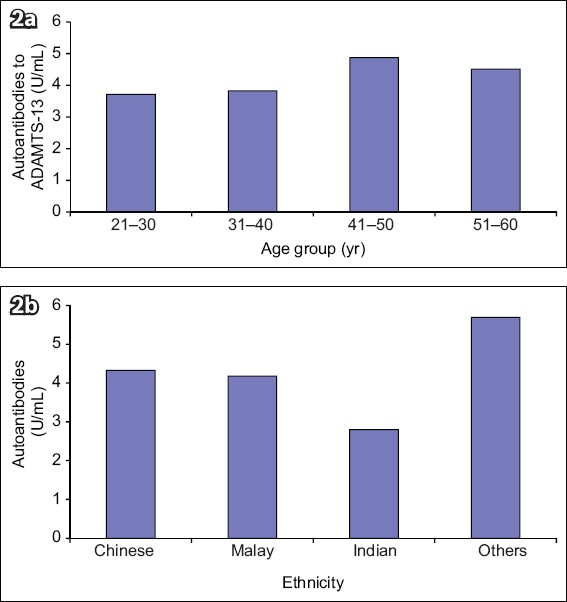
Table I
ADAMTS13 antigen, activity and autoantibody levels according to gender, age group and ethnicity.
DISCUSSION
Historically, in Singapore, the diagnosis of TTP has been based on clinical features supported by laboratory findings of thrombocytopaenia and peripheral blood film features of microangiopathic haemolytic anaemia. ADAMTS13 activity and autoantibodies, if obtained, are typically sent to the Mayo Clinic, USA, for analysis, but with long turnaround times of over two weeks. As the presenting features of TTP are often indistinguishable from those of other conditions, therapy for TTP, including plasmapheresis and corticosteroids that are not without risks, is often initiated as emergency treatment for suspected patients. With a turnaround time of ≤ 2 working days, the availability of this ADAMTS13 laboratory service in Singapore would have a significant impact on the clinical management of suspected TTP, providing clinicians the support to stop unnecessary treatment for patients for whom there is low clinical suspicion of TTP but who have nevertheless been commenced on plasma exchange, thereby reducing both risk to patients as well as healthcare expenditure. It can also be used to monitor the response to therapy and help predict relapses, especially for patients with chronic relapsing TTP. Furthermore, our reference range was standardised against WHO standard plasma, allowing reporting in international units (IU/mL), which is now encouraged by the WHO, as part of harmonisation of results across laboratories worldwide.
There are two types of ADAMTS13 inhibitors – neutralising antibodies (approximately two-thirds of patients) and non-neutralising antibodies (approximately one-third of patients). While neutralising antibodies block the substrate-cleaving activity, non-neutralising antibodies reduce the half-life of ADAMTS13 by enhancing antibody-mediated clearance. The advantage of our choice of assay (ELISA) to detect autoantibodies, over the traditional Bethesda assay utilised by some external reference laboratories, is the enhanced sensitivity that ELISA provides, as the Bethesda assay primarily detects only neutralising antibodies (seen in approximately two-thirds of patients) and cannot detect antibody titres < 0.5 BU (Bethesda units). In contrast, the ELISA methodology detects both types of inhibitors, thereby improving the sensitivity of the test as well as the diagnosis of TTP. One of the limitations of our assay was that samples with high concentrations of antibodies other than anti-ADAMTS13 antibodies might result in weak positive or borderline results.
Where the FRET assay is concerned, ADAMTS13 activity can be artificially low owing to high bilirubin (> 15 mg/dL) as well as haemolysed plasma containing free haemoglobin (> 2 g/dL). It is important to note the timing of the blood drawn in relation to whether the patient has already received plasma infusion or undergone plasma exchange, as the ADAMTS13 activity level would then be elevated. Activity can also be affected by high levels of endogenous vWF, the presence of heparin, fondaparinux, dabigatran or other direct thrombin inhibitors, and hyperlipidaemia.
As age can affect the ADAMTS13 activity by up to 3.9%–6.5%, the reference range established for healthy adults in Singapore may not be applicable to the paediatric population, despite our range being comparable to those used by paediatric centres in the USA.(32,33) Establishing a reference range of ADAMTS13 for the Singapore paediatric population is, therefore, warranted. Similarly, as genetic polymorphism can affect ADAMTS13 activity levels by up to 20%, we may not be able to confidently presume that the same cut-off values of ADAMTS13 activity in diseased states, as quoted in the guidelines, would apply to patients of Asian origin. Studying the pathological levels of ADAMTS13 in Singapore and Asian patients may inform practice guidelines and influence the clinical management of these patients.
ACKNOWLEDGEMENTS
The authors thank the National Healthcare Group, Singapore, for its generous and unconditional support of this study through the awards of the Small Innovation Grant and Pitch-For-Fund Grant in 2015 and 2016, respectively.
About the First Author

Dr Allison Ching Yee Tso is a Senior Consultant Haematologist and Adjunct Assistant Professor at Nanyang Technological University’s Lee Kong Chian School of Medicine, and Senior Clinical Lecturer at NUS Yong Loo Lin School of Medicine, Singapore. Dr Tso graduated from the University of Oxford and was trained in London. She is a Fellow of the Royal Colleges of Physicians of London and Edinburgh and a Fellow of the Royal College of Pathologists (United Kingdom). Her research interests include thrombotic microangiopathies, myeloma, haemophagocytic lymphohistiocytosis and haemoglobinopathies.


