Abstract
INTRODUCTION
Children with an anterior mediastinal mass (AMM) need general anaesthesia (GA) or deep sedation for diagnostic procedures more often than adult patients. Anaesthetic management to prevent such complications includes maintenance of spontaneous ventilation (SV) and prebiopsy corticosteroids/radiotherapy.
METHODS
We reviewed the medical records of children with AMM who were brought to the operating theatre for diagnostic procedures (prior to chemotherapy) between 2001 and 2013. Our aim was to describe the clinical features, radiological findings and anaesthetic management, as well as determine any association with complications.
RESULTS
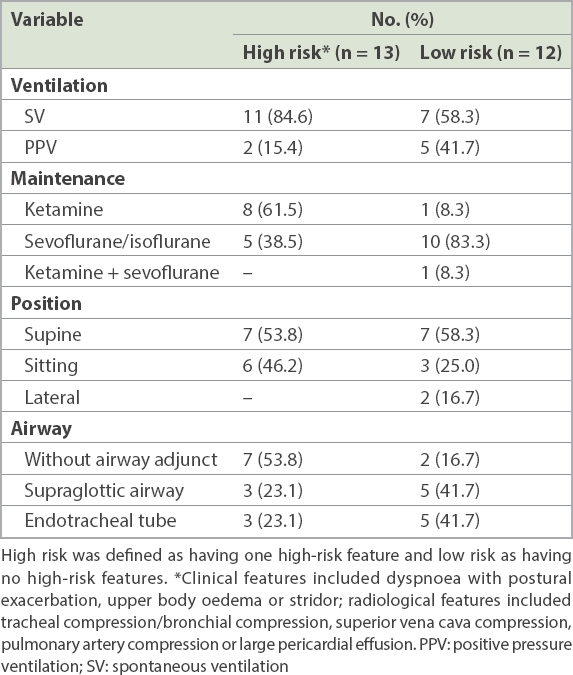
25 patients (age range 10 months–14 years) were identified during the study period. Corticosteroid therapy was started before the biopsy for one patient. All 25 patients had GA/sedation. A senior paediatric anaesthesiologist was involved in all procedures. Among 13 high-risk patients, SV was maintained in 11 (84.6%) patients, ketamine was used as the main anaesthetic in 8 (61.5%) patients, 6 (46.2%) patients were in a sitting position and no airway adjunct was used for 7 (53.8%) patients. There were 3 (12.0%) minor complications.
CONCLUSION
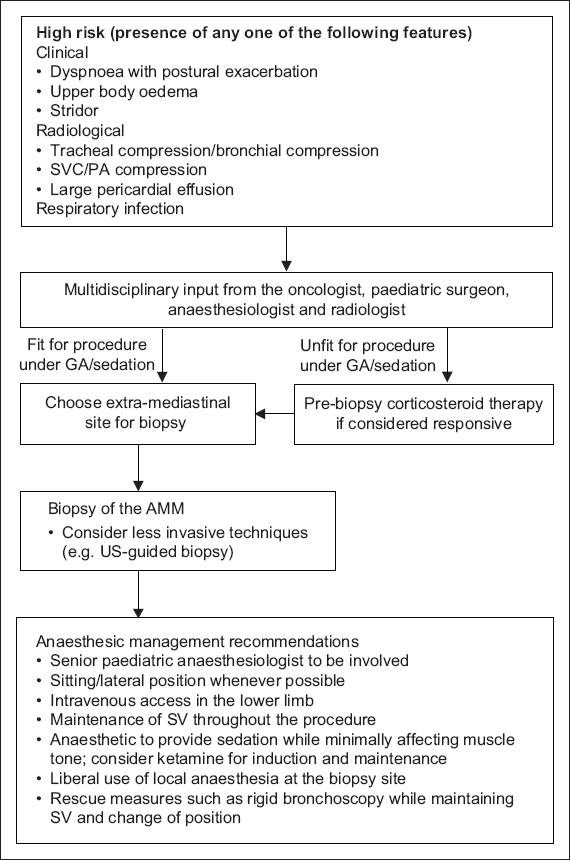
Based on our results, we propose a simplified workflow, wherein airway compression of any degree is considered high risk. For patients with high-risk features, multidisciplinary input should be sought to decide whether the child would be fit for a procedure under GA/sedation or considered unfit for any procedure. Recommendations include the use of less invasive methods, involving experienced anaesthesiologists to plan the anaesthetic technique and maintaining SV.
INTRODUCTION
Children with an anterior mediastinal mass (AMM) need general anaesthesia (GA) or deep sedation for diagnostic procedures more often than adult patients. Anaesthesia-related morbidity and mortality have been associated with the presence of orthopnoea, upper body oedema, stridor, major vessel compression, tracheal/bronchial compression, dynamic compression on spirometry and large pericardial effusion.(1-8) Anaesthetic management to prevent such complications includes the maintenance of spontaneous ventilation (SV) and prebiopsy corticosteroids/radiotherapy.(1,4-11)
We reviewed the medical records of children with AMM who were at the operating theatre for diagnostic procedures. Our aim was to describe the clinical features, radiological findings and anaesthetic management methods used for these patients as well as to determine whether there was any association with complications.
METHODS
Following approval from the institutional review board, retrospective case note evaluation was done of children diagnosed with AMM between 2001 and 2013 at KK Women’s and Children’s Hospital, Singapore. We included all children who had undergone diagnostic procedures in the operating theatre while excluding children who had already received chemotherapy other than corticosteroid therapy. Information on the demographic characteristics, diagnosis, clinical presentation, computed tomography/magnetic resonance imaging findings, anaesthetic management and perioperative critical incidents was recorded.
Anaesthetic management was analysed based on the type of ventilation maintained, anaesthetic agents used and the risk status of the patient. Patients who presented with upper body oedema, stridor, major vessel compression, tracheal/bronchial compression and large pericardial effusion were considered high risk.(2,6-8) As the term ‘orthopnoea’ was not consistently used in the patients’ case notes, all patients who presented with dyspnoea with postural exacerbation were considered to be at high risk.
RESULTS
Within the study period, 25 patients with AMM, aged between 10 months and 14 years, were identified. There were 14 (56.0%) boys and 11 (44.0%) girls. The various diagnoses included lymphoma in 21 (84.0%) patients, and neuroblastoma, ganglioblastoma, malignant rhabdoid tumour and mucoepidermoid carcinoma in 1 (4.0%) patient each. 6 (24.0%) patients had airway compression at the tracheal or bronchial level (
Table I
Relationship between radiological features of cardiorespiratory involvement on CT/MR imaging and preoperative symptoms suggestive of mass effect among patients with anterior mediastinal mass (n = 25).
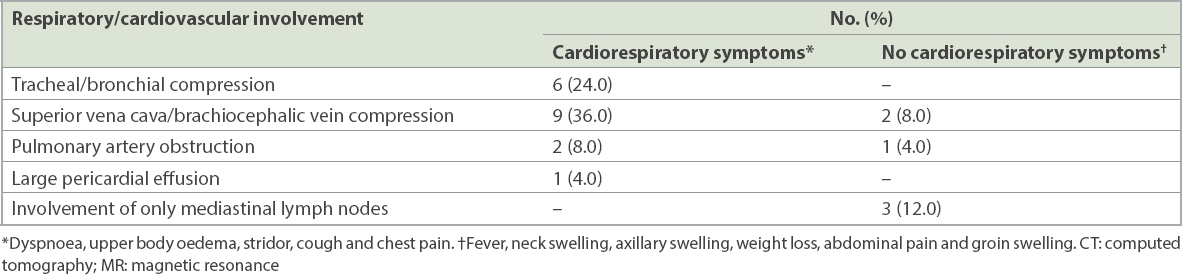
2 (8.0%) patients received corticosteroids to reduce mass size before surgery. One of them had been diagnosed at another hospital and had been administered prednisolone prior to transfer to our centre.
All procedures were performed under GA or sedation. A senior paediatric anaesthesiologist (with over five years’ experience) was in charge in the operating theatre for all procedures. For 6 (24.0%) patients, the biopsy sample was taken from extramediastinal sites; for 3 (12.0%) patients, ultrasonography-guided needle biopsy of the AMM was performed; and for 16 (64.0%) patients, open/thoracoscopic biopsy of the AMM was performed.
In our study, 13 (52.0%) patients were considered as high risk (
Table II
Anaesthetic management of children considered high risk and those without any high-risk features (n = 25).
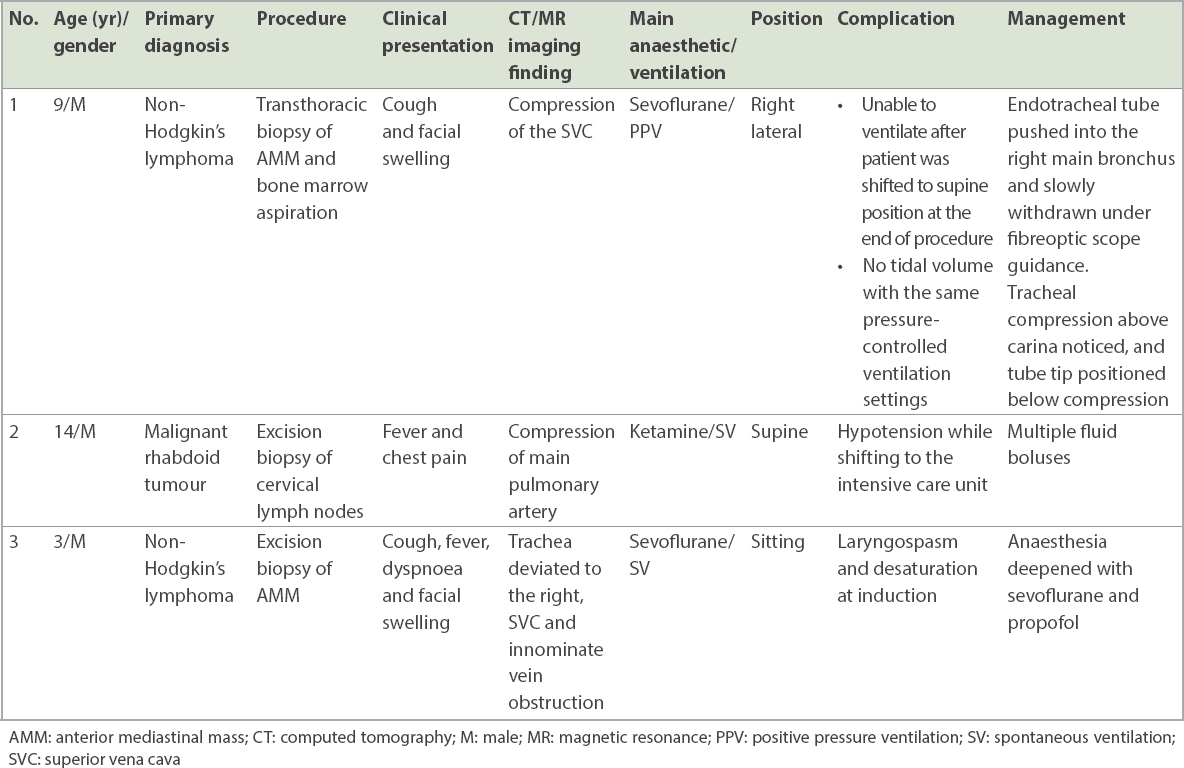
There were 3 (12.0%) complications – respiratory events in two patients and a cardiovascular event in one patient – all of which occurred in high-risk patients (
Table III
Details of patients with anaesthetic complications.
DISCUSSION
The absence of cardiorespiratory symptoms in our patients did not necessarily imply low risk (
Corticosteroids or radiotherapy before biopsy can considerably reduce the size of the mass and thereby decrease the risk of GA or sedation, but it may also preclude the biopsy of AMM and thereby hinder accurate diagnosis.(2,10,12) Corticosteroid therapy was started before biopsy in one patient at our hospital, and the procedure was done after the risk was deemed to be lowered. This 11-year-old patient was only able to assume the right lateral decubitus position, and computed tomography showed that AMM was causing compression of the trachea and narrowing of the left and right main bronchi, superior vena cava and main pulmonary artery. After multidisciplinary input from oncologists, surgeons, radiologists and anaesthesiologists, the patient was considered unfit to undergo biopsy of AMM due to a combination of cardiorespiratory symptoms and radiological findings.
It has been suggested that a tracheal cross-sectional area or peak expiratory flow rate less than 50% of the predicted value based on age and gender indicates that a patient is unfit for GA.(1,10,13) The calculation for tracheal cross-sectional area compression was not done for any patient in our study, and neither was any test for dynamic compression, such as peak expiratory flow rate. Testing for peak expiratory flow rate in children and obtaining reliable readings is difficult and is thus not used to assess the risk associated with GA at our hospital.(14,15)
Biopsy in high-risk patients under local anaesthesia, with or without conscious sedation, has been reported, but it is not clear whether all such patients received an anaesthetic intervention.(5,15) In our series, all patients did receive anaesthetic intervention – possibly because a senior paediatric anaesthesiologist was in charge of anaesthetics in the operating theatre for all our patients.
For high-risk patients with AMM, maintaining SV is important, as the negative intrathoracic pressure during inhalation pulls the mass away from the airway and major vessels. Positive pressure ventilation may cause turbulence and ineffective gas exchange in the airways compressed and narrowed by the AMM.(4,8) Both inhalation anaesthetic agents (e.g. sevoflurane and isoflurane) and the intravenous anaesthetic agent ketamine can be used to anaesthetise patients while maintaining SV. Ketamine maintains intercostal and chest wall tone better than inhalational agents; as a result, it does not cause atelectasis in spontaneously breathing patients.(8,11,16) The increase in the use of ketamine in the later years of our study was probably due to greater awareness of its advantages. Assuming the sitting or lateral position during the diagnostic procedure may be beneficial, as in the supine position, gravity pushes the mass onto the airways and major vessels, and also reduces the intrathoracic space (cephalad movement of the diaphragm and increased central thoracic volume).(4,13) The choice of anaesthetic and positioning seemed to have been based on the risk status of patients in our study. For high-risk patients, ketamine was more frequently used to maintain anaesthesia/sedation (8/13 patients, 61.5%), patients were more likely to be positioned in the sitting position (6/13 patients, 46.2%) and fewer patients needed an airway adjunct (7/13 patients, 53.8%). However, such a correlation could not be shown because of our small sample size.
The complications in the present series of patients were minor and comparable to those reported in other series.(1,10,14,15) Complications in our study highlight the delicate balance needed for anaesthetic management. In the case of the first patient who developed complication, when the patient was placed in a supine position after biopsy was done in the right lateral position, there was complete airway compression. It is possible that the use of the inhalational agent (sevoflurane), positive pressure ventilation and oedema/bleeding into the mass due to the biopsy may all have contributed to this.
Patients for whom AMM compresses the major vessels may be relatively asymptomatic while awake but on loss of muscle tone after administration of an anaesthetic agent, may develop severe life-threatening hypotension due to complete compression and resultant interruption of cardiac output.(8) The second patient who developed complication had compression of the pulmonary artery by AMM. This patient was in a sitting position throughout the procedure and anaesthetic management was done with ketamine, which preserves muscle tone. Although there was significant hypotension at the end of the procedure, it was corrected with fluid boluses and there was no further deterioration in the patient. Respiratory infection contributes to the risks associated with GA in AMM.(3) Pulmonary consolidation/collapse can cause ventilation-perfusion mismatch and slower induction with less soluble inhalational agents, such as sevoflurane. The slower inhalational induction may have resulted in a lighter plane of anaesthesia during airway intervention and subsequent laryngospasm, which was the complication seen in the third patient in our study.
Based on our results, we propose a simplified (albeit conservative) workflow that can be applied to children with AMM undergoing diagnostic procedures to enable safe anaesthetic intervention (
Fig. 1
Flowchart shows risk stratification and anaesthetic management of children with AMM. AMM: anterior mediastinal mass; GA: general anaesthesia; LA: local anaesthesia; PA: pulmonary artery; PPV: positive pressure ventilation; SV: spontaneous ventilation; SVC: superior vena cava; US: ultrasonography