Abstract
Haemoptysis is commonly seen in the healthcare setting. It can lead to life-threatening complications and therefore requires careful evaluation of the severity and status of the patient. Common causes of haemoptysis can be broadly grouped into five main categories: infective, neoplastic, vascular, autoimmune and drug-related. Detailed history-taking and careful physical examination are necessary to provide a diagnosis and assess the patient’s haemodynamic status. Physicians must have a clear understanding of the criteria for further investigations and the need for a specialist or inpatient referral for management.
Mrs Lim, a 55-year-old homemaker whom you last saw two years before, visited your clinic complaining of blood in her sputum when she clears her throat. She coughed up approximately 10 mL of bright red blood in the morning on the day of her visit. She had no complaints of chest pain or shortness of breath. Mrs Lim has a past medical history of hypertension and was diagnosed with type 2 diabetes mellitus during her recent hospital admission for acute appendicitis. She does not smoke and has no family history of lung disease.
WHAT IS HAEMOPTYSIS?
Haemoptysis is defined as the expectoration of blood originating from the tracheobronchial tree or pulmonary parenchyma.(1) It can be classified as massive or non-massive based on the volume lost. Massive haemoptysis has been described as blood loss ranging from 100–600 mL. However, for the purpose of this article, massive haemoptysis is defined as loss of at least 200 mL of blood in 24 hours or 50 mL per episode.(2,3) Although massive haemoptysis accounts for only 5% of haemoptysis cases, it is an alarming symptom with an associated mortality of more than 50%.(4)
HOW RELEVANT IS THIS TO MY PRACTICE?
Haemoptysis is a common presentation in the primary, secondary and tertiary care setting.(5) Hence, it is essential for primary care physicians to be well-versed in the evaluation and management of patients with haemoptysis. This article aimed to discuss the more common differential diagnosis, indications for referral to the emergency department or specialty clinics, and a general management plan for patients with non-massive haemoptysis.
WHAT CAN I DO IN MY PRACTICE?
In the primary care setting, it is important to differentiate between pseudohaemoptysis and haemoptysis. Thereafter, the patient’s clinical status needs to be assessed, as well as the quantity of blood lost. The physician should then attempt to identify the cause of the bleeding and determine if a specialist or inpatient referral is needed.
True haemoptysis versus pseudohaemoptysis
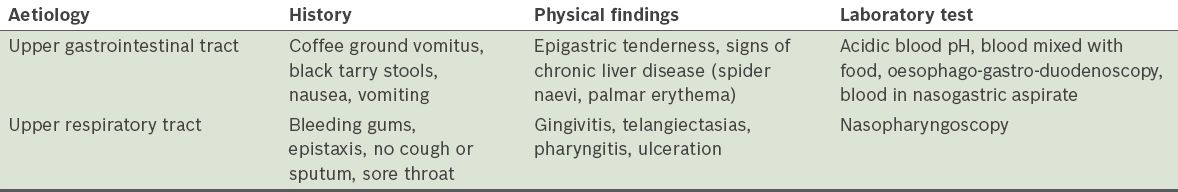
When evaluating a patient presenting with expectoration of blood, one must determine the source of the bleeding and determine whether the patient is presenting with true haemoptysis or pseudohaemoptysis. Aetiologies of pseudohaemoptysis include the upper gastrointestinal tract and upper respiratory tract (
Table I
Differentiating features of pseudohaemoptysis.
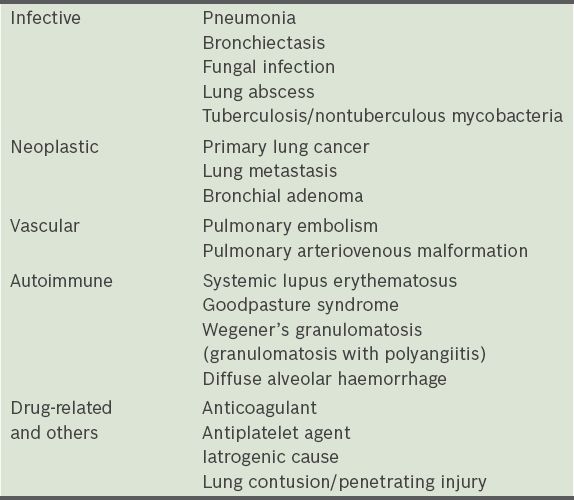
Differential diagnosis for haemoptysis
There are multiple causes of haemoptysis. This article focuses on the more common causes such as (a) infective; (b) neoplastic; (c) vascular; (d) autoimmune; and (e) drug-related. A more detailed differential list can be found in
Table II
Differentials for haemoptysis.
Infective causes
Pneumonia is a common cause of haemoptysis; in such patients, physicians must look for symptoms and signs of infection such as fever, chest pain and productive cough. It is also important to obtain any relevant contact and/or travel history. Tuberculosis is a common cause of massive haemoptysis, with an incidence rate of 38 per 100,000 population in 2015.(8) It is hence prudent for a practitioner to look for risk factors of tuberculosis. This includes immunosuppression, which may be secondary to immunosuppressive medications or conditions such as human immunodeficiency virus infection or diabetes mellitus.
Neoplastic causes
Neoplastic lesions that can cause haemoptysis can be divided into primary pulmonary lesions and metastatic lesions. If a malignant cause is suspected, physicians should consider a thorough history and examination to look for other primary malignancies such as history of breast, kidney, gastrointestinal, ovarian and cervical cancers.(9) It is important to also elicit constitutional symptoms such as loss of weight and appetite, and to consider the risk factors for primary lung carcinoma such as smoking and occupational exposure.
Vascular causes
Vascular causes of haemoptysis include pulmonary embolism. In patients with suspected pulmonary embolism, the physician should ask if they have a history of recent surgery or immobilisation.(10) The limbs should be assessed for any signs of deep vein thrombosis as well. Other signs and symptoms to look out for include dyspnoea, chest pain, tachypnoea and tachycardia.(11)
Autoimmune causes
Vasculitic rash, haematuria, joint pain or swelling may be suggestive of underlying autoimmune diseases such as Wegener’s granulomatosis (granulomatosis with polyangiitis), systemic lupus erythematosus (SLE) or Goodpasture syndrome.(12,13) The characteristic butterfly rash or other skin lesions such as alopecia in SLE may also be found.(14) If autoimmune disease is suspected, the patient should be referred to a specialist centre for further evaluation.
Drug-related causes
Common drugs that may cause haemoptysis include anticoagulants and antiplatelet agents. It is important to elicit further history (i.e. medication history) in order to predict the consequence of stopping medications as part of management.
HOW SHOULD I APPROACH HAEMOPTYSIS?
When approaching a patient with haemoptysis, a proper history has to be taken to narrow down the aetiology of the patient’s symptoms. The physician should first exclude the possibility of pseudohaemoptysis and then narrow down the groups of causes for true haemoptysis. It is also paramount to quantify the amount of blood loss and evaluate for any complications due to blood loss. This includes evaluation of signs and symptoms of anaemia and searching for signs of haemodynamic compromise. The patient’s vital signs should be measured and documented. Criteria for admission to the emergency department or referral to specialist clinics are as follows:(6,15) (a) high risk of massive bleed; (b) gas exchange abnormalities (respiratory rate > 30 breaths/minute, oxygen saturation < 88% room air); (c) haemodynamic instability (e.g. tachycardic, tachypnic, hypotensive patient); (d) other respiratory comorbidities (e.g. previous pneumonectomy, chronic obstructive pulmonary disease); and (e) other comorbidities (e.g. ischaemic heart disease, need for anticoagulants/antiplatelet agents).
Chest radiography is typically recommended for all patients who present with haemoptysis. It is a quick, readily available and cheap modality that can assist in revealing any focal or diffuse parenchymal involvement as well as pleural abnormalities.(4) Other diagnostic modalities include bronchoscopy, multidetector computed tomography (CT), multidetector CT angiography and digital subtraction angiography.
HOW SHOULD I MANAGE HAEMOPTYSIS?
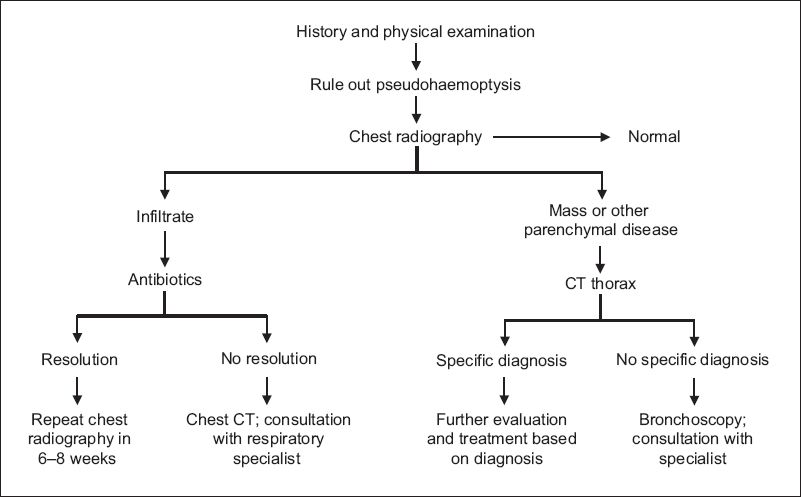
When a patient with non-massive haemoptysis has no haemodynamic instability, a normal chest radiograph and low risk of massive bleeding in the acute setting, no additional testing is required. In a patient with increased risk of malignancy (i.e. 30 pack-years of smoking, age ≥ 40 years), a referral for CT should be considered (
Fig. 1
Flowchart shows evaluation of non-massive haemoptysis. CT: computed tomography
On further questioning, you noted that Mrs Lim was recently started on aspirin following the diagnosis of type 2 diabetes mellitus. Chest radiography showed no abnormalities and her haemoptysis stopped once the aspirin was discontinued. She has been asymptomatic since then.
TAKE HOME MESSAGES
-
Haemoptysis is expectoration of blood originating from the tracheobronchial tree.
-
It is important to differentiate pseudohaemoptsysis from true haemoptysis.
-
Causes of haemoptysis can be divided into five broad categories: infective, neoplastic, vascular, autoimmune and drug-related.
-
Chest radiography is recommended as a first-line investigation for all patients complaining of haemoptysis.
-
Massive haemoptysis is defined as loss of at least 200 mL of blood in 24 hours or 50 mL per episode. It warrants an inpatient or specialist referral.
SMJ-57-418.pdf


