Abstract
The use of implantable cardioverter defibrillators (ICDs) in young women has been increasing in recent years owing to greater awareness about inherited cardiac conditions that increase the risk of sudden death. Traditional placement of ICDs in the infraclavicular region among young women often leads to visible scars, a constant prominence that causes irritation from purse or bra straps and can result in body image concerns and device-related emotional distress. In this case series, two women with long QT syndrome required placement of ICDs for prevention of sudden cardiac death. Submammary placement of ICDs was performed in collaboration with electrophysiologists. We describe our local experience and technique in submammary placement of ICDs as well as the challenges faced.
INTRODUCTION
The use of implantable cardioverter defibrillators (ICDs) has become the mainstay treatment for prevention of sudden cardiac death secondary to haemodynamically intolerable ventricular arrhythmias.(1,2) Traditional placement of ICDs in the infraclavicular region leads to visible scars and a constant prominence over the infraclavicular region. This will often lead to discomfort and irritation from purse or bra straps. Young women are particularly vulnerable to these negative effects of ICD placement. Numerous studies have shown that ICD placement in young women leads to a sense of loss over their body image perception owing to the forced limitations of clothing, including the inability to wear bathing suits. It has also shown to interfere with moderate activities such as pushing a vacuum cleaner or moving a table.(3-5) The loss of body image and possible drop in self-esteem may have negative effects on their sexuality and place them at higher risk for developing psychiatric illnesses.(4-6) Furthermore, women are more likely to be affected compared to men.(7)
To overcome this, techniques for submammary or subpectoral placement of ICDs were described by various authors.(8-11) In such cases, the prominence of the ICDs would be hidden beneath the thoracic soft tissues, with more concealed incisions. Giudici et al showed that 97% of women undergoing submammary placement of ICDs felt they had made the right choice, with 95% of them suggesting that they would undergo the same procedure again and recommend it to their friends. This suggests that submammary placement of ICDs has high patient acceptance, with long-term satisfaction.(8)
This case series aims to highlight the technique of submammary placement of ICDs from a plastic surgeon’s perspective, with the goal of increasing awareness about such a technique, addressing the challenges faced and increasing the potential for future collaborations with our cardiology colleagues.
CASE SERIES
The patients described in this paper are young Chinese women with newly diagnosed familial long QT syndrome. They shared a similar family history of syncope with seizures and a relative with sudden cardiac death in the early 30s.
Case 1
A 20-year-old Chinese woman with no significant past medical history suffered an out-of-hospital collapse during a netball game. She was resuscitated successfully by her coach. This was complicated by seizures secondary to cerebral anoxia from torsades des pointes at the emergency department. She was admitted to the intensive care unit for further monitoring. Subsequent electroencephalogram (EEG), coronary angiogram, transthoracic echocardiogram and cardiac magnetic resonance images were normal. Her electrocardiogram (ECG) showed a long QTc interval of 528 ms.
Case 2
A 28-year-old Chinese woman with no noteworthy past medical history suffered a seizure secondary to cerebral anoxia from torsades des pointes. EEG and cardiac investigations were largely normal. However, long QTc intervals of 571 ms were seen on ECGs.
Our technique
For secondary prevention of sudden death, ICD placement, in addition to long-term beta-blocker therapy, was discussed with the patients and their families. In addition, the choice of placing the ICD in the conventional subcutaneous infraclavicular position versus a submammary pocket was discussed with the patients. The patients favoured the latter. Subcutaneous placement in the infraclavicular region remained our salvage procedure should submammary placement fail. The plastic surgery team was approached for consideration of placement of the ICD in the submammary region given the young age of the patients.
The procedure was performed in the electrophysiology (EP) lab under general anaesthesia. Patients were placed in a supine position with the left arm abducted at 90º to expose the lateral edge of the breast and axilla. Patients were cleaned and draped in the usual sterile manner with povidone iodine 10% solution and chlorhexidine gluconate 0.05% solution. The surgical field of exposure spanned from the sternal clavicular notch to the left anterior axillary line and from the left supraclavicular fossa to the left costal margin, inferior to the breast. A nipple shield was placed after draping to prevent contamination from ductal bacteria.
Our EP colleagues started the procedure with a venogram from the left upper limb to check for patency of the left axillary and subclavian veins. Local anaesthesia (a mixture of Marcaine, lidocaine and adrenaline) was infiltrated in the infraclavicular region. A 1.5-cm horizontal incision inferior and parallel to the clavicle was made. The guide wire was inserted into the left axillary vein using the Seldinger technique and under fluoroscopic guidance. The right ventricular lead was then inserted and advanced under fluoroscopic guidance. A standard-length 62-cm lead was utilised. Once the leads were in position, the anchoring sheath was placed and they were anchored to the clavipectoral fascia with three silk 0/0 sutures.
Once the leads were secured, the plastic surgery team proceeded with the development of the inframammary pocket. A 5-cm inframammary incision was made. The length of the incision was predetermined by measuring the width of the device, as this would be the minimum size for insertion of the ICD. Dissection down to the pectoralis fascia was performed with diathermy. The submammary pocket was then developed. Similar to breast augmentation techniques, careful attention was paid to the size of the submammary pocket. Making the pocket too large would predispose the ICD to migration and increase the risk of lead dislodgement. The pocket was sited under the most prominent portion of the breast mound to reduce the palpability of the implant. Meticulous haemostasis and preservation of perforators and nerves were ensured during the development of the submammary pocket. The pocket was irrigated with copious amounts of gentamicin saline wash.
After development of the submammary pocket, a submammary tunnel towards the infraclavicular incision was made using a long Roberts artery forceps by careful blunt dissection. The estimated length of this tunnel was 22 cm in our patients. It is important to create the submammary tunnel from the submammary pocket towards the infraclavicular incision to avoid tunnelling through the wrong plane. The tunnel was created along the submammary plane, and care was taken to prevent tunnelling into the pectoralis major muscle and prevent penetration into the breast parenchyma. Once the tunnel was created, the leads were ready to be railroaded through.
To prevent damaging the leads by directly grasping them with the artery forceps, we utilised a 22-fr Foley catheter. The catheter tip was cut off and the tip was railroaded through the tunnel under the guidance of the Roberts artery forceps. The tip of the lead was then inserted into the cut end of the catheter and anchored with silk 2/0 sutures. Subsequently, the catheter was slowly withdrawn from the tunnel with the left hand and the lead was fed into the infraclavicular end of the tunnel with the right hand. Once the lead was out in the submammary pocket, its excess length was pulled into the submammary pocket to minimise excess length in the infraclavicular pocket, while still maintaining some slack to allow full range of movement of the shoulder without the lead being dislodged. The lead was then connected to the pulse generator, and an on-table testing of the ICD was conducted to ensure good pacing and sensing parameters with no diaphragmatic capture. The device was then anchored to the pectoralis fascia with silk 2/0 sutures. The final orientation of the device was positioned to ensure minimal coiling and minimal tension on the lead (
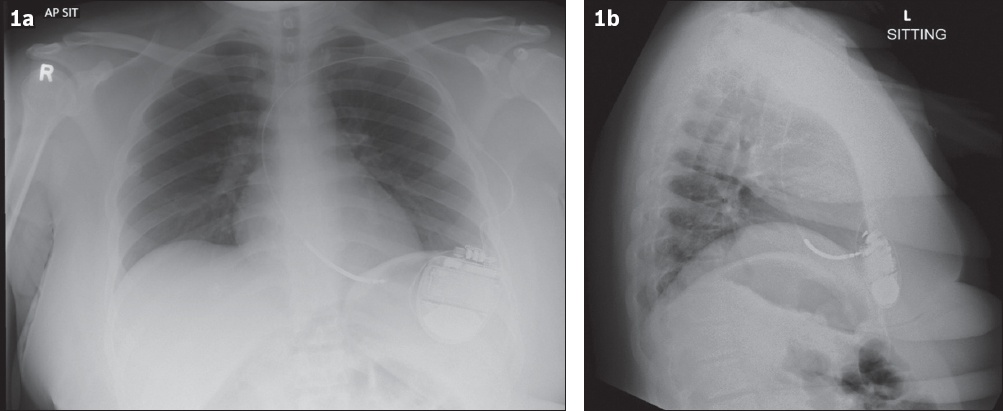
Fig. 1
(a) Anteroposterior sitting and (b) lateral chest radiographs show the chest region of Case 1 after submammary placement of implantable cardioverter defibrillators.
Postoperatively, the patients were encouraged to wear a supportive bra without an underwire to avoid pressure on the incision. Patients were advised against excessive abduction and straining of the left upper limb to avoid unnecessary tension on the leads. Standard wound care advice was given, and patients were discharged after post-placement checks.
Follow-up
Both patients were given follow-up appointments at one week, one month and two months after surgery. Patients reported minimal pain immediately after the operation and at the one-week follow-up. At the one-month follow-up appointment, the wound incisions were well healed, with minimal palpability of the ICD under the left breast. Both patients were satisfied with the procedure and reported minimal impact on their daily activities.
DISCUSSION
ICD placement is a life-saving therapy for secondary prevention of sudden death in familial syndromes such as long QT syndrome. Traditional ICD placement in the subcutaneous infraclavicular region has been associated with poorer body image and sexual self-esteem, placing young women at higher risk for developing psychiatric illnesses.(4-7) This negative association has created a need for alternative methods of ICD placement.
Subcutaneous placement of ICD was a suitable consideration for both patients in our series. In terms of cosmetic results, the subcutaneous ICD would result in an incision scar and protrusion of the device in the mid-axillary line, which is cosmetically less acceptable compared to the submammary devices. The choice of placing the ICD in the conventional subcutaneous infraclavicular position versus a submammary pocket was discussed with the patients, and the patients favoured the latter. Subcutaneous placement in the infraclavicular region remained our salvage procedure in case submammary placement failed.
Numerous methods for submammary or subpectoral placement of ICDs have been described, with minor differences in incision placement and plane of dissection.(8-10,12-14) Both submammary and subpectoral methods have been proven to be effective and cosmetically acceptable by patients when compared to conventional implant placement.(8,14) More importantly, during long-term follow-up, no difference in the technical performance of the device nor the need for wound or device revision was observed with both methods of ICD placement.(14)
The technique described in this series closely resembles that described by Ozin et al(12) and Schaverien et al,(13) with some differences in tunnelling methods. In our cases, we achieved transmission of the leads through the subcutaneous tunnel using a Foley catheter to minimise damage to the leads, while Schaverien et al achieved the same through blunt dissection with a rigid oesophagoscope. Other methods such as use of a pericardiocentesis needle or a thoracic catheter for transmission of the leads have also been described.(10,15)
While no comparative studies between subpectoral and submammary placements have been performed, we prefer a submammary plane of placement. Firstly, placing the ICD in a submammary plane completely avoids the need for any form of pectoralis major transection or subpectoral dissection, and also avoids the placement of anchoring sutures on the perichondrium, which can be very painful. In our practice of performing dual-plane breast augmentation techniques, extended release of the pectoralis major muscles often led to increased postoperative pain. By avoiding a subpectoral placement of the ICD, we aimed to reduce the postoperative pain of the patient.
Secondly, a submammary plane of placement avoids the risk of causing a pneumothorax during dissection of the subpectoral pocket, although the risk of causing a pneumothorax during subpectoral placement is low, as shown by the fact that Giudici et al(8) reported only one incident of pneumothorax in his vast experience. Persichetti et al(10) and Oh et al(9) reported no pneumothoraxes in their studies; therefore, placing the ICD in a submammary plane circumvents the risk of a pneumothorax from pocket dissection. The risk of causing a pneumothorax during venous lead access remains unchanged with either plane of placement.
Lastly, selecting a submammary plane of placement reduces the movement and animation deformity of the ICD on pectoralis muscle contraction as well as the stress of the device on the ribs, as described by Giudici et al.(8) However, even in a submammary plane of placement, it does not completely eliminate potential movement of the ICD on muscle contraction, as the anchoring sutures are placed on the pectoralis major fascia.
As cosmesis is one of the goals of submammary placement of ICDs, incision placement is an important factor that should be considered. We opted for an inframammary incision over an axillary incision, as both our patients had large ptotic breasts of Regnault grading 2 and 3. This made placing the ICD directly under the breast mound easier via an inframammary incision, as the breast parenchyma had descended, making the upper pole somewhat deficient. Ptosis of the breast also concealed the inframammary incision more than it would have normally done in a normal non-ptotic breast. We believe that both the axillary and inframammary incisions are cosmetically pleasing and effective in allowing for submammary placement of the ICD and should cater to patient preferences and characteristics.
To the best of our knowledge, studies on the technique of submammary placement of ICDs have not been published in Singapore. Although Giudici et al(8) mentioned that they performed the technique themselves, numerous other authors have described the benefits of collaboration between plastic surgery and cardiology in such procedures, as submammary or subpectoral pocket dissections are commonplace in plastic surgery procedures.(9,10,12,13) With this paper, we hope to increase the collaborative opportunities with our colleagues in cardiology. This collaboration also allows this surgery to be performed under monitored anaesthesia and sedation, eliminating the need for general anaesthesia.
In conclusion, submammary placement of ICDs is a well-described technique to avert the downsides of subcutaneous placement. Numerous techniques have been described in the literature for alternative planes of placements and incisions; however, all these techniques have the common goals of improving the cosmesis of the procedure, reducing the palpability of the device and minimising the impact of daily activities. We described our local technique and experience in submammary placement of ICDs with an aim to improve awareness of the availability of such a technique and expand opportunities for further collaborations with our cardiology colleagues.


