Abstract
Intraosseous haemangiomas (IOHs) are benign vascular bone tumours that account for 1% of all primary bone tumours. They are most frequently seen in the vertebrae and skull, and are rarely found in long bones. Herein, we present an uncommon case of a 25-year-old woman with a solitary IOH that occupied the left femoral neck. We describe the clinical, radiological and histological details of the case, as well as the three-year outcome of the surgical treatment, which successfully preserved the femoral head. We also conducted a review of the literature on this uncommon entity.
INTRODUCTION
Intraosseous haemangiomas (IOHs) are benign bone tumours that arise from blood vessels; they account for 1% of all primary bone tumours.(1) IOH is one of the few primary bone tumours that has female predominance, with a female-to-male ratio of 3:2.(2) It is most frequently seen in the vertebrae (30%–50%) and skull (20%), while involvement of the appendicular skeleton, such as long tubular bones, is less frequent.(3) Solitary IOH in long bones is usually located in the diaphyseal and metadiaphyseal regions.(2) Herein, we present an uncommon case of a large, solitary IOH in the femoral neck. We report the clinical, radiological and histological details of the case, as well as the three-year outcome of the surgical treatment, which aimed to preserve the femoral head.
CASE REPORT
A 25-year-old woman presented with left groin pain that persisted for two months. She had no history of trauma or fever, and no history of any remarkable medical or surgical illnesses. On physical examination, there was left groin tenderness and the motion of her left hip joint was limited due to pain. She walked with an antalgic gait and the hip pain intensified with weight-bearing activities. Imaging was undertaken, including plain radiography (Figs.
Fig. 1
(a) Anteroposterior radiograph of the pelvis and (b) lateral radiograph of the left hip show a multilocular lucent lesion over the left femoral neck. The lesion extends to the femoral head and intertrochanteric area, with sclerotic borders and internal trabeculation. Three-week postoperative (c) anteroposterior radiograph of the pelvis and (d) lateral radiograph of the left hip show that the lesion was curetted and an allogenic fibular cortical strut, with proximal femoral locking plate and screws in situ, was applied. (e & f) Repeat radiographs taken during follow-up at 39 months show no recurrence and a well-incorporated allograph.
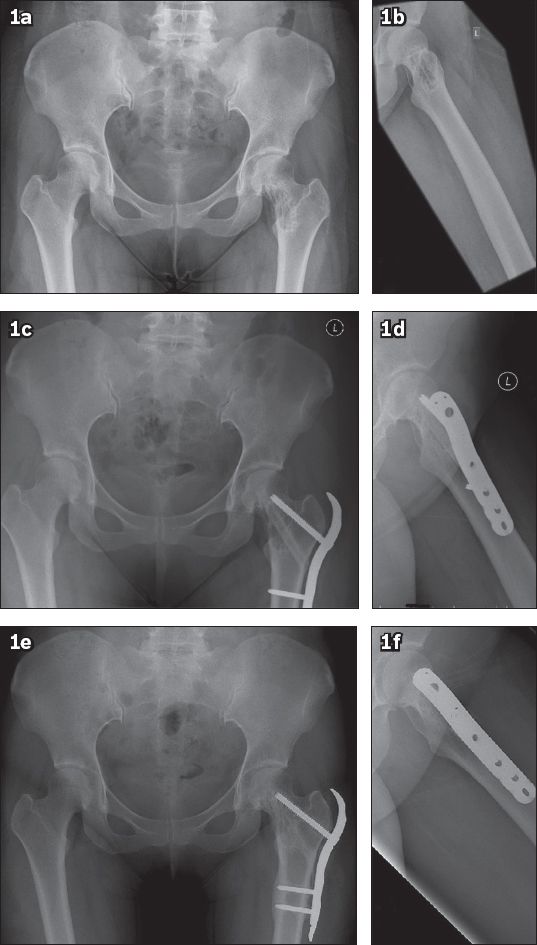
Fig. 2
(a) Coronal CT image of the left hip shows a lesion occupying the medial aspect of the left femoral head and neck. The lesion has an irregular margin, a narrow transition zone and coarse trabeculation within. (b) Axial CT image shows multiple ‘dots’ within the lesion.
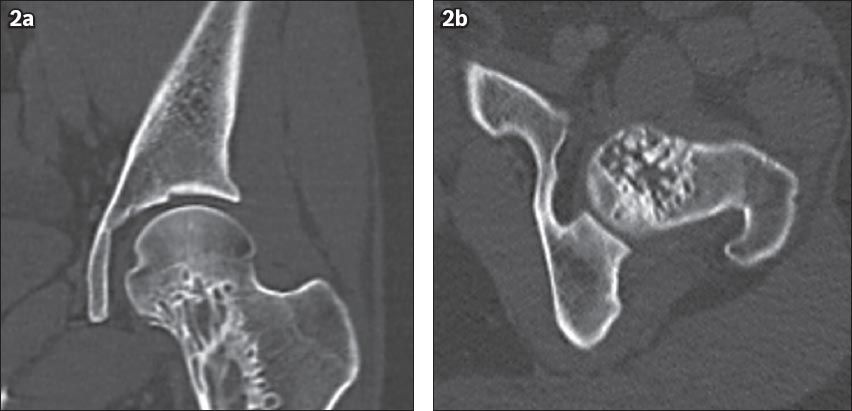
Fig. 3
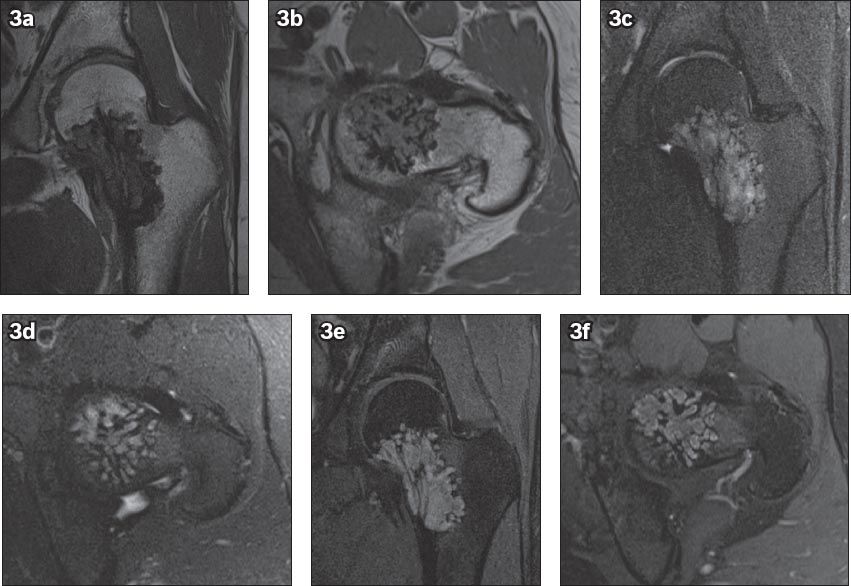
MR images of the left hip show a 5.7 cm (craniocaudal) × 2.7 cm (mediolateral) × 3.6 cm (anteroposterior) irregularly marginated lesion with a narrow zone of transition. The lesion occupies the medial aspect of the left femoral neck, and extends into the femoral head and upper metaphyseal region. Thickened trabeculae is observed within the lesion. The lesion is of low signal intensity on (a & b) T1-W images, and of high signal intensity on (c & d) T2-W images. The spaces between the trabeculae within the lesion show profound enhancement on (e & f) post-contrast images.
Prior to surgery, we carried out an angiography to explore the possibility of embolisation. However, the angiogram revealed a cavernous haemangioma with no multiple arterial feeders; there was no suitable artery to be considered for embolisation (
Fig. 4
(a) Radiograph and (b) angiogram of the left hip with selective injections of arteries supplying the hip region show no multiple arterial feeders to the lesion. No arteries were identified to be appropriate for embolisation.
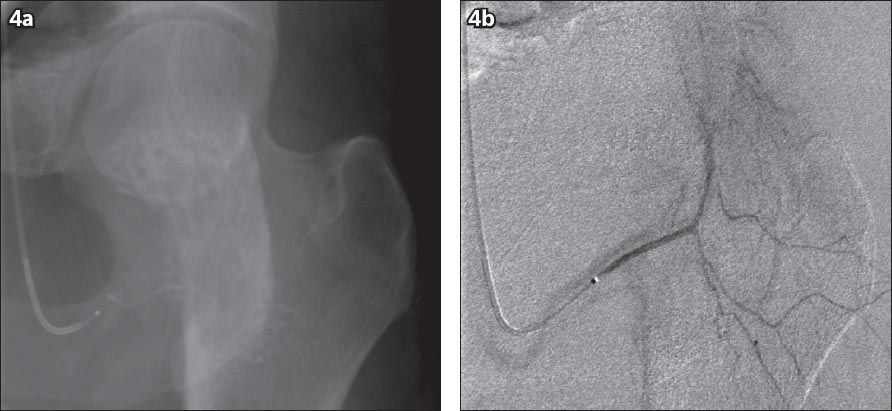
Fig. 5
Intraoperative fluoroscopic images show (a) the lesion being curetted with a long handle curette, and (b) a freeze-dried allograft fibular cortical strut being inserted into the channel, which was created by a 11-mm cannulated reamer.
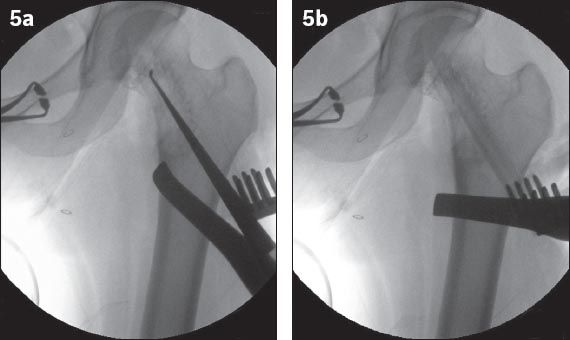
Fig. 6
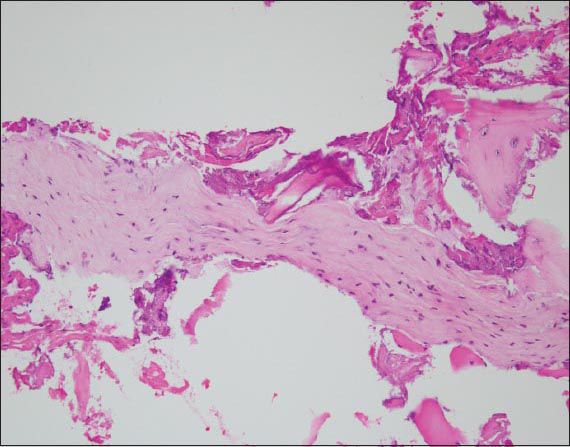
Photomicrograph shows elongated strips of cellular fibroblastic tissue, consistent with septae from a haemangioma. No endothelial lining or malignancy is identified. These features are consistent with a cavernous haemangioma (Haematoxylin & eosin, × 200).
Postoperatively, repeat plain radiography was conducted (Figs.
DISCUSSION
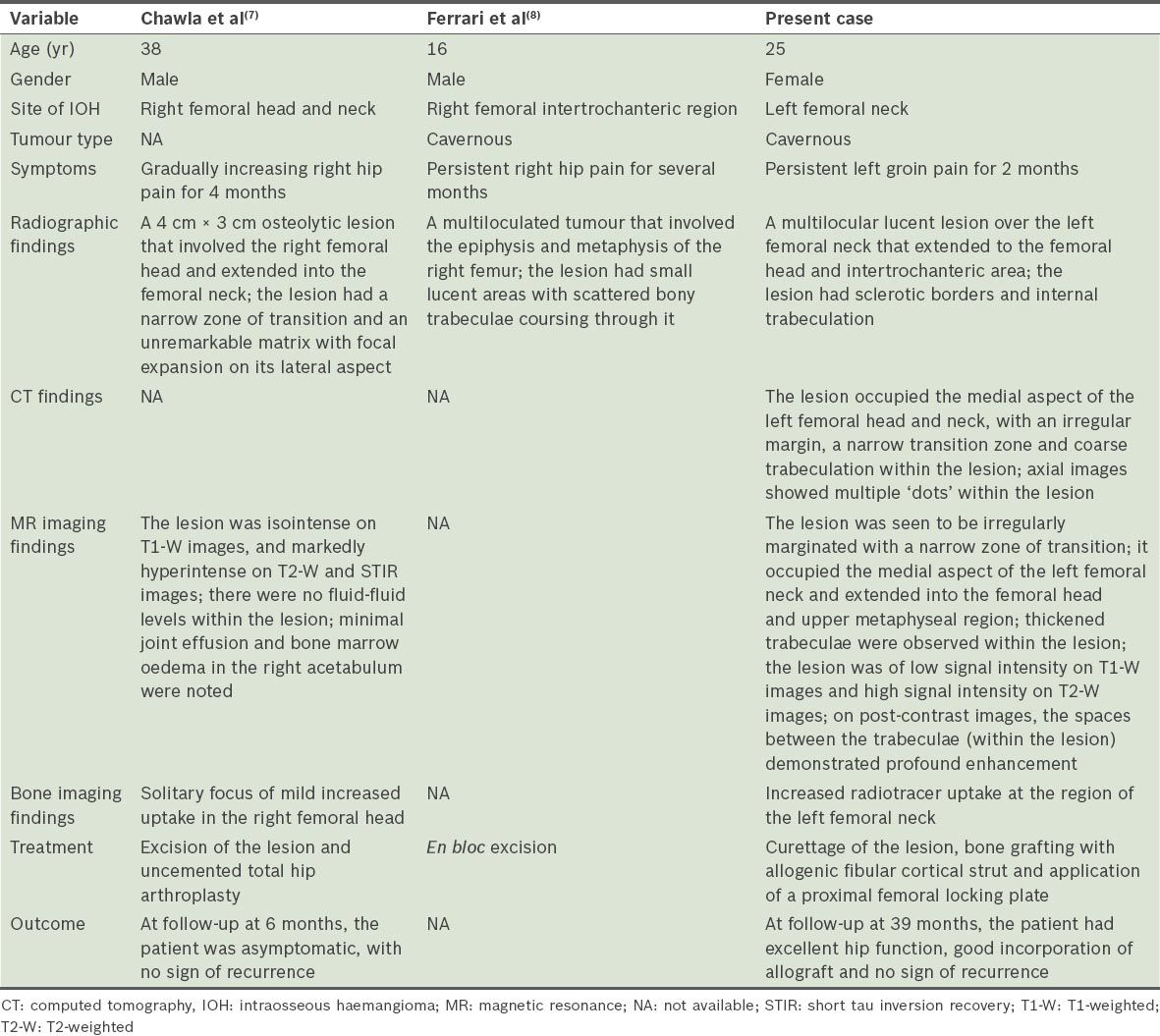
IOH was first described in the literature by Toynbee in 1845.(4) Hitzrot reported the first complete and illustrated case of a solitary IOH in an extremity in 1917.(5) As mentioned in the study by Kaleem et al, it is extremely rare for a radiologist to encounter a primary haemangioma in the long bones.(2) The rarity of IOH in the extremities is often emphasised. In fact, we were only able to find two other articles on solitary IOH in the proximal femur in the literature. This was despite the fact that the proximal femur is one of the most common locations for bony benign lesions.(6) Kaleem et al conducted a literature review of solitary IOH of the extremities in 2000.(2) In that review, the authors yielded a large group of cases (n = 34) in which substantial clinical, radiological and histological details were provided, but none of the cases involved the proximal femur.(2) Chawla et al(7) reported one case of IOH involving the femoral head in 2006 and Ferrari et al(8) described a case of IOH at the intertrochanteric area of the femur in 2011. The comparison between the previous studies and the present case is summarised in
Table I
Comparison of two cases of previously reported solitary proximal femoral intraosseous haemangioma (IOH) and the present case.
IOHs of the axial skeleton are usually asymptomatic, while those over the appendicular skeleton, depending on the size and site of lesion, are often associated with symptoms such as pain, swelling and pathological fractures.(2) According to the site of the lesion, IOHs of tubular bones can be classified into medullary, periosteal and intracortical IOHs. Medullary IOHs can be further subcategorised into diaphyseal (48%), metadiaphyseal (30%), metaphyseal (12%), metaepiphyseal (4%), epimetadiaphyseal (3%), epiphyseal (1.5%) IOHs, and IOH over the total length of the bone (1.5%).(7) In the present case, the patient presented with groin pain and her IOH lesion was of the metaepiphyseal medullary type.
The literature describes the radiological patterns of IOH in the appendicular skeleton as being highly variable and usually nonspecific; a preoperative diagnosis is made only in a minority of cases.(2) On radiography, medullary IOHs may show lytic, well-defined, slightly expansile lesions that have a coarse trabecular pattern, which produces a characteristic sunburst, soap bubble/honeycomb appearance due to the expansive proliferation of engorged vessels and thickened, remodelled bone trabeculae.(3,7) Alternatively, it may present with an expansile osteolytic appearance.(9) The appearance of IOHs on CT corresponds with its appearance on plain radiography. The cross-sectional images may demonstrate the lesion with a typical ‘polka dot’ pattern, as the thickened vertical trabeculae are seen as small punctuate areas of sclerosis.(1) MR imaging of the medullary IOH usually shows variable signal intensity on T1-weighted sequences and high signal intensity on T2-weighted sequences due to water in the stagnant blood within the haemangioma; contrast-enhanced images can show avid enhancement.(1,3) Corresponding to the ‘polka dot’ pattern observed in axial CT images, axial MR images may also reveal multiple dots.(2) Tc-99m MDP bone scintigraphy is considered nonspecific, with either variable degrees of radiotracer uptake or absence of tracer accumulation. The classical findings of these different diagnostic modalities help contribute to a definite imaging-based diagnosis. This, however, can only be made in a small number of patients with IOH; in patients without pathognomonic radiological features, histology is required for diagnosis. In the present case, our patient’s lesion had the typical radiological characteristics of medullary IOH – a honeycomb appearance on radiographs, a ‘polka dot’ pattern on axial CT and MR images, high signal intensity on T2-weighted MR images, and marked enhancement on MR images after contrast administration. It was one of those cases in which a definite imaging-based diagnosis of IOH could be made preoperatively.
Due to the rarity of IOHs and the lack of consistent radiological features, histology plays an important role in the final diagnosis. This is especially so for the examination of en bloc specimens. For the examination of biopsy specimens, curettage or needle biopsy procedures may disrupt the thin-walled blood vessels of the lesion, rendering histological findings nonspecific.(1) In such cases, the final diagnosis usually relies on a comprehensive analysis of the clinical, radiological and histological findings. Histologically, IOH can be classified as cavernous, capillary, venous or mixed, depending on the type of vascular involvement.(2) Among these, cavernous IOH is the most common type of vascular involvement, accounting for 50% of all types of vascular involvement, while capillary IOH accounts for 10%.(7) In the present case, the diagnosis of cavernous IOH was made based on radiographic and angiographic findings. The histological features of the biopsy specimens from curettage were consistent with the diagnosis made.
Treatment of benign bone lesions of the proximal femur depends on the size and site of the lesion, as well as the effect that the lesion has on the bony framework. For small, silent lesions that occur without any weakening of the bony framework, close observation can be considered. Some nonoperative treatments specific to IOH may also be considered; they include direct sclerotherapy, radiotherapy and selective arterial embolisation.(2,10) If the lesion is large and causes weakening of the bony framework (thus rendering impending or actual pathological fracture), surgical treatment is frequently indicated.(6) For lesions involving the articular surface of the femoral head, resection and reconstruction with hip endoprosthesis is often required.(7,11) If the lesion involves the femoral neck or intertrochanteric area with a risk of impending pathological fracture, but preservation of the hip articular surface is possible, curettage and bone grafting with application of an internal fixation device is preferred and has been described with excellent outcomes in previous studies.(6,12,13)
For bone grafting, cortical bone grafts such as fibular cortical strut grafts are favoured, as they work as a mechanical strut and biological bone graft. The choice between autogenous or allogenous cortical grafts depends on multiple factors. Autogenous cortical grafts are superior to allografts in their rate and completeness of incorporation; this is especially the case for large solitary lesions.(6) The drawbacks include donor site morbidity and limited supply.(14) Allografts, on the other hand, offer the advantages of reduced rate of graft resorption, no donor site morbidity and wide availability; however, they may have a higher rate of infection, a reduced rate of incorporation and a small risk of disease transfer and immunogenicity.(14,15) Augmentation with an internal fixation device can provide additional mechanical support, so as to lessen the risk of a pathological stress fracture, especially during the initial 6–12 months before significant graft incorporation.(13,15) Based on previous studies,(6,12,13) the compression hip screw appears to be the most commonly applied internal fixation device. In the case described by Chawla et al,(7) excision of the femoral head and total hip arthroplasty was unavoidable even though the patient was young, as there was involvement of the articular surface of the femoral head. In the case reported by Ferrari et al,(8) the teenage patient who had IOH with no direct involvement of the articular surface of the femoral head underwent en bloc resection of the lesion, as the lesion over the intertrochanteric area was considered too large to allow for preservation of the femoral head.
In the surgical treatment of solitary IOH of the extremities, a common concern is intraoperative bleeding. Yu et al(10) described a case in which a patient who had an IOH over the left humerus was initially misdiagnosed with fibrous hyperplasia. In that case, the first attempt at surgery was abandoned when the surgeons encountered uncontrolled haemorrhage during the attempt to curette the lesion.(10) Therefore, in the surgical treatment of an IOH, detailed preoperative plans are essential so as to avoid massive blood loss. Preoperative angiography can be used to identify the major blood vessels that supply the IOH. If the IOH is supplied by a non-trunk vessel, embolisation should be performed; however, if the blood supply of the IOH is mainly from a trunk vessel, a technique introduced by Yu et al, in which a balloon was preset to temporarily block the blood flow,(10) could be used. Other considerations may include preoperative preparation of adequate blood products, and perioperative use of recombinant human erythropoietin and iron.
Similar to the case described by Ferrari et al,(8) our patient was young and the IOH did not involve the articular surface of the femoral head. The primary objective in the present case was to remove the IOH, prevent a pathological fracture and preserve the femoral head. Although preoperative embolisation was considered, angiography revealed no major arterial feeders for the IOH, thus rendering embolisation unnecessary. Curettage of the lesion was successfully performed without gross blood loss. Following curettage, we chose to use an allograft fibular cortical strut for our patient, as we were concerned about the possibility of recurrence and an allograft strut is less prone to resorption by the vascular lesion than an autograft strut in recurrence cases. As the lesion involved over 50% of the femoral neck, we decided to use a proximal locking plate with a 7.3-mm locking screw applied to the superior aspect of the femoral neck. We did not use a compression hip screw with a 12.5-mm lag screw due to concerns that it would be too large for the femoral neck, thus possibly increasing the likelihood of femoral neck fracture during screw placement. The outcome of the surgery in the present case was successful. The patient recovered with excellent hip function, and the allograft fibula strut incorporated well and strengthened the femoral neck. No recurrence was seen during follow-up at 39 months.


