Abstract
INTRODUCTION
Shortening of the tendon and muscle is recognised as a strong predictor of surgical failure of supraspinatus tendon tears. Changes in muscle architecture following repair have not been thoroughly investigated. Hence, we aimed to compare the pre- and postoperative architecture of the supraspinatus.
METHODS
We recruited eight participants with full-thickness supraspinatus tears. Images of the supraspinatus were captured preoperatively (pre-op) and postoperatively at one month (post-op1), three months (post-op2) and six months (post-op3) in relaxed and contracted states (0º and 60º glenohumeral abduction). Fibre bundle length (FBL), pennation angle (PA) and muscle thickness were quantified. Self-reported function, and maximal isometric abduction and external rotation strengths were assessed.
RESULTS
The mean FBL increased from pre-op to post-op1 (p = 0.001) in the relaxed state and from pre-op to post-op2 (p = 0.002) in the contracted state. Decrease in FBL was observed from post-op2 to post-op3 in the relaxed state. The mean PA decreased from pre-op to post-op1 (p < 0.001) in the relaxed state, but increased from post-op2 to post-op3 in both relaxed (p = 0.006) and contracted (p = 0.004) states. At post-op3, external rotation (p = 0.009) and abduction (p = 0.005) strengths were greater than at post-op2. Overall function increased by 47.67% from pre-op to post-op3.
CONCLUSION
Lengthening of the supraspinatus occurs with surgery, altering the length-tension relationship of the muscle, which can compromise muscle function and lead to inferior surgical outcomes. These findings may guide clinicians to optimise loads, velocities and shoulder ranges for effective postoperative rehabilitation.
INTRODUCTION
Rotator cuff tears involving the supraspinatus tendon are common and can be associated with debilitating pain and dysfunction in the shoulder.(1) The size and degree of supraspinatus tendon tears can range from low-grade partial-thickness tears to massive full-thickness tears. In the general population, the prevalence of rotator cuff tears was reported to be up to 22%.(2) A partial-thickness tear of the supraspinatus can progress to become a full-thickness tear involving the other rotator cuff tendons if not detected and addressed early.(3)
Supraspinatus tendon tears are associated with structural and architectural changes in the tendon and muscle.(4) The importance of muscle architecture in elucidating the functional properties of the muscle and its role in clinical decision-making have been underscored in numerous studies.(5-9) Architectural parameters of the muscle, such as fibre bundle length (FBL), pennation angle (PA), physiological cross-sectional area (CSA) and muscle volume, directly influence muscle excursion and its force-producing capacity;(5) they can predict the forces on the musculoskeletal system and provide a mechanical basis for muscle injury during various loading conditions.(10)
Changes in muscle architecture due to tendon injuries have been reported in numerous studies. Meyer et al(11) reported shortening of fibre bundles and an increase in PA following tendon release of the infraspinatus in a sheep model. Similarly, Kim et al(12) reported a shortening of FBL and an increase in PA in cadaveric specimens that had full-thickness supraspinatus tendon tears with retraction. In an in vivo study, Kim et al(4) examined the architectural changes distinctive of the anterior and posterior regions of the muscle in patients with full-thickness tendon tears. Compared to normal controls, shortening in FBL was greater in the posterior region than in the anterior region. Contrary to cadaveric studies,(12,13) in vivo investigation by Kim et al(4) found the PA of the anterior region to be smaller in patients who had full-thickness tears with retraction compared to normal controls.
The treatment of rotator cuff tendon injuries is multifaceted and ranges from conservative physical therapy to surgical interventions such as arthroscopic debridement and reattachment of torn tendons. Tears that do not respond to non-surgical treatments are often surgically treated with pain relief, with functional improvement being the primary goal.(14) Despite advances in surgical techniques, the incidence of re-tear and failure of surgical repair is high, ranging from 11.4%(15) to 94%.(16) There has been a great deal of interest in the factors that contribute to the success of surgical repair of full-thickness tears, including the extent of fatty infiltration, atrophy and size of the tear; however, little is known about the association of muscle morphology with surgical repair and functional outcomes. Moreover, despite the clinical and functional relevance, muscle architectural changes in humans following surgical repair at different time intervals have not been investigated.
A greater understanding of these architectural changes may allow for surgeons and rehabilitation specialists to be better informed regarding the functional consequences of surgical repair techniques. This would also allow better prediction of the amount of tension that can be safely applied to reattach the tendon during surgical repair as well as the type of exercises that would improve function without causing further trauma to the muscle.
The morphology of supraspinatus has been investigated using cadaveric dissection(17-19) and several imaging modalities, including ultrasonography (US)(20) and magnetic resonance (MR) imaging.(21) US is an ideal imaging technique in clinical and rehabilitative settings because it is portable, easily accessible and non-invasive.(22) It is also an established method for quantifying FBL, PA, CSA and muscle thickness (MT) in both normal and pathological supraspinatus muscle.(4,20,23) Therefore, the main objectives of this prospective observational study were: (a) to quantify and compare the fibre bundle architecture within the anterior region of the pathologic supraspinatus preoperatively and postoperatively at multiple time points using real-time US and (b) to assess and discuss the possible functional implications.
METHODS
Ethics approval was obtained from the Biomedical Research Ethics Board, University of Saskatchewan, Saskatchewan, Canada (BioREB #11-53). Participants were recruited from a single orthopaedic surgeon’s practice in Saskatoon, Saskatchewan, Canada. To be included, participants were required to have a full-thickness tear of the supraspinatus tendon confirmed with US or MR imaging and be scheduled for arthroscopic surgical repair. Participants were excluded if they had any prior shoulder surgery, neuromuscular disease or any conditions other than a rotator cuff tear that could limit active shoulder abduction. Eligible participants were identified by the surgeon and, with the participants’ consent, were contacted by the study investigator via telephone. Participants’ medical history was collected from files and screened again for the study inclusion criteria. Each participant was provided with the study information sheet for careful review, and signed informed consent was obtained prior to the first testing session.
Each participant attended four US scanning sessions: one preoperatively and three postoperatively at different time intervals. The postoperative sessions were scheduled at one month (post-op1), three months (post-op2) and six months (post-op3) after the surgery. For each of the scanning sessions, the participants were seated with their back supported in a chair with adjustable height. A previously validated US protocol by Kim et al(20) was used for landmarking and scanning of the muscle. In all testing sessions, the scans were performed in two shoulder positions, with the exception of the one-month follow-up (post-op1), where active muscle contraction was contraindicated. First, the muscle was scanned in a relaxed state – 0º glenohumeral (GH) abduction, with the arm resting by the participant’s side and the palm of the hand facing medially. Next, the muscle was scanned in the contracted state with the participants holding their arm against gravity at 60º GH abduction, with the elbow flexed at 90º and neutral GH rotation. A universal goniometer was used to measure the shoulder positions. To maintain consistency, all scans were performed by one researcher (RS) who was trained in US scanning of the supraspinatus. A portable US scanner (LOGIQ e BT08, GE Healthcare, Milwaukee, WI, USA) fitted with a linear (38.4 mm) 12-Hz transducer (resolution 0.3 mm) was used. As outlined in previous studies,(4,8,20,24) panoramic US images were taken to measure the fibre bundles of the anterior region of the supraspinatus; the US probe was placed along the length of the muscle. To assess the thickness of the muscle, sagittal US images were captured by placing the probe perpendicular to the muscle belly.(20) In each arm position, 15–20 images (panoramic and sagittal) were captured. Measurement errors were minimised by keeping the tilt angle of the US probe perpendicular to the fibre bundle plane and applying minimum pressure to ensure that the underlying tissues were not deformed.
All data was saved on the US unit for subsequent analyses. The architectural parameters measured on the saved images were FBL, PA and MT. All the measurements were performed by one investigator (RS), who received adequate training to perform measurements on the US images. Among the saved images, one image with the clearest fibre bundles in the middle part of the muscle belly was chosen; the middle part forms the majority of the anterior region.(18) The fibre bundles in which the medial and lateral attachment ends were apparent and that could be seen along the majority of its length were used. FBL was computed as a linear distance between the medial and lateral attachment sites. PA was measured as the angle between the fibre bundle and its attachment to the intramuscular tendon (
Fig. 1
Evaluation of fibre bundle length and pennation angle using ultrasonography. Panoramic US images show the right supraspinatus following injury and surgical repair in the relaxed state (a) preoperatively; (b) one month postoperatively; (c) three months postoperatively; and (d) six months postoperatively. Fibre bundle from the anterior region is demarcated by a line with double-headed arrows and the intramuscular tendon by a dotted line. Ø: pennation angle; FBL: fibre bundle length; SC: subcutaneous tissue; SF: supraspinous fossa
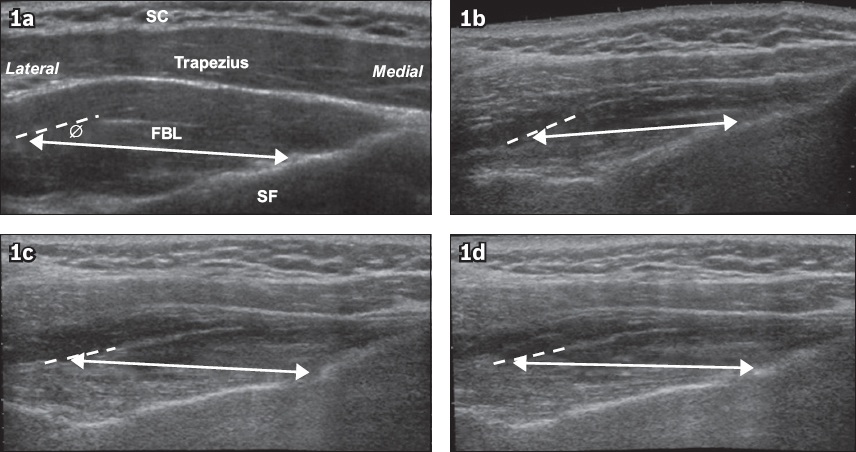
Fig. 2
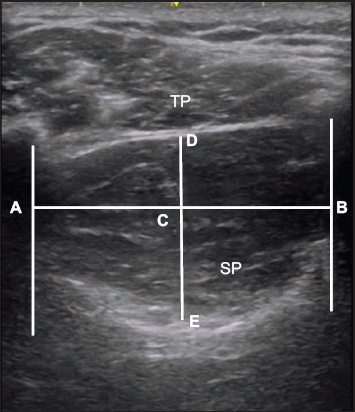
Evaluation of muscle thickness using ultrasonography. Sagittal US image shows the right supraspinatus in the relaxed state. The distance between D and E represents supraspinatus muscle thickness. A: acoustic shadowing of the clavicle; B: acoustic shadowing of the scapular spine; C: midpoint of distance between A and B; D: upper border of the supraspinatus muscle; E: lower border of the supraspinatus muscle; SP: supraspinatus; TP: trapezius
Strength testing was conducted after US imaging during each session, except at post-op1, in which active abduction was contraindicated and the arm on which surgery was performed was still immobilised in an abduction sling with a bolster (UltraSling®, DJO Global, Lewisville, TX, USA or SmartSling®, Össur, Foothill Ranch, CA, USA). All participants were seated with their back supported and feet flat on the floor. Maximal isometric shoulder strength was assessed using a handheld dynamometer (HHD; Lafayette Manual Muscle Test System, Lafayette Instrument Company, Lafayette, IN, USA). The HHD was chosen because it is portable, is easy to use and has been shown to be a valid and reliable tool for strength testing.(25-27) Two shoulder motions were tested: abduction and external rotation. Each position was tested with the arm at 0º of GH abduction, 90º of elbow flexion and neutral supination/pronation position (palm facing medially) of the forearm. The padded stirrup of the HHD was positioned perpendicular to the distal end of the humerus on the lateral side for shoulder abduction. For external rotation, the HHD was placed proximal to the ulnar styloid process, perpendicular to the ulna. The HHD was held stationary against the arm or forearm (depending on the position being tested), and the participants received standard verbal encouragement to push into the HHD with maximum effort. For all the positions tested, the participants were instructed to progressively increase their force and reach their maximum by the end of three seconds. Strength was measured twice in each position, with a 60-second rest between the two trials. The average value of the two measurements was determined as the peak value (in kg). To maintain consistency, the HHD was placed at the same position during all the strength testing sessions.
To assess self-reported function, the Western Ontario Rotator Cuff Questionnaire (WORC) was used. The WORC has been shown to be a valid and reliable tool for assessing rotator cuff conditions and repairs.(28) A higher score represents a more symptomatic shoulder and a lower quality of life. The total score can also be reported as a percentage of normal. The total final WORC score can vary from 0% (dysfunctional shoulder) to 100% (the highest functional status level). The WORC was assessed during each assessment session, preoperatively and postoperatively at one, three and six months.
All statistical analyses were performed using IBM SPSS Statistics version 20.0 (IBM Corp, Armonk, NY, USA). Data was screened for normality, skewness and kurtosis before parametric tests were applied. A repeated measures analysis of variance (ANOVA) was used to analyse and compare the architectural parameters (i.e. FBL, PA and MT) and strength values between pre- and postoperative measurements. For architectural parameters, separate ANOVAs were conducted for relaxed and contracted states. Greenhouse-Geisser adjustment was applied when the assumption of sphericity was violated (Mauchly’s test of sphericity, p < 0.05). Paired Student’s t-tests were used to compare the differences in measured variables between two time points. Statistical significance was set at p < 0.05, with Bonferroni adjustments made where appropriate (p = 0.05/4 = 0.013 for strength values and architectural measurements in relaxed state; p = 0.05/3 = 0.017 for architectural measurements in the contracted state). Percentage change was used to analyse WORC scores. Percentage score was calculated by subtracting the pre-training score from the post-training score, dividing by the pre-training score and multiplying by 100.
RESULTS
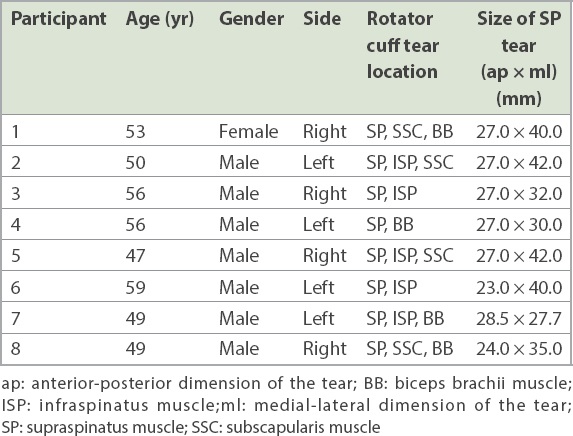
Eight participants (seven male and one female) aged 53.9 ± 4.8 (range 47–59) years participated in this study. Their demographics, tear sites and tear measurements are presented in
Table I
Participants’ demographics, tear sites and dimensions.
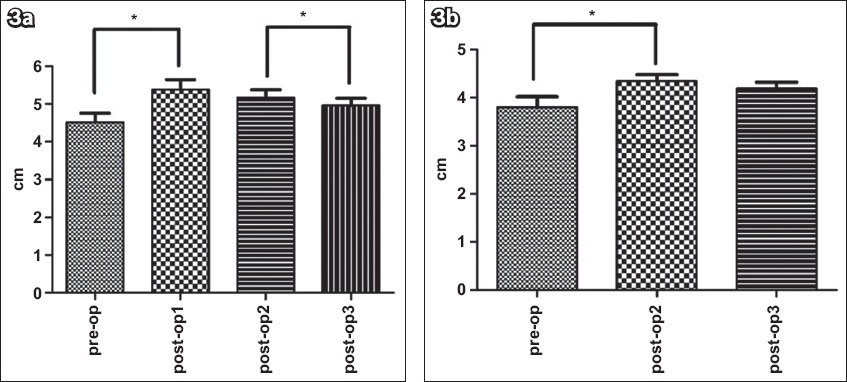
For FBL, there was a significant main effect of time in both the relaxed (p < 0.001) and contracted (p < 0.001) states. In the relaxed state, the mean FBL significantly increased from pre-op to post-op1 (p = 0.001). A significant decrease in the mean FBL was observed from post-op1 to post-op2 (p = 0.029), which further decreased at post-op3 (p = 0.004). Mean FBL at post-op3 was found to be significantly greater than the mean pre-op value (p = 0.014). When Bonferroni adjustments were made, mean FBL at post-op1 and post-op3 remained significantly different from that at pre-op (p < 0.013) and post-op2 (p < 0.013), respectively (
Fig. 3
Graphs show fibre bundle length changes pre- and postoperatively in the (a) relaxed and (b) contracted states. Values are presented as mean ± standard error. The data shows a significant time main effect (p < 0.001).
Similar to the FBL, a significant main effect of time was found for PA in both the relaxed (p = 0.002) and contracted (p = 0.023) states. In the relaxed state, the mean PA significantly decreased at post-op1 (p < 0.001) but increased significantly at post-op3 from post-op2 (p = 0.006). No significant differences for mean PA were found between pre-op and post-op3 (p = 0.21). The results did not change after Bonferroni adjustments for multiple comparisons (p < 0.013;
Fig. 4
Graphs show pennation angle changes pre- and postoperatively in the (a) relaxed and (b) contracted states. Values are presented as mean ± standard error. The data shows a significant time main effect (p < 0.001). *Indicates significant differences (Bonferroni adjustment: relaxed state: p < 0.013; contracted state: p < 0.017). Pre-op: preoperatively; post-op1: one month postoperatively; post-op2: three months postoperatively; post-op3: six months postoperatively
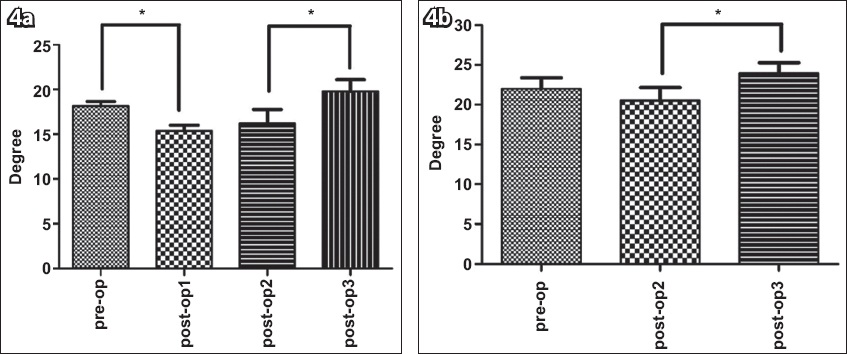
Fig. 5
Graphs show muscle thickness changes pre- and postoperatively in the (a) relaxed and (b) contracted states, with no significant differences across time. Values are presented as means ± standard error. Pre-op: preoperatively; post-op1: one month postoperatively; post-op2: three months postoperatively; post-op3: six months postoperatively
For external rotation strength, there was a significant main effect of time (p = 0.002). The mean external rotation strength at post-op3 was found to be significantly higher than that at pre-op (p = 0.011) and post-op2 (p = 0.009;
Fig. 6
Graph shows shoulder external rotation strength changes pre- and postoperatively. The data shows a significant time main effect (p = 0.002). Values are presented as means ± standard error.
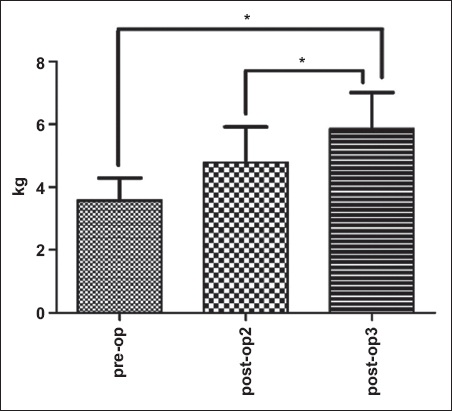
Fig. 7
Graph shows shoulder abduction strength changes pre- and postoperatively. The data shows a significant time main effect (p = 0.015). Values are presented as means ± standard error.
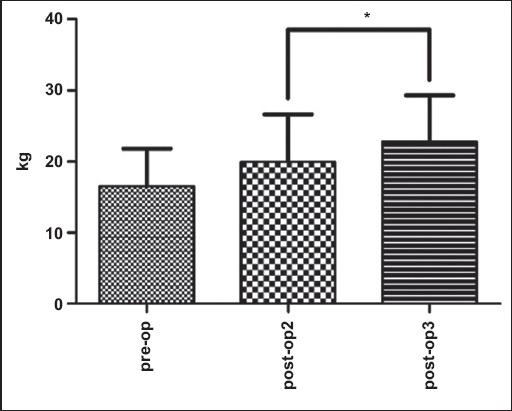
After Bonferroni adjustment for multiple comparisons for strength, no significant differences were detected for abduction strength between pre-op and post-op3 (p > 0.017).
The subjective assessment of participants showed improvement over time. An increase of 47.67% in the overall function was observed from pre-op to post-op3. The overall WORC scores were preop: 37.96% ± 19.21%; post-op1: 40.84% ± 14.98%; post-op2: 70.27% ± 19.07; and post-op3: 85.63% ± 12.34%, with higher percentages indicating better function.
DISCUSSION
The primary objectives of this study were to quantify and compare the architecture of the supraspinatus muscle pre- and postoperatively to better understand these changes over the recovery period and to elucidate the impacts on muscle function and functional recovery of the shoulder. To our knowledge, this is the first study performed in humans to investigate the changes in the supraspinatus architecture after rotator cuff repair. The findings of the study demonstrate significant differences in architectural parameters of the supraspinatus following surgical repair, and these changes show a distinct pattern. The lengthening of fibre bundles was observed after one month of surgery, which then decreased significantly by six months. By contrast, an initial decrease followed by an increase in PA over time was observed. The architectural data collected in this study has several clinical implications and may aid in improving the treatment of supraspinatus tendon tears. Additionally, the data available from this study has the potential to inform biomechanical and finite element models, which may be able to simulate different surgical scenarios, predict functional outcomes and assist in planning postoperative rehabilitation protocols.
When a tendon is torn and the musculotendinous unit retracts, it is common to see fibre bundles within the muscle shorten.(4,29) The shortening can continue to progress as long as the tendon remains torn.(30) The mean FBL of the supraspinatus in participants with retracted tears in our study was shorter than the mean FBL reported by Kim et al.(4) The difference is likely attributed to the varying lengths of time since the initial tear and symptoms. The mean symptomatic period reported by our participants was 40 weeks, whereas it was 24 weeks in the study by Kim et al.(4) Thus, the tears were more chronic in nature for the participants in the present study.
As the torn tendon is re-attached to its footprint, the applied tension will result in stretching of the muscle and tendon, hence lengthening the fibre bundles. The tension applied to the tendon during repair has been suggested to be an important factor for the repair outcome(31) and the force-generating capacity of a muscle.(10) In the literature, inferior clinical outcomes have been shown to be attributable to the chronicity of tears, as tendons tend to lose their elasticity and the tension required to repair increases significantly.(31,32)
To postulate the changes occurring at the sarcomere level following surgical repair of a torn supraspinatus tendon, we considered the following example. The mean sarcomere length of the normal supraspinatus is 3.0 μm.(29) If the mean FBL of the supraspinatus with an intact tendon is 5.60 cm,(20) there would be approximately 18,666 sarcomeres (number of sarcomeres = length of fibre bundle/length of a sarcomere). The mean FBL of the supraspinatus with a torn tendon, found preoperatively in our study, was 4.52 cm, which would correspond to approximately 15,066 sarcomeres. Following repair, the mean FBL at post-op1 lengthened to 5.38 cm. Assuming that the muscle has not yet had a chance to adapt to the new length change, the number of sarcomeres preoperatively remained consistent and each sarcomere was stretched equally, the new length of each sarcomere at post-op1 would be approximately 3.6 μm. At a sarcomere length of approximately 3.6 μm, there is no overlap between actin and myosin.(33,34) The optimum fibre length for human fibres has been reported to be 2.7 μm.(35) On a length tension curve, a change in the sarcomere length from 2.7 μm to 3.6 μm could result in a 30%–40% decrease in the force-producing capability of a muscle.(34) Thus, in theory, tension applied to the musculotendinous unit during surgical repair to re-attach the tendon will lengthen each sarcomere to an extent beyond their optimal length. This, in turn, will compromise the contractile force of the muscle owing to the decrease in the myofilament (actin-myosin) overlap.(36) To exacerbate this suboptimal state, in comparison to other rotator cuff muscles, the supraspinatus has been reported to be more sensitive to stretch because of its short fibres and long resting sarcomere lengths,(37) leading to fewer sarcomeres for a given FBL.
Several animal studies have shown that skeletal muscles have the ability to adapt to lengthened or shortened positions by increasing or decreasing the number of sarcomeres, respectively.(38-40) In addition to adapting to new lengths by adding or decreasing the sarcomere number in series, these studies have shown that skeletal muscles can maintain optimal sarcomere length and thus, their maximal force-producing capability. However, this mechanism of sarcomere adaptation may only apply to particular muscles in animals. The results are not easily extrapolated to human muscles.(41) This finding may be true for the highly adaptive soleus muscle of rodents but is not necessarily true even for other muscles of the rodent hind limb.(42) In addition, Fridén and Lieber(41) found that in several tendon transfer cases in humans, the adjusted sarcomere length following surgery was greater than the optimal sarcomere length.
In a cadaveric study that investigated the sarcomere length of rotator cuff muscles with intact tendons and those with full-thickness tendon tears, no significant differences in sarcomere lengths of the supraspinatus were reported between muscles with torn tendons and those with intact tendons.(29) With a chronic supraspinatus tendon tear, it may be possible that over time, the number of sarcomeres would decrease in order to regain their optimal length. However, whether this sarcomere adaptation occurs in human muscles following lengthening with surgical repair has yet to be investigated. Furthermore, it has been postulated that the sarcomere number-sensing mechanism may be disrupted or may respond inappropriately with excessive stretch during surgical repair.(43)
Different methods have been suggested in the literature to avoid over-tensioning during surgical repair. Continuous lengthening of the muscle-tendon unit has been suggested in a previous animal study to improve tendon-to-bone healing. This involves continuous progressive traction onto the tendon and muscle.(44) However, this is not a one-step surgical procedure. Use of reinforcement grafts and human dermal allografts has also been suggested to repair large and massive tears to enhance healing and restore muscle function.(45) However, neither method is used regularly, and these techniques are technically more challenging.
Although tendon properties were not investigated in this study, it is important to consider the changes in mechanical and structural properties of the tendon that occur with time, from injury to repair. The amount of tension applied to the tendon is dependent on the elasticity of the tendon during surgical repair. In a muscle with short FBL such as the supraspinatus, the force-producing capability of the muscle is sensitive to changes in the tendon length.(10) In a rat shoulder model, Gimbel et al(46) reported an initial decrease in tendon stiffness after tendon detachment, followed by a progressive increase with time. Furthermore, in another study using the same animal model, Gimbel et al(32) demonstrated a rapid increase in musculotendinous stiffness and tension required at repair to reappose tendon to bone in the first few weeks after the tendon tear. Similar results (i.e. an increase in stiffness of the musculotendinous unit) have been reported in chronic rotator cuff tears in humans.(31)
A significant reduction in FBL from post-op2 to post-op3 was observed in our study. This pattern of FBL reduction could be attributed to the specific adaptation patterns of the supraspinatus muscle and tendon to the surgical repair. In an animal study, Takahashi et al(47) found an asynchronous pattern between the muscle and tendon in response to a chronic length increase in the extensor digitorum of the second toe. They found an initial rapid increase in the number of sarcomeres in response to the stretch. This increase in sarcomeres was then reversed following the delayed adaptive response of the tendon. It was thought that the muscle adapted to the tendon elongation by subtracting the number of sarcomeres. Hence, at the end of eight weeks, there was no significant difference between the tendon and muscle length of the experimental and control groups.(47) It is possible that this process of muscle and tendon adaptation takes longer than eight weeks in the human supraspinatus. Factors such as the nature of injury (acute vs. chronic), muscle investigated (extensor digitorum vs. supraspinatus) and subjects studied (animals vs. humans) could influence the length of time required for both the muscle and tendon to adapt. Recently, Meyer et al(30) found a dramatic increase in tendon length (8 mm) postoperatively in patients with full-thickness tears using MR imaging performed at an average of 24 (range 6–50) months after surgery. They hypothesised that this ‘pseudo’ lengthening of the tendon could be due to unpacking of the central tendon owing to degeneration and subsequent absorption of atrophied and retracted muscle fibres.
Postoperative rehabilitation is another important factor in the structural and functional recovery of the musculotendinous unit and may explain the significant reduction in FBL observed from post-op2 to post-op3. The muscular toning stage, which generally starts at three months after the repair and may last up to six months depending upon the recovery of the patient, involves strengthening exercises, facilitation of functional movements to a near normal range, and work- and sports-specific activities.(48) These rehabilitation exercises may stimulate the muscle to adjust and adapt to its length, allowing the muscle to optimally contract. Further studies are needed to confirm these hypotheses.
Finally, another possible reason for a significant reduction in FBL could be a re-tear. As mentioned earlier, the re-tear rate following rotator cuff repair is very high.(16) Thus, the reduction in FBL seen at post-op3 may be a result of surgical failure. However, based on the significant improvement in WORC scores, re-tear was not likely the cause in this sample.
Preoperative PA measurements in full-thickness tears at rest (18.13º ± 1.55º) found in this study are comparable to those reported by Thompson et al(49) (17.6º ± 8.6º) and Zuo et al(13) (18.6º ± 10º) using MR imaging and Photoshop imaging software, respectively. In contrast to FBL, a decrease in PA was found at post-op1, likely owing to stretching of fibres in the plane of the muscle fibres. The fibre bundle changes observed at post-op1 are consistent with a previous animal study(50) and reflect lengthening of the muscle.
A significant increase in PA over time observed in the present study may be explained by the adaptation and reduced tension on the muscle fibres and the hypertrophy induced by postoperative rehabilitation. Despite the shortening of muscle fibres and significant increase in PA, no significant changes in MT were seen at any time point. A possible explanation for this is that the muscle was initially inflamed owing to the tension applied to stretch the muscle and microtrauma induced by re-attaching the tendon to its insertion. At post-op2, still no change was observed in MT. Inflammation at 12 weeks post surgery would likely have subsided, so the lack of change could be explained by new muscle fibre growth, accounting for the space formerly occupied by inflammation. These changes cannot be easily differentiated using US. Although we did not observe significant changes in MT at post-op3, a trend towards an increase in MT was observed, likely as a result of postoperative rehabilitation.
For the WORC, the decrease in composite score with time indicates improvement in perceived function after surgery. In order to assess a true clinically meaningful change in function from one time point to the other, a minimum change of 245 points in the WORC has been suggested.(28) In our study, we observed a drop in composite score of more than 245 points from pre-op to post-op2 and post-op3. There was a change of 1,001 points (an increase of 47.67% in the overall function) from the preoperative period to six months post surgery. Our findings of improvements in patient-reported outcomes following surgery are in agreement with previous studies.(51-53)
Our study has several limitations. First, the sample size was small and a power estimate to determine the sample size was not performed. In addition, two participants did not attend the post-op3 session for personal reasons and their data was not included in the study. This further compounded the limitation of small sample size. However, a conservative approach for statistical analysis (Bonferroni adjustments) was taken in this study. Second, strength testing and the WORC were added to the protocol to measure functional improvement after the first three participants had already been tested. Third, we did not assess the interactions between strength gains and WORC scores owing to our small sample size. Fourth, we did not assess the sarcomere length of the supraspinatus. Quantification of sarcomere length changes with stretching of the tendon during surgical repair may have provided a better understanding of the functional recovery of the muscle and the amount of tension that can be applied without damaging the sarcomeres. Fifth, we investigated the anterior region of the muscle, which produces most of the force; however, the architecture of the posterior region of the supraspinatus also needs to be investigated, as the posterior aspect of the supraspinatus tendon has been shown to be significantly affected by tendon injury.(4)
In conclusion, repair of the supraspinatus tendon during surgery lengthens the fibre bundles of the muscle. The amount of lengthening from pre-op to one month after the surgical repair likely results in overstretching of the sarcomeres, affecting the length-tension relationship of the muscle. The amount of lengthening needed may be greater with more chronic tears owing to even further reductions in FBLs. Overall, this lengthening can compromise muscle function and may be associated with the prevalence of surgical failure. Future studies should investigate any changes in tendon integrity (i.e. presence of re-tears) in addition to these architectural parameters in a larger clinical population, with different surgical repair techniques. This may better inform surgeons regarding the impact of repair timing (i.e. early vs. delayed) and provide insights on the influence of different surgical repair techniques on architectural parameters and functional improvements.
ACKNOWLEDGEMENT
The study was conducted with financial support from the Saskatchewan Health Research Foundation, Canada.


