Dear Sir,
The liver is the internal organ that is most commonly affected in cases of drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome.(1) While Cytomegalovirus (CMV) reactivation has been described in patients with DRESS syndrome, to our knowledge, histologically confirmed CMV hepatitis has never been described in patients with DRESS syndrome. We herein report a case that highlights the complexity in the diagnosis and treatment of CMV hepatitis in a DRESS patient who received immunosuppressants.
A 44-year-old woman with gout was started on allopurinol and colchicine. Five weeks later, she presented to the outpatient clinic with ten days of fever and a pruritic dermatosis. Physical examination revealed discrete and confluent erythematous maculopapular lesions affecting her face, trunk and limbs (body surface area [BSA] 70%), and anasarca. Relevant negative findings included hepatosplenomegaly, absence of lymphadenopathy, and stigmata of chronic liver disease. Laboratory tests showed an elevated eosinophils count of 2.4 × 109/L, presence of atypical lymphocytes, raised levels of aspartate transaminase (AST; 585 IU/L), alanine transaminase (ALT; 633 IU/L), alkaline phosphatase (535 IU/mL) and total bilirubin (136 µmol/L), increased creatinine clearance of 11.8 mL/min and prothrombin time of 13.4 seconds. Urinary eosinophils were detected.
A dermatologist was consulted and a diagnosis of DRESS syndrome was made based on the RegiSCAR criteria(2) – fever, Grade 2 eosinophilia, skin involvement with typical lesions suggestive of DRESS (> 50% BSA), and cardiac, renal and liver involvement. Skin biopsy showed basal vacuolar degeneration with apoptotic keratinocytes demonstrating perivascular infiltrate of lymphocytes and eosinophils suggestive of DRESS. Allopurinol was ceased immediately and prednisolone 60 mg/day (1 mg/kg) was initiated in view of cardiac and renal involvement. At the time of presentation, the patient’s CMV immunoglobulin M and cytospin were both negative.
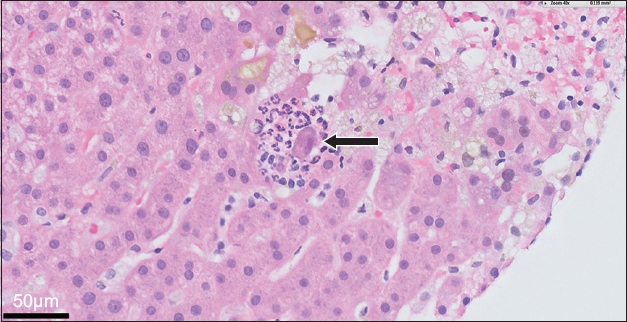
After three weeks of being on prednisolone, the patient’s liver biochemistries showed a mixed pattern with worsening cholestasis. The working diagnosis was allopurinol-induced DRESS syndrome or viral hepatitis (Hepatitis A, B, C and E as well as CMV in an immunocompromised host).(3) Serology for Hepatitis A, B, C and E and autoimmune liver diseases, and blood cultures were negative. Abdominal ultrasonography and magnetic resonance cholangiopancreatography did not reveal any intrahepatic or extrahepatic biliary dilatation. Serial CMV DNA polymerase chain reactions showed CMV reactivation with rising CMV DNA titre from < 150 copies/mL to 10,370 copies/mL. As histology is the gold standard for the diagnosis of invasive CMV hepatitis, liver biopsy was performed.(4) The presence of scattered inclusion bodies and surrounding microabscesses from liver biopsy confirmed the diagnosis of CMV hepatitis (
Fig. 1
Photomicrograph shows ‘owl-eye’ appearance (arrow) of inclusion bodies with surrounding neutrophilic infiltrates and microabscesses suggestive of Cytomegalovirus hepatitis (Haemotoxylin & eosin, × 40).
Intravenous ganciclovir was initiated for the patient and the dosage of prednisolone was halved. As the CMV DNA titre continued to rise to 1,043,055 copies/mL, the patient also received intravenous artesunate and two doses of intravenous immunoglobulin, after which the CMV titre started to decline, liver biochemistries started to improve, and ALT and AST levels normalised. On Day 28 of admission, the patient developed Escherichia coli bacteraemia. She suffered multiple episodes of ventricular fibrillation before her demise on Day 44 of admission.
DRESS syndrome is a severe adverse cutaneous reaction with multiorgan involvement. It has a reported prevalence of 3.6 in 1,000 hospitalised patients following drug exposure.(5) Reactivation of CMV resulting in CMV viraemia has been reported in 17.6% of patients with DRESS syndrome.(6,7) CMV reactivation may result in multisystemic manifestations, including gastrointestinal bleeding,(8) glomerulonephritis, myocarditis(9) and pneumonitis. Immunosuppressants are the cornerstone of treatment in DRESS syndrome after drug cessation, but they can be detrimental if there is CMV reactivation resulting in end-organ disease.
The diagnosis of CMV hepatitis was challenging in this patient for a few reasons. Firstly, as deranged liver biochemistries are common in DRESS syndrome, CMV infection may be overlooked. Secondly, prolonged recovery was expected in this patient with renal impairment. This was because the excretion of the toxic metabolites of allopurinol and oxypurinol is dependent on renal function, and oxypurinol itself can cause interstitial nephritis, resulting in a vicious cycle that delays recovery. Lastly, the reported demographic risk factors for CMV infections in patients with DRESS syndrome were absent in this case. A retrospective case series of DRESS syndrome reported by Yusuke et al showed that CMV infection was more common in older males.(8) In our patient, persistent derangement in liver biochemistries and a rising CMV viral load led to the diagnosis of CMV hepatitis, which was confirmed on liver biopsy. To our knowledge, this is the first case report of histological confirmation of CMV hepatitis in a patient with DRESS syndrome.
The exact mechanism of CMV reactivation in patients with DRESS syndrome remains uncertain. CMV reactivation can be a consequence of immunosuppressive treatment used in DRESS syndrome, or a consequence of disease. Immunosuppressants, which are often used to treat DRESS syndrome, impair host immunity and may result in reactivation of CMV.(10) Ishida et al reported a higher CMV viral load in 34 subjects with DRESS syndrome who were treated with corticosteroids compared with those who were not.(7) Interestingly, reactivation of CMV has also been reported in patients with DRESS syndrome who were not receiving immunosuppressants.(6) As CMV infections may alter immune response, early initiation of treatment is beneficial. Sequential monitoring of CMV viral load in patients with DRESS syndrome allows pre-emptive treatment of CMV viraemia.
Unlike immunocompetent hosts with CMV infection who do not require treatment, CMV reactivation in critically ill immunocompromised hosts is associated with poorer outcome.(11,12) After two weeks of intravenous ganciclovir and artesunate, the patient’s CMV viral load fell from 1,403,055 copies/mL to 370,987 copies/mL and her liver biochemistries improved, with normalisation of ALT and AST levels. This suggests that we should consider pre-emptive treatment of CMV viraemia in patients with DRESS syndrome before it progresses to CMV disease.
This case highlights the complexity of diagnosis and management of CMV hepatitis as an uncommon complication of DRESS syndrome. Sequential monitoring for CMV viraemia in DRESS syndrome allows early diagnosis and prompt pre-emptive treatment. Physicians should have a high index of suspicion for CMV hepatitis in DRESS syndrome, especially when there are deranged liver biochemistries with prolonged recovery.
Yours sincerely,


