Abstract
About one-third of patients with suspected gastro-oesophageal reflux disease (GERD) do not respond symptomatically to proton pump inhibitors (PPIs). Many of these patients do not suffer from GERD, but may have underlying functional heartburn or atypical chest pain. Other causes of failure to respond to PPIs include inadequate acid suppression, non-acid reflux, oesophageal hypersensitivity, oesophageal dysmotility and psychological comorbidities. Functional oesophageal tests can exclude cardiac and structural causes, as well as help to confi rm or exclude GERD. The use of PPIs should only be continued in the presence of acid reflux or oesophageal hypersensitivity for acid reflux-related events that is proven on functional oesophageal tests.
Ms Olivia has been troubled by symptoms that resemble gastro-oesophageal reflux disease since her 40th birthday several years ago. She was prescribed proton pump inhibitors by several different doctors and has been taking them regularly with no improvement. She underwent gastroscopy about five years ago that reported normal results. In recent months, she underwent a cardiac evaluation that was also normal. She did not experience dysphagia, weight loss or vomiting.
WHAT IS GASTRO-OESOPHAGEAL REFLUX DISEASE?
The Montreal definition proposed that gastro-oesophageal reflux disease (GERD) is a condition that develops when the reflux of gastric contents into the oesophagus causes troublesome symptoms and/or complications.(1) Heartburn and regurgitation are typical symptoms of GERD. The Montreal consensus also includes a number of extra-oesophageal symptoms potentially attributed to GERD.(1) It further defines ‘troublesome’ symptoms as an impairment in health-related quality of life for ≥ 2 days/week for mild symptoms or ≥ 1 day/week for moderate symptoms. The latest Rome IV classification proposes a more restrictive definition of GERD, limiting it to patients who have endoscopic features of reflux oesophagitis and/or elevated oesophageal acid exposure on functional oesophageal tests.(2)
HOW COMMON IS THIS IN MY PRACTICE?
Although GERD is a predominantly Western disease, with a prevalence of 10%–20% in North America and Western Europe,(3) changing dietary patterns and a rise in obesity have resulted in the increasing prevalence of GERD in Asia.(4) The prevalence of symptom-based GERD in Southeast Asia has been rising(5) and was estimated to be 6.3%–18.3% from 2005–2010.(6)
Current guidelines recommend the use of acid-suppressive therapy with proton pump inhibitor (PPI) therapy as the first-line approach to GERD treatment.(7-9) PPIs suppress gastric acid secretion and have a profound effect on oesophageal mucosal healing.(10) Despite the high efficacy of PPIs, up to 30% of patients continue to experience GERD-like symptoms even when adequately dosed.(7,8) Patients who do not respond to PPIs or have any alarm symptoms (e.g. dysphagia, odynophagia, weight loss, vomiting and/or abdominal pain) require further evaluation. Gastroscopy is useful to exclude sinister conditions, especially in patients who have additional risk factors such as smoking, older age and a family history of upper gastrointestinal cancers.
After exclusion of any underlying serious aetiology, it is not uncommon for patients to continue to experience GERD-like symptoms. With the widespread use of PPIs, the failure to resolve GERD symptoms is now one of the most common presentations of GERD in gastroenterology clinics as well as the general practice setting. Patients who have atypical symptoms, such as unexplained chronic cough and throat symptoms, and do not respond well to PPIs are also commonly encountered. In such situations, the diagnosis of GERD remains doubtful(11,12) and ambulatory oesophageal reflux monitoring is useful to determine if the symptoms are indeed due to GERD.(12)
GERD is a costly disease, especially if PPI treatment failure results in patients seeking a second opinion, or receiving a higher dose or different course of PPIs. Compared to patients with erosive oesophagitis, patients with non-erosive reflux disease (i.e. the endoscopic features of reflux esophagitis are absent) have a 20% reduction in therapeutic gain from PPIs.(13) Apart from the high treatment costs, it has been observed that up to 55% of patients with persistent GERD symptoms report an impaired quality of life.(14)
WHAT CAN I DO IN MY PRACTICE?
History
There is no gold standard for the diagnosis of GERD. In our daily practice, we often rely on the subjective reporting of a constellation of symptoms and attribute these to GERD. For patients who fail to respond to PPIs, a variety of causes are possible, both GERD-related and non-GERD-related.
A good history can clarify the nature of the patient’s symptoms and which symptoms respond or persist despite PPI therapy. Heartburn is characterised by a painful retrosternal burning sensation lasting for a short duration. Regurgitation is described as a backflow of gastric contents into the chest or mouth. Establishing compliance to PPIs, including the timing of medications and proper dosages, is important in the initial evaluation. PPIs are typically started at a standard dose on a once-daily basis. If the patients do not respond, the physician can increase the dose to twice daily for a defined period of time (approximately eight weeks) and monitor the response to establish if the patients should continue taking PPIs for presumptive GERD or undergo further diagnostic tests. In clinical trials, PPIs are more efficacious for relieving symptoms of heartburn compared to regurgitation.(15,16)
It is also important to establish if a non-GERD-related aetiology (
Box 1
Causes of persistent reflux-like symptoms despite PPIs

Functional gastro-oesophageal disorders have a negative impact on treatment outcomes.(19) Based on the latest Rome IV criteria,(2) functional heartburn and reflux hypersensitivity are part of the functional oesophageal disorders spectrum. These are characterised as disorders presenting with typical oesophageal symptoms that are not explained by a structural lesion, oesophageal motility disorder or underlying GERD. A diagnosis of functional oesophageal disorder has an arbitrary requirement of symptom onset at least six months before diagnosis and symptoms of at least three months’ duration.(2) In addition, patients with functional heartburn may have symptom overlap with functional dyspepsia(20) or irritable bowel syndrome.(21)
Questionnaires
Various questionnaires have been employed to diagnose GERD, but as these are neither sensitive nor specific for reflux,(22) their use does not increase diagnostic accuracy.(23) The GerdQ questionnaire(24) comprises positive predictor questions about heartburn and regurgitation, and negative predictor questions about epigastric pain and nausea. Its sensitivity (65%) and specificity (71%) for GERD diagnosis were reported to be comparable to the clinical judgment of gastroenterologists.(25) However, most of these questionnaires lack sensitivity and specificity, and cannot reliably distinguish GERD from functional dyspepsia. Language barriers, complexities in symptom description and cross-cultural differences are the main difficulties preventing the widespread use of GERD questionnaires in our local population.(26)
Response to proton pump inhibitors
Response to PPIs remains one of the most specific predictors of GERD and is useful in the primary care setting. The PPI test consists of measuring the symptomatic response to a 1–2-week course of a high-dose PPI in patients with GERD symptoms. The rationale for a high-dose PPI (typically a double dose) is the need to suppress gastric acid secretion and heal erosive oesophagitis.(27)
There is a lack of consensus on the definition of a PPI non-responder.(28) The dose of PPIs and duration of treatment required to fulfil the criteria is not well defined, varying from single to double dose, and ranging from 8–12 weeks, respectively. Sifrim and Zerbib(29) defined refractory GERD as heartburn and/or regurgitation symptoms occurring at least three times per week despite a stable double dose of PPI, during a treatment period of at least 12 weeks. In a prospective study of 544 patients with typical GERD symptoms, a 75% symptom response to a double dose of PPIs of one week’s duration showed a sensitivity of 96.5% (95% confidence interval [CI] 94%–98%) and specificity of 34.6% (95% CI 25%–43%) for a diagnosis of GERD.(30)
Lifestyle changes
Dietary modifications and lifestyle measures are useful for GERD patients. Weight loss in patients who are overweight or have recent weight gain has been proven to improve GERD symptoms, as well as elevation of the head of the bed.(31) Patients should also avoid a supine position immediately after meals and having meals up to two hours before bedtime. Dietary triggers for reflux include caffeine, chocolate, carbonated beverages and foods with high fat content.
WHAT INVESTIGATIONS ARE PERFORMED IN A SPECIALIST SETTING?
Gastroscopy
The American Society for Gastrointestinal Endoscopy recommends endoscopy for patients with GERD symptoms that persist despite appropriate medical therapy.(32) Gastroscopy has poor sensitivity(26,33) but good specificity (90%–95%)(33) for the diagnosis of GERD in the presence of the endoscopic features of reflux oesophagitis, peptic strictures, erosions, ulcers and Barrett’s oesophagus. However, the majority of patients with refractory reflux symptoms will have normal endoscopy. Nevertheless, gastroscopy remains an important screening tool to exclude an underlying organic lesion of the upper gastrointestinal tract and allow for oesophageal biopsies in conditions such as pill-induced oesophagitis, skin diseases with oesophageal involvement, peptic ulceration of the oesophagus and eosinophilic oesophagitis.
Functional oesophageal tests
In patients who remain symptomatic despite PPIs, functional oesophageal tests provide a useful objective measure of oesophageal function after exclusion of any structural lesion or cardiac cause. Oesophageal manometry is an important diagnostic tool to exclude achalasia(34,35) as well as other major motility disorders, including oesophageal spasm, aperistalsis and hypercontractile oesophagus.(36) Ambulatory 24- or 48-hour oesophageal pH studies or 24-hour combined pH-impedance studies are well-described diagnostic modalities used by gastroenterologists to confirm or exclude GERD as a cause of persistent symptoms. This would avoid the unnecessary use of PPIs when there is no objective evidence of GERD.
Oesophageal pH monitoring
Ambulatory reflux monitoring plays an important role in the diagnosis of GERD.(37) Oesophageal pH monitoring can be performed using either the conventional nasopharyngeal pH catheter monitoring system, for a period of 24 hours, or the wireless 48-hour pH monitoring device (i.e. Bravo® capsule). Compared to the nasopharyngeal catheter system, the wireless capsule is cosmetically more acceptable to most patients, as it is attached to the lower oesophageal mucosa and allows for a longer duration of monitoring of up to 48 hours. Based on normal values that were previously published for the 24-hour nasopharyngeal catheter system(38) and wireless Bravo capsule,(39) we can objectively quantify oesophageal acid exposure (usually expressed as the percentage of time during which pH < 4). GERD is diagnosed in the presence of an elevated oeosphageal acid exposure time. In addition, ambulatory reflux monitoring allows physicians to establish the temporal association between the patient’s symptom episodes and acid reflux events. A symptom episode is considered to be reflux-related if symptom onset occurs within two minutes of a reflux event. The importance of symptom association in a patient who continues to have persistent reflux-like symptoms is that it allows us to distinguish reflux hypersensitivity (positive symptom association) from functional heartburn (negative symptom correlation).(2)
Combined pH-impedance monitoring
In recent times, there has been an increasing awareness of non-acid reflux as a cause of persistent symptoms. Unlike the conventional 24-hour nasopharyngeal pH monitoring device, which measures only events related to acid reflux, the combined pH-impedance monitoring device can measure both events related to acid reflux (pH < 4) and non-acid reflux (pH ≥ 4). In addition, it permits further characterisation of the nature of the refluxate (i.e. air, reflux or mixed air-liquid reflux),(40) and the number of acid and non-acid reflux events. Apart from the objective measurement of reflux events, pH-impedance studies also allow the patient’s symptoms to be directly correlated with both acid and non-acid reflux events. Studies have shown that non-acid reflux accounts for persistent reflux-like symptoms in patients who respond poorly to PPIs.(41,42) Based on the results of functional diagnostic tests, the causes of PPI-refractory GERD can be classified as reflux-related or non-reflux-related (
Fig. 1
Flow chart shows stratification of patients evaluated for persistent gastro-oesophageal reflux disease (GERD) symptoms. AET: acid exposure time; NERD: non-erosive reflux disease; SAP: symptom association probability; SI: symptom index
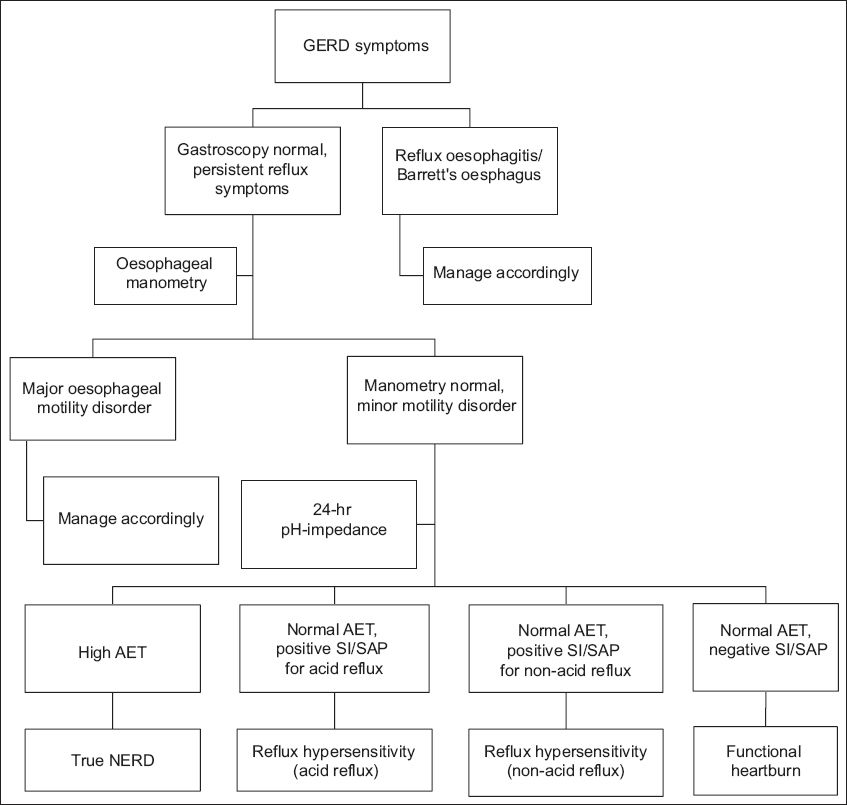
For PPI non-responders who have an elevated number of reflux episodes on 24-hour pH-impedance studies, the use of transient lower oesophageal sphincter relaxation (tLESR) inhibitors can be considered. Baclofen, a gamma-aminobutyric acid agonist that reduces the frequency of tLESRs and number of reflux events, can be beneficial for patients who continue to have persistent reflux symptoms despite PPIs. However, the central side effects of baclofen, especially dizziness and drowsiness, have restricted its widespread use. Once the diagnosis of a functional oesophageal disorder has been made, appropriate treatment should be instituted, while PPIs should be stopped. The use of visceral pain modulators may be beneficial in patients who have functional heartburn or oesophageal hypersensitivity.(44-46) However, psychological comorbidities can also affect oesophageal perception, resulting in reduced pain thresholds.(45) The use of pain modulators, including tricyclic antidepressants, selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors, was reported to be beneficial.(46)
Ms Olivia underwent gastroscopy, which returned normal results, and oesophageal manometry to exclude any underlying oesophageal motility disorders. Her 24-hour combined pH-impedance study showed a normal oesophageal acid exposure time and negative symptom association profile (i.e. symptoms of heartburn did not correspond to any reflux event during ambulatory reflux monitoring). Further history revealed that she was having difficulty coping with her job while taking care of her three school-going children, as her husband was often overseas. She was referred to a psychologist and underwent cognitive behavioural therapy for underlying anxiety and stress management. Her husband was supportive and changed his job to spend more time at home. At her last clinic visit, Ms Olivia showed much improvement and had stopped her acid suppressants.
CONCLUSION
Despite major advances in our understanding of GERD, management remains a challenge. Due to their established efficacy and safety, PPI treatment is often used as a therapeutic trial to diagnose GERD in the absence of alarm symptoms such as bleeding, weight loss, anaemia or dysphagia.(47) A single dose of PPI provides adequate symptom relief in most patients; however, some may require dose escalation to twice a day. Patients unresponsive to PPI therapy are often labelled as having refractory GERD.
Failure to respond to a complete course of PPIs should alert the clinician to a non-GERD cause. The continued use of PPIs without objective evidence of GERD often leads to high costs. Once a structural cause has been excluded following gastroscopy, discussion with the patient on the role of further functional evaluation, including manometry and ambulatory studies, is useful to obtain a definitive diagnosis. This would avoid unnecessary costs from the overuse of PPIs and identify patients with functional heartburn. Treatment options for functional heartburn include a trial of pain modulators or psychological treatment such as cognitive behavioural therapy and muscle relaxation techniques.
TAKE HOME MESSAGES
-
GERD is a common condition, but overdiagnosis (especially in patients with atypical manifestations) leads to the unnecessary prescription of PPIs and high costs for patients.
-
Primary care physicians play a crucial role in assessing the patient and should take into account the symptom profile as well as any underlying psychological comorbidities.
-
The presence of any alarm symptoms (e.g. dysphagia, odynophagia, weight loss, abdominal pain and/or vomiting) warrants early referral to a gastroenterologist for further evaluation.
-
Start PPIs at a once-daily standard dose and monitor the patient’s response. Patients who continue to remain symptomatic should be referred for further evaluation. The most common first-line investigation is gastroscopy. In patients without structural lesions, functional oesophageal tests that gastroenterologists may perform include oesophageal manometry and 24-hour ambulatory pH-impedance studies. These tests measure oesophageal acid exposure and reflux numbers, and assess the temporal association between the patient’s symptom episodes and reflux events.
-
In the absence of high oesophageal acid exposure or a positive symptom association, repeated prescriptions for PPIs and escalation of acid-suppressive therapy are ineffective and lead to unnecessary costs.
SMJ-57-551.pdf


