Abstract
INTRODUCTION
This study aimed to describe the clinical presentation, treatment and outcome of patients with relapsing polychondritis (RP) who were seen at a large tertiary-care academic medical institution in Singapore.
METHODS
The medical records of all patients diagnosed with RP at the Department of Rheumatology and Immunology, Singapore General Hospital, Singapore, between 2005 and 2013 were reviewed. The diagnosis of RP was made using the modified McAdam criteria.
RESULTS
Ten patients were diagnosed with RP during the study period. Among these patients, five fulfilled the modified McAdam criteria and five were probable cases of RP. The most common clinical presentations were auricular chondritis (n = 9), episcleritis or scleritis (n = 5), and large airway involvement (n = 3). All of the patients received prednisolone. Five patients developed haematological disorders. Patients with both RP and haematological disorders had a longer duration of RP symptoms prior to diagnosis, compared to patients with RP who did not develop haematological disorders (average duration of symptoms 14.7 months vs. 4.2 months).
CONCLUSION
The high frequency of patients with haematological malignancies in this series was unexpected. Myelodysplastic syndrome has been reported, but other haematological malignancies are rarely associated with RP. As the association between haematological malignancies and RP is currently unclear, the threshold for haematological/lymphoproliferative screening should be lowered in patients with RP.
INTRODUCTION
Relapsing polychondritis (RP) is a rare immune-mediated condition, which is characterised by recurrent and progressive inflammation of cartilaginous structures and proteoglycan-rich structures.(1) RP particularly affects the cartilaginous areas of the ears and nose, and it is not uncommon for the eyes, joints and respiratory tract to be affected as well. RP can occur in isolation or in association with other diseases. The objective of the present study was to describe the clinical presentation, treatment and outcome of patients with RP who were seen at a large tertiary-care academic medical institution in Singapore.
METHODS
Singapore General Hospital is a 1,700-bed tertiary-care academic medical institution in Singapore. In this study, we retrospectively reviewed the medical records of all patients diagnosed with RP at the Department of Rheumatology and Immunology between January 2005 and December 2013. The medical records that were retrieved and reviewed included information on both inpatient and outpatient visits.
The diagnosis of RP was made according to the modified McAdam criteria.(2) The McAdam criteria suggest that a diagnosis of RP is almost certain if at least three of the following six criteria are present: (a) bilateral auricular chondritis; (b) non-erosive seronegative inflammatory polyarthritis; (c) nasal chondritis; (d) ocular inflammation such as conjunctivitis, keratitis, scleritis/episcleritis and uveitis; (e) respiratory tract chondritis such as inflammation of the laryngeal and/or tracheal cartilages; or (f) cochlear and/or vestibular dysfunction such as neurosensory hearing loss, tinnitus and vertigo. According to the modified McAdam criteria, a diagnosis of RP can be made if one of the following criteria is fulfilled: (a) at least three of the clinical criteria stated in the McAdam criteria; (b) one or more of the McAdam criteria with positive histological confirmation; or (c) chondritis at two or more separate anatomic locations with response to steroids and/or dapsone.
The clinical presentation, laboratory results, treatment response and clinical outcome of the patients were recorded using a standardised data collection sheet; the collected data was then summarised. This study design received approval from the local institutional review board.
RESULTS
A total of ten patients with RP symptoms were seen at the Department of Rheumatology and Immunology, Singapore General Hospital, Singapore, during the study period. Five of these patients fulfilled the modified McAdam criteria, while the remaining five patients fulfilled only part of the diagnostic criteria and were thus classified as probable cases of RP. The average age of onset of RP was 48 (range 22–72) years and the average duration of follow-up was 3.5 years (range 17 months–7 years). The patients were all of Chinese ethnicity and the female-to-male ratio was 7:3. The clinical presentation, laboratory results, and therapy and clinical outcomes of the ten patients are summarised in
Table I
Summary of the clinical presentation, laboratory results, and therapy and clinical outcomes of the patients with relapsing polychondritis (RP) (n = 10).
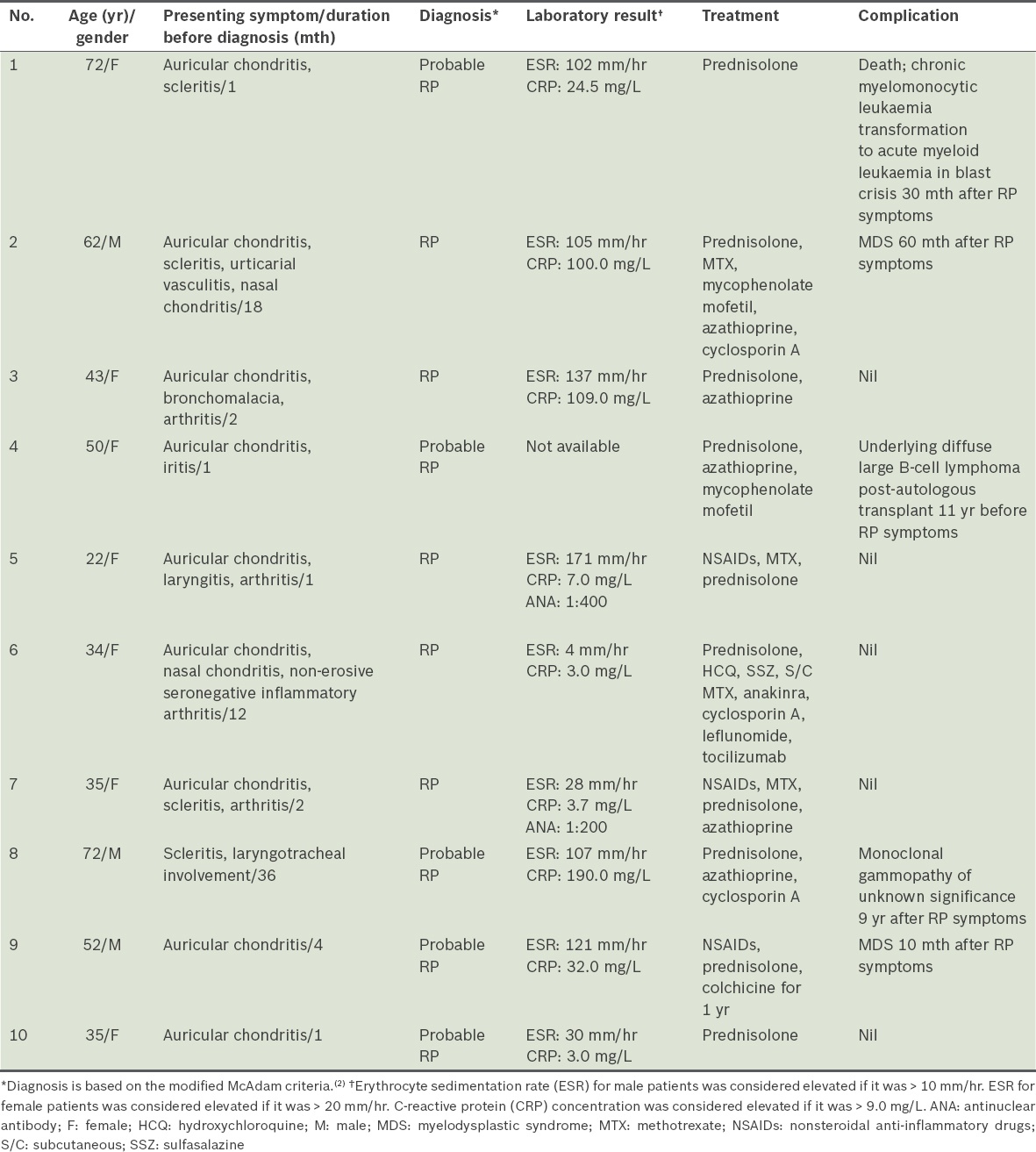
The most common clinical presentation in our study cohort was auricular chondritis (9/10 patients, 90%). The next most commonly affected organ was the eyes, with episcleritis and scleritis being the presenting complaint in five patients. Three patients had large airway involvement (two were diagnosed using bronchoscopy and one using computed tomography). One of the three patients with large airway involvement (Patient 3) developed severe bronchomalacia three years after the diagnosis and required continuous positive airway pressure support.
Inflammatory markers were generally elevated in the patients with RP. An elevated erythrocyte sedimentation rate (ESR) was observed in 80.0% of the patients at their first presentation, as well as during acute exacerbation. An elevated concentration of C-reactive protein (CRP) was present in 50.0% of patients at their first presentation.
All ten patients received courses of prednisolone at least once during their treatment; the maximum dose administered was 60 mg. The longest duration of steroid treatment was eight years (in Patient 3, who had features of bronchomalacia). Although three patients were treated with nonsteroidal anti-inflammatory drug (NSAID) monotherapy for a month, they were later switched to prednisolone due to a lack of response to the NSAID. All except one patient required second-line agents. The second-line agents used included azathioprine, methotrexate, cyclosporin A, mycofenolate mofetil, hydroxychloroquine, sulfasalazine and leflunomide. One patient (Patient 6) received anakinra, a biologic therapy, but was later initiated on tocilizumab every 4–6 weeks in view of the patient’s poor response to the previous therapy. At the time of writing, this patient was still on tocilizumab every 4–6 weeks.
Five of the ten patients in this case series had haematological disorders. One patient (Patient 4) was diagnosed with diffuse large B-cell lymphoma 11 years before the diagnosis of RP, while the remaining four patients developed haematological disorders after the diagnosis of RP. In the latter four patients, the haematological disorder that developed was myelodysplastic syndrome (MDS). The patients with both RP and haematological disorders had a longer duration of RP symptoms prior to diagnosis, compared to the patients with RP who did not develop haematological disorders (average duration of symptoms 14.7 months vs. 4.2 months).
DISCUSSION
Interestingly, despite the multi-ethnic composition of Singapore’s population (74.3% Chinese, 13.4% Malay and 9.1% Indian), all of the patients with RP in the present study were of Chinese ethnicity.(3) It is unclear whether the lack of representation of the other ethnic groups was related to ethnic-specific factors or the presence of selection bias. More female (n = 7) than male (n = 3) patients were affected in this case series, although previous series reported that RP occurs at the same frequency in both genders.(2)
The most common presenting feature in our case series was auricular chondritis, which was seen in nine out of the ten patients. In contrast, a case series conducted by Kong et al reported that airway complication was the most common presenting feature of RP in their Oriental population.(4) Airway complications were seen in only three of our ten patients. One of these three patients developed severe bronchomalacia three years after the diagnosis of RP while on a tapering dose of prednisolone; this patient required home continuous positive airway pressure therapy. Nasal chondritis has been reported to be present in 61% of patients with RP over the course of the disease.(5) However, this condition was only present in two of the patients in our case series, despite our study’s relatively long duration of follow-up (the longest duration of follow-up was seven years). One of the patients in our series was pregnant when the RP was active, but the disease did not affect the baby. One case study, which was published in 1960, reported that the baby of a woman with RP was affected at birth.(6)
Eight of the ten patients in our case series had elevated ESR at presentation and during flare. Pearson et al reported raised ESR in 80% of their patients with RP.(7) On the other hand, CRP levels were elevated in only five of the ten patients in our series. This suggests that ESR may be a more useful biomarker than CRP for the diagnosis and monitoring of RP activity in our study cohort.
An unusual finding in the present study was the high incidence of haematological malignancy in our cohort. Five of the ten patients had associated haematological malignancy. The association of RP with MDS was previously described by Hebbar et al, who reported that up to 27% of their RP patients had concomitant MDS.(8) Two patients in our case series developed MDS after being diagnosed with RP; one of them received azathioprine (50–100 mg) for six months.
Table II
Summary of cases of relapsing polychondritis (RP) and its association with haematological malignancy, as reported in the literature and the present study.
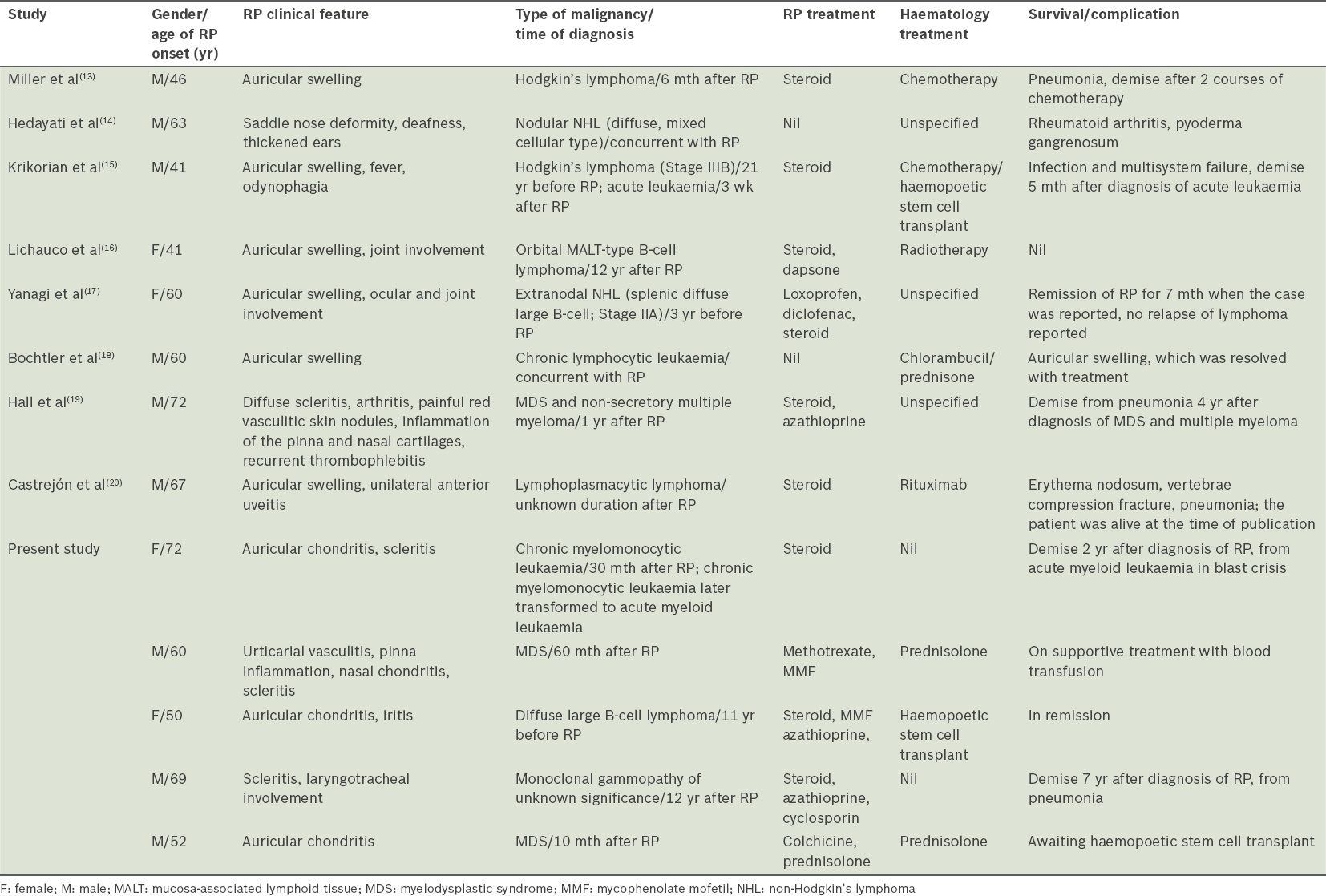
A possible explanation for the pathophysiology underlying the association between RP and lymphoma may be inferred from the pathophysiology of rheumatoid arthritis (RA), as RA is the archetypal autoimmune-mediated inflammatory disease. A meta-analysis of 21 publications demonstrated a twofold increase in lymphoma risk among patients with RA as compared to the general population.(9) In another large case-control study, Baecklund et al showed that RA patients with the highest inflammatory disease activity have a considerably increased risk of lymphoma.(10) That study also concluded that treatment with the most common disease-modifying antirheumatic drugs is in itself not a risk factor for RA-associated lymphomas. Similarly, the use of biologic response modifiers among patients with RA was not significantly associated with an increased risk of malignancy when compared with other disease-modifying antirheumatic drugs or placebos.(11)
The link between MDS and RP is presently unclear. A prospective study conducted on MDS patients with autoimmune manifestations did not identify chromosome aberrations that are typically associated with therapy-related MDS.(12) Nonetheless, there is a distinct subset of RP patients who develop haematological malignancies through an unknown pathophysiological process. Future research in this area may include studies examining whether such patients have clonal abnormalities that predispose them to leukaemic transformation.
The increased risk of malignancy among patients with autoimmune disease may be driven by a combination of factors, including sustained inflammatory burden, immunological defects, and personal and environmental exposure such as smoking and some viral infections.(21,22) For example, it has been postulated that sustained excessive salivary and lacrimal gland B-cell stimulation, and impaired B-cell apoptosis in patients with Sjögren’s syndrome may lead to tumorigenesis and clonal expansion of B-cells that ultimately leads to the development of lymphomas.(21) The relationship between chronic inflammation and carcinogenesis in RP patients may be elucidated by studying inflammatory markers such as plasma cytokines, lipid peroxides, p53, BAX, cyclins and caspases.
In conclusion, we found an unexpectedly high frequency of RP patients with haematological malignancies in our series. Although MDS has been reported to occur with RP, the association of other haematological malignancies with RP is rare. As the correlation between haematological malignancies and RP is currently unclear, we recommend that the threshold for haematological/lymphoproliferative screening be lowered in patients with RP.
ACKNOWLEDGEMENT
We thank our colleagues from the Department of Rheumatology and Immunology, Singapore General Hospital, Singapore, for contributing their cases to this study.


