Abstract
INTRODUCTION
International normalised ratio (INR) control is an important factor in patients with non-valvular atrial fibrillation (NVAF) being treated with warfarin. INR control was previously reported to be poorer among Asians compared to Westerners. We aimed to validate the SAMe-TT2R2 score for prediction of suboptimal INR control (defined as time in therapeutic range [TTR] < 65% in the Thai population) and to investigate TTR among Thai NVAF patients being treated with warfarin.
METHODS
INR data from patients enrolled in a multicentre NVAF registry was analysed. Clinical and laboratory data was prospectively collected. TTR was calculated using the Rosendaal method. Baseline data was compared between patients with and without suboptimal INR control. Univariate and multivariate analyses were performed to identify variables independently associated with suboptimal INR control.
RESULTS
A total of 1,669 patients from 22 centres located across Thailand were included. The average age was 69.1 ± 10.7 years, and 921 (55.2%) were male. The mean TTR was 50.5% ± 27.5%; 1,125 (67.4%) had TTR < 65%. Univariate analysis showed hypertension, diabetes mellitus, heart failure, renal disease and SAMe-TT2R2 score to be significantly different between patients with and without optimal TTR. The SAMe-TT2R2 score was the only factor that remained statistically significant in multivariate analysis. The C-statistic for the SAMe-TT2R2 score in the prediction of suboptimal TTR was 0.54.
CONCLUSION
SAMe-TT2R2 score was the only independent predictor of suboptimal TTR in NVAF patients being treated with warfarin. However, due to the low C-statistic, the score may have limited discriminative power.
INTRODUCTION
The annual incidence of ischaemic stroke in patients with non-valvular atrial fibrillation (NVAF) was reported to be approximately 5%.(1) Antithrombotic therapy is an acknowledged first-line treatment that both reduces mortality and improves quality of life by reducing the incidence of ischaemic stroke.(2) Although many practice guidelines recommend the use of non-vitamin K antagonists (NOACs) for stroke prevention in patients with NVAF,(2,3) warfarin remains the most prescribed anticoagulant drug for stroke prevention, especially in Asian countries, due to its relative affordability.(4) As a complication of warfarin therapy, intracerebral haemorrhage is more common among Asian populations than Western populations.(5) For those who receive warfarin, efficacy of treatment for stroke prevention depends on the level of international normalised ratio (INR) control, measured as time in therapeutic range (TTR).(6) It has been recommended that the minimum TTR should be 60%–70%.(2,7,8) Data from the Global Anticoagulant Registry in the FIELD-Atrial Fibrillation (GARFIELD-AF registry), the world’s largest cohort of patients with newly diagnosed atrial fibrillation, revealed that level of INR control (as reflected by TTR) was associated with bleeding and thromboembolic complications, and that the Asian population had a substantially lower TTR compared to other regions of the world (31% vs. 54%) and a greater proportion of patients with INR lower than 2 (59% vs. 28%).(6)
SAMe-TT2R2 score has been shown to be a good predictor of suboptimal INR control.(9) Component factors include female gender, age < 60 years, medical history (i.e. hypertension, diabetes mellitus, coronary artery disease, peripheral arterial disease, heart failure, stroke, pulmonary disease, hepatic and renal disease), interacting drugs, tobacco use and ethnicity (non-Caucasian).(9) Given that non-Caucasian status is immediately assigned 2 points, SAMe-TT2R2 scores in Asian populations tend to be high. A recent review showed that SAMe-TT2R2 scores may be useful to aid in the clinical decision of anticoagulant use.(10) However, data from Asian populations is limited. Hence, this study aimed to validate the SAMe-TT2R2 score for prediction of suboptimal INR control (defined as TTR < 65% in the Thai population) and to investigate TTR among Thai NVAF patients being treated with warfarin.
METHODS
The study protocol was approved by the institutional review board of each participating hospital. Written informed consent was obtained from all participating patients. Patients were enrolled during the 2014–2017 study period from 22 hospitals located across Thailand: 12 of those hospitals were university hospitals and ten were regional or general hospitals. The study sites were distributed across all regions in the country. We encouraged investigators at each site to enrol consecutive cases.
Patients who were enrolled were: older than 18 years of age; had NVAF diagnosed by electrocardiography or Holter monitoring; were taking warfarin; and were included in the COOL-AF Thailand (
The following data was collected: medical history including risk factors for ischaemic stroke (CHA2DS2-VASc score) and bleeding (HAS-BLED score); history of ischaemic stroke; history of bleeding; blood pressure and heart rate; electrocardiogram and echocardiogram; antithrombotic drugs and other medications; and laboratory data, including INR. We recorded the data from real-world practice. The investigator selected the choice of antithrombotic treatment. In the case record form of the main study, investigators had to record their target INR for each patient. Those with target INR outside the range of 2–3, as indicated by the physician, were excluded from this study. Each component of the [CHA2DS2-VASc] score was scored and recorded as follows: C = congestive heart failure (1 point); H = hypertension (1 point); A = age > 75 years (2 points); D = diabetes mellitus (1 point); S = stroke (2 points); V = vascular disease (1 point); A = age 65–74 years (1 point); and Sc = female sex category (1 point).
Each component of the HAS-BLED score was scored and recorded, giving 1 point each for uncontrolled hypertension, abnormal renal or liver function; history of stroke; history of bleeding; labile INR; elderly age (> 65 years); and taking drugs or alcohol. Each component of the SAMe-TT2R2 score was recorded as follows: S = female sex (1 point); A = age < 60 years (1 point); Me = medical history consisting of at least two conditions: diabetes mellitus, hypertension, heart failure, coronary artery disease, peripheral arterial disease, previous stroke, pulmonary disease, and hepatic or renal disease (1 point); T = treatment with interacting drugs such as amiodarone, nonsteroidal anti-inflammatory drugs, antifungal medications and many antibiotics (1 point); T = tobacco use in two years (2 points); and R = non-white race (2 points). Renal disease was defined based on laboratory data within the last six months as glomerular filtration rate < 60 mL/min, according to the Chronic Kidney Disease Epidemiology Collaboration formula, or as a diagnosis of chronic kidney disease or end-stage kidney disease in medical records, excluding the setting of acute kidney injury. Hepatic disease was defined as an alanine aminotransferase level of more than twice the upper normal limit, based on laboratory data within the last six months or a diagnosis of chronic liver disease or cirrhosis in the medical record, excluding the setting of acute liver injury. Pulmonary disease was defined as a diagnosis of chronic lung disease or chronic lung infection in the medical records. TTR was calculated for each patient using the Rosendaal method(11) and defined as the percentage of time the INR result was between 2 and 3. We defined suboptimal INR control as TTR < 65%.(8,12)
All data was collected in the case record form and keyed into a Web-based system. The investigator sent the case record form to the central site for data verification. The central data management site performed double data entry, verified the data and sent a query to study a site if needed. Site monitoring was performed in approximately 70% of study sites for quality control of study data.
Data was analysed using IBM SPSS Statistics version 22.0 (IBM Corp, Armonk, NY, USA). Continuous data was presented as mean ± standard deviation and categorical data as number and percentage. Student’s t-test was used to compare continuous unpaired data, and chi-square test was used to compare categorical data. Univariate and multivariate logistic regression analysis was performed to identify factors associated with suboptimal INR control, and those results were described as odds ratio (OR) and 95% confidence interval (CI). Variables with a p-value < 0.2 in univariate analysis were included in multivariable analysis. A p-value < 0.05 was regarded as being statistically significant.
RESULTS
Among 2,800 patients enrolled in the COOL-AF study, 1,919 (68.5%) received warfarin with or without antiplatelet therapy, 154 (5.5%) received NOAC with or without aspirin, 513 (18.3%) received antiplatelet therapy alone, and 214 (7.6%) had no antithrombotic drugs. Among the 1,919 patients who were on warfarin, 1,669 patients had enough INR data to calculate TTR and were included in this study. The other 250 patients could not be included as warfarin was discontinued for 113 (5.9%) patients and follow-up data was not available for 137 (7.1%) patients because of their short duration of follow-up.
The average age of the 1,669 patients was 69.1 ± 10.7 years, and 921 (55.2%) were male (
Table I
Baseline characteristics of patients with TTR ≥ 65% compared with those of patients with TTR < 65%.
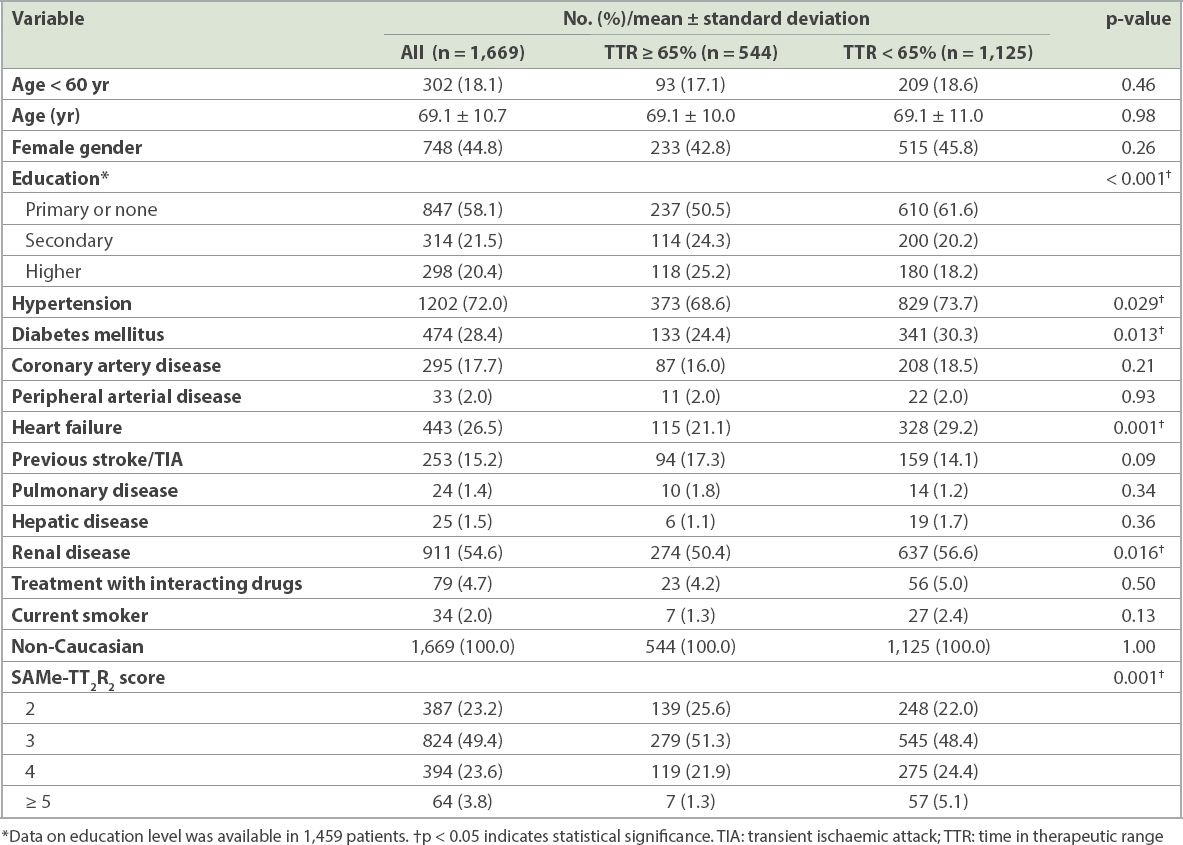
The results of univariate and multivariable analyses are shown as a forest plot in
Fig. 1
Forest plot shows univariate and multivariable analysis of factors associated with suboptimal TTR control. CI: confidence interval; OR: odds ratio; TIA: transient ischaemic attack; TTR: time in therapeutic range
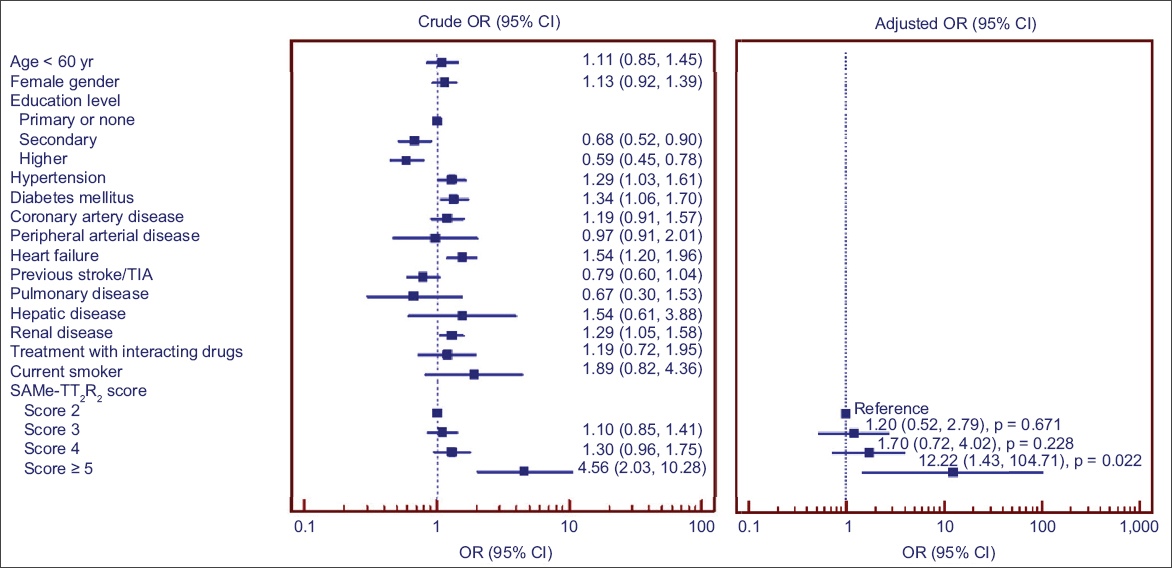
Fig. 2
Box plot shows TTR distribution stratified by SAMe-TT2R2 score group. The table shows the proportion of patients with TTR ≥ 65% for each SAMe-TT2R2 score. TTR: time in therapeutic range
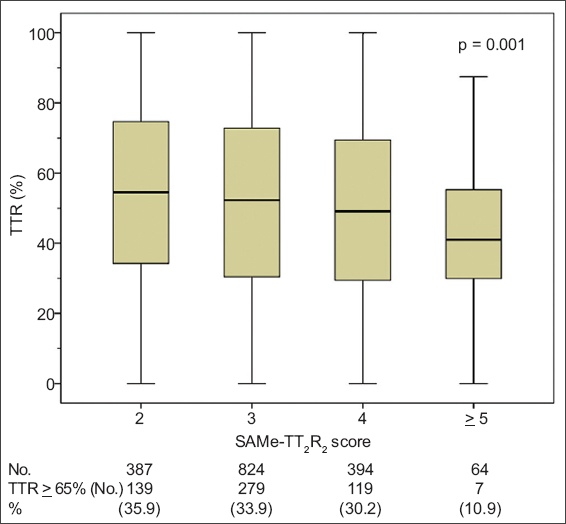
Table II
SAMe-TT2R2 score for the prediction of time in therapeutic range < 65%.
DISCUSSION
The main results of this study showed that SAMe-TT2R2 score was significantly associated with suboptimal INR control. The best cut-off of the SAMe-TT2R2 score (> 3) can be used to predict a TTR < 65%. However, due to the low C-statistic, the score may not be sensitive enough to discriminate well in the Asian population being studied.
As high TTR was associated with a good outcome,(8) practice guidelines emphasise the assessment of TTR in NVAF patients who are on warfarin.(2) However, previous studies showed the level of TTR control to be lower in Asian than in Western populations.(8,13) Post-hoc analysis of data from the 2006 ACTIVE-W (
Data from a global registry indicated that INR readings were within the range of 2.0–3.0 in 34%–38% of patients in Asian countries, 67% of patients in Western Europe, 59% of patients in Eastern Europe, and 54% of patients in North America.(14) Moreover, the median of INR reading in the GARFIELD registry was 1.8 (interquartile range [IQR] 1.4–2.3) for Asian regions compared to 2.3 (IQR 1.9 to 2.8) for non-Asian regions.(6) Data from our study revealed that 32.6% of Thai NVAF patients taking warfarin had TTR ≥ 65%. One of the possible reasons our INR control data is better than the Asian data from the GARFIELD study and the global AF registry may be that our data reflects the adoption of recent practice guidelines for management of patients with NVAF. As shown in the GARFIELD registry, the proportion of NVAF patients taking anticoagulants has increased from Cohort 1 (2010–2011) to Cohort 4 (2014–2015), and this is likely due to the same reason. The publication of many practice guidelines for stroke prevention in atrial fibrillation,(2,3) including new guidelines from the Asia Pacific Heart Rhythm Society(7) and new guidelines in Asian countries such as Taiwan,(15) Korea(16) and Japan,(17) have led to an increased rate of antithrombotic use in NVAF.(7) These guidelines focus not only on the use of anticoagulant in the ‘not low-risk’ group, but also recommended a high TTR in patients on warfarin in order to achieve a good outcome.(2) It should also be noted that all principal investigators in the present study are cardiologists, which should lead to better anticoagulant control. In this NOAC era, guidelines from Taiwan prefer the use of NOACs;(15) as shown in a recent publication, 66.4% of those who used anticoagulant utilised NOACs instead of warfarin.(18)
There are many possible reasons for poorer TTR control in Asians as compared to Westerners. A fear of bleeding is one of the major reasons due to the increased risk (up to four times) of intracranial haemorrhage in Asians who are on warfarin compared to Westerners.(5) A study from Singapore described other common reasons given by physicians, that patients are too old and that they frequently fall.(19) Many physicians, therefore, prefer to maintain a lower INR than the recommended level. Hence, factors related to both physicians and patients contribute to poorer warfarin control. In addition, a significant proportion of patients with NVAF take herbal medications that can affect INR control. Genetic predisposition may be another factor: the proportion of CYP2C9 and VKORC1 polymorphism has been reported to be different in Asian compared to Western populations.(20) It remains unclear how much influence this genetic factor has on suboptimal INR control in Asian populations, although data from a pharmacokinetic and pharmacodynamic study in Chinese and Indians showed warfarin response to be influenced by the VKORC1 haplotypes.(21) Data from a randomised study on a Chinese population showed that a lower INR target within the range of 1.6–2.0 may be used due to the lower rate of bleeding without a significant increase in the rate of stroke compared to an INR target range of 2.1–2.6.(22) This data is somewhat different from data from Western populations. In general, the recommendation for INR target is still 2.0–3.0, even in Asian populations.(7) The high proportion of patients with TTR < 65% in this study may be related to the fact that not all patients (90.2%) had a target INR of 2–3.
Data on INR control among regions may differ between clinical trials and real-world settings. In the Randomized Evaluation of Long Term Anticoagulant Therapy (RE-LY) study, TTR from Thailand, China and India was 56%, 55% and 49%, respectively.(13) However, TTR results from the RE-LY registry of 15,400 NVAF patients in 46 countries from nine geographic regions indicated that the TTR in Southeast Asia, China and India was 36%, 35.5% and 33.7%, respectively.(13) This data discrepancy emphasises the importance of suboptimal INR control in our regular practice and suggests that results from clinical trials may not reflect real-world data. Although TTR results from Asian populations have been reported in NOAC clinical trials (i.e. RE-LY 56.5%, ROCKET 52.4% and ARISTOTLE 60.0%), real-world INR control could be much worse.(13)
Asian patients have a SAMe-TT2R2 score of 2 on the basis of their ethnicity alone, which indicates a significant increase in the risk of suboptimal INR.(9) It is recommended that patients with a SAMe-TT2R2 score > 2 have regular INR checks and apply strategies to improve TTR.(2) Since the risk of stroke in patients who are on warfarin is highly dependent on their level of INR control,(12) a tool like the SAMe-TT2R2 score, if valid in Asian populations, could be helpful to determine which patients would do well on warfarin. A recent review indicated that the SAMe-TT2R2 score had a C-statistic of 0.52–0.72 for the prediction of suboptimal INR control in eight studies,(10) compared to 0.54 in our study. Most studies defined poor INR control as TTR < 65% or 70%. The eight studies also investigated the association of SAMe-TT2R2 score with clinical events, with five studies showing some positive correlation.(10)
To our knowledge, ours is the third study to attempt to validate the SAMe-TT2R2 score in Asian populations, after previous studies from Hong Kong(23) and Singapore.(24) The earlier studies used retrospective data collection with data from a single centre, whereas our study collected data prospectively from a registry from 22 centres in Thailand. There were 1,428 participants in the Hong Kong study, 1,137 in the Singapore study and 1,669 in our study. The report from Hong Kong included outcome data, but this was not reported in the papers from Singapore and the present study. Furthermore, the study from Hong Kong showed that SAMe-TT2R2 score > 2 predicted poor TTR, defined as TTR < 70%, and was associated with an increased risk of stroke. The Singapore study found that the TTR of patients with SAMe-TT2R2 score > 2 was lower than that of patients with score ≤ 2. Our study showed that SAMe-TT2R2 score > 3 had the best C-statistic for the prediction of poor TTR, defined as TTR < 65%, and the C-statistic was highest for a TTR < 65% compared to < 60% and < 70%. However, the highest C-statistic was 0.54, which is low and reflects poor prediction of suboptimal TTR using the SAMe-TT2R2 score. Therefore, use of the SAMe-TT2R2 score in clinical routine is limited.
Some experts have commented on the external validity of the original paper on the SAMe-TT2R2 score,(9) stating that its discriminative power is lacking and that it should not be used in daily practice.(25) A recent systematic review on 16 studies summarised that although the SAMe-TT2R2 score can predict low TTR, the effect is too small to be clinically useful.(26) In our study, 67.4% of the patients had TTR < 65%, which is considered suboptimal. Even at a SAMe-TT2R2 score of 2, 64.1% of the patients had TTR < 65%, indicating that the discriminative power of the score is limited; this correlated with the low C-statistic.
The present study has some limitations. As our study population was mainly recruited from tertiary care or large regional hospitals, we may not be able to extrapolate the findings to the NVAF population at large. Another limitation is that the data on clinical outcomes is not yet complete, and hence we could not correlate the main finding with clinical outcomes. Nevertheless, one of the strengths of this study is the double data entry and data checking.
In conclusion, the SAMe-TT2R2 score was identified as the only independent predictor of suboptimal TTR in NAVF patients being treated with warfarin. However, due to the low C-statistic, the score may not be sensitive enough to discriminate in the Asian population being studied.
ACKNOWLEDGEMENTS
The authors gratefully acknowledge the patients who generously agreed to participate in this study. The authors would also like to thank Pontawee Kaewcomdee, Olaree Chaiphet, Wipaporn Wangworatrakul, and Wilasinee Phromawan from Siriraj Hospital, Mahidol University, Thailand, for data management, and all investigators and research nurses for their assistance with patient enrolment and their commitment to maintaining a high level of data quality. This study was funded by grants from the Health Systems Research Institute (HSRI, 59-053), the Heart Association of Thailand under the Royal Patronage of H.M. the King, and the Royal College of Physicians of Thailand.


